Important Notes
HL7 licenses its standards and select IP free of charge. If you did not acquire a free license from HL7 for this document, you are not authorized to access or make any use of it. To obtain a free license, please visit http://www.HL7.org/implement/standards/index.cfm.
If you are the individual that obtained the license for this HL7 Standard, specification or other freely licensed work (in each and every instance "Specified Material"), the following describes the permitted uses of the Material.
A. HL7 INDIVIDUAL, STUDENT AND HEALTH PROFESSIONAL MEMBERS, who register and agree to the terms of HL7’s license, are authorized, without additional charge, to read, and to use Specified Material to develop and sell products and services that implement, but do not directly incorporate, the Specified Material in whole or in part without paying license fees to HL7.
INDIVIDUAL, STUDENT AND HEALTH PROFESSIONAL MEMBERS wishing to incorporate additional items of Special Material in whole or part, into products and services, or to enjoy additional authorizations granted to HL7 ORGANIZATIONAL MEMBERS as noted below, must become ORGANIZATIONAL MEMBERS of HL7.
B. HL7 ORGANIZATION MEMBERS, who register and agree to the terms of HL7's License, are authorized, without additional charge, on a perpetual (except as provided for in the full license terms governing the Material), non-exclusive and worldwide basis, the right to (a) download, copy (for internal purposes only) and share this Material with your employees and consultants for study purposes, and (b) utilize the Material for the purpose of developing, making, having made, using, marketing, importing, offering to sell or license, and selling or licensing, and to otherwise distribute, Compliant Products, in all cases subject to the conditions set forth in this Agreement and any relevant patent and other intellectual property rights of third parties (which may include members of HL7). No other license, sublicense, or other rights of any kind are granted under this Agreement.
C. NON-MEMBERS, who register and agree to the terms of HL7’s IP policy for Specified Material, are authorized, without additional charge, to read and use the Specified Material for evaluating whether to implement, or in implementing, the Specified Material, and to use Specified Material to develop and sell products and services that implement, but do not directly incorporate, the Specified Material in whole or in part.
NON-MEMBERS wishing to incorporate additional items of Specified Material in whole or part, into products and services, or to enjoy the additional authorizations granted to HL7 ORGANIZATIONAL MEMBERS, as noted above, must become ORGANIZATIONAL MEMBERS of HL7.
Please see http://www.HL7.org/legal/ippolicy.cfm for the full license terms governing the Material.
Ownership. Licensee agrees and acknowledges that HL7 owns all right, title, and interest, in and to the Materials. Licensee shall take no action contrary to, or inconsistent with, the foregoing.
Licensee agrees and acknowledges that HL7 may not own all right, title, and interest, in and to the Materials and that the Materials may contain and/or reference intellectual property owned by third parties (“Third Party IP”). Acceptance of these License Terms does not grant Licensee any rights with respect to Third Party IP. Licensee alone is responsible for identifying and obtaining any necessary licenses or authorizations to utilize Third Party IP in connection with the Materials or otherwise. Any actions, claims or suits brought by a third party resulting from a breach of any Third Party IP right by the Licensee remains the Licensee’s liability.
Following is a non-exhaustive list of third-party terminologies that may require a separate license:
| Terminology | Owner/Contact |
|---|
| Current Procedures Terminology (CPT) code set | American Medical Association
https://www.ama-assn.org/practice-management/cpt-licensing |
| SNOMED CT© | SNOMED CT® International http://www.snomed.org/snomed-ct/get-snomed-ct or info@ihtsdo.org |
| Logical Observation Identifiers Names & Codes (LOINC©) | Regenstrief Institute, Inc. |
| International Classification of Diseases (ICD) codes | World Health Organization (WHO) |
| NUCC Health Care Provider Taxonomy code set | American Medical Association. Please see www.nucc.org. AMA licensing contact: 312-464-5022 (AMA IP services) |
Obtaining a CPT Sublicense from HL7
Contact hq@hl7.org about how to obtain a sublicense from HL7 for non-production use of CPT for (i) the development and publication of value sets, profiles, and other artifacts as part of the HL7 Implementation Guides, (ii) as part of defined VSAC value sets, and (iii) to support HL7's terminology services within the Territory.
Flow Down Clauses for CPT Sublicense from HL7
CPT content is copyrighted by the American Medical Association and CPT is a registered trademark of the AMA.
HL7, as a party to a license agreement with the AMA, is authorized to grant user a limited, non-exclusive, non-transferable, non-sublicensable license for user to use CPT content for (i) the development and publication of value sets, profiles, and other artifacts as part of the HL7 Implementation Guides, (ii) as part of defined VSAC value sets, and (iii) to support HL7's terminology services within the Territory, each of which shall be considered a non-production use. The sublicense granted hereunder shall automatically terminate upon termination of the agreement between HL7 and AMA, unless prior written consent of AMA is obtained.
The provision of updated CPT content is dependent on a continuing contractual relationship between HL7 and the AMA.
User acknowledge a separate license agreement shall be required, and shall govern any proposed use, including any distribution of CPT content for any other purposes not expressly permitted under this Agreement, and the terms of such agreement will govern such use (e.g., a separate license agreement shall govern production use and commercial purposes). AMA reserves the right to accept or reject licenses based on AMA's evaluation of the proposed use of the CPT content.
User acknowledge that User's development and commercialization of CPT-informed works developed with reference to Licensed Products may only be implemented in the Territory.
User is prohibited from making CPT content publicly available, creating derivative works (including translating), transferring, selling, leasing, licensing, or otherwise making available to any unauthorized party the CPT content, or a copy or portion of CPT content to any unauthorized party, including a subsidiary, affiliate, or other legal entity, however designated, for any purpose whatsoever except as expressly permitted under a separate agreement.
User expressly acknowledges and agrees to the extent permitted by applicable law, use of CPT content is at User's sole risk and CPT content is provided "as is" without warranty of any kind. The AMA does not directly or indirectly practice medicine or dispense medical services. Fee schedules, relative value units, conversion factors and/or related components are not assigned by the AMA, are not part of CPT, and the AMA is not recommending their use. CPT content herein does not replace the AMA's Current Procedural Terminology book or other appropriate coding authority. The coding information contained in CPT content should be used only as a guide.
U.S. Government End Users. CPT is commercial technical data, which was developed exclusively at private expense by the American Medical Association (AMA), 330 North Wabash Avenue, Chicago, Illinois 60611. This agreement does not grant the Federal Government a direct license to use CPT based on FAR 52.227- 14 (Data Rights - General) and DFARS 252.227-7015 (Technical Data - Commercial Items).
User expressly consents to the release of its name to the AMA.
| Role
|
Name
|
Organization
|
Contact
|
| Primary Editor
|
Giorgio Cangioli
|
Consultant, HL7 Italy
|
giorgio.cangioli@gmail.com
|
| Primary Editor
|
Rob Hausam
|
Hausam Consulting LLC
|
rob@hausamconsulting.com
|
| Primary Editor
|
Dr Kai U. Heitmann
|
Heitmann Consulting and Services, ART-DECOR Open Tools GmbH, HL7 Germany
|
info@kheitmann.de
|
| Primary Editor
|
François Macary
|
Phast
|
francois.macary@phast.fr
|
| Contributor
|
Dr Philip Scott
|
HL7 UK
|
philip.scott@uwtsd.ac.uk
|
| Contributor
|
Dr Christof Geßner
|
HL7 Germany
|
christof.gessner@mxdx.de
|
| Contributor
|
Dr Stefan Sabutsch
|
ELGA, HL7 Austria
|
stefan.sabutsch@elga.gv.at
|
| Contributor
|
Gary Dickinson
|
CentriHealth
|
gary.dickinson@ehr-standards.com
|
| Contributor
|
Catherine Chronaki
|
HL7 International Foundation
|
chronaki@gmail.com
|
| Contributor
|
Dr Stephen Chu
|
HL7 Australia
|
chuscmi88@gmail.com
|
| Contributor
|
Didi Davis
|
The Sequoia Project
|
ddavis@sequoiaproject.org
|
| Contributor
|
George Dixon
|
Allscripts LLC
|
george.dixon@allscripts.com
|
| Contributor
|
Kenneth Sinn
|
Ontario Health Digital Services
|
ken.sinn@ontariohealth.ca
|
| Contributor
|
John D'Amore
|
More Informatics
|
johnd@moreinformatics.com
|
Thanks to Alexander Berler (a.berler@gnomon.com.gr); Carina Seerainer (carina.seerainer@elga.gv.at); John Roberts (John.A.Roberts@tn.gov); Julie James (julie_james@bluewaveinformatics.co.uk); Mark Shafarman (mark.shafarman@earthlink.net); Fernando Portilla (fportila@gmail.com); Ed Hammond (william.hammond@duke.edu); Steve Kay (s.kay@histandards.net).
Preface to this STU ballot
The goal of this Implementation Guide is to specify how to represent in HL7 CDA R2 the International Patient Summary (IPS) (see also the ART-DECOR repository). An alternative representation as HL7 FHIR is also provided (see the HL7 build site).
The initial focus of the International Patient Summary (IPS) was the unplanned care across national borders. This specification can be used and be useful also in local applications and be supportive of planned care.
Compared to the previous STU specification, developments took place mainly around additional data to be conveyed (Alert, Patient Story), input that comes from the underlying ISO Standard 27269:2021 Health informatics — International patient summary and its updates. In addition, terminology aspects have been discussed and further developed. The terminology changes are listed in details below. With a few exceptions, FHIR and CDA terminologies used are the same. One exception is the update Allergy and Intolerances Value Set, in FHIR made for binding at one place in the associated profile but in CDA these are still two Value Sets as substances and conditions are bound to different elements in CDA.
Here is the list of change categories:
- Medications updated due to new Value Sets
- Problem Entry updated
- Procedure updated due to new Value Sets, especially Certainty
- Immunization updated due to new Value Sets
- Specimen Collection updated due to new Value Sets
- Allergy and Intolerances have been updated re/ Value Sets, defined for substances and conditions
- Laboratory/Pathology updated due to new Value Sets
- Radiology updated due to new Value Sets,
- Radiology added the result component accordingly as modelled in FHIR
- Social History (Smoking) updated due to new Value Sets
- Pregnancy updated, especially the Estimated Delivery Date Observation
- New section: Alert
- New section: Patient Story
- A new Document Level Template has been created (OID 2.16.840.1.113883.10.22.1.2) to address all changes,
The Template Definition of the document level template in ART-DECOR can be found here https://art-decor.org/ad/#/hl7ips-/rules/templates/2.16.840.1.113883.10.22.1.2/2024-08-02T13:02:39. From that all other header, section and entry level templates are listed.
Expressing HL7 CDA® R2 template definitions as FHIR Structure Definitions
As a pilot efforts have been made to convert the definitions of the HL7 CDA® R2 Implementation Guide of the International Patient Summary, that were done in ART-DECOR using the former HL7 Templates Standard: Specification and Use of Reusable Information Constraint Templates, in short Templates ITS into FHIR Structure Definitions and to use the FHIR IG publisher for the CDA IG. This is still experimental and offered as an informational IG, reachable under HL7 build site.
Introduction
An International Patient Summary (IPS) document is an electronic health record extract containing essential healthcare information about a subject of care. As specified in EN ISO 27269, it is designed for supporting the use case scenario for ‘unplanned, cross border care’, but it is not limited to it. It is intended to be international, i.e., to provide generic
solutions for global application beyond a particular region or country.
The IPS dataset is minimal and non-exhaustive; specialty-agnostic and condition-independent; but still clinically relevant.
The IPS document is composed by a set of robust, well-defined and potentially reusable sets of core data items (indicated as IPS library in the figure below). The tight focus of the IPS on unplanned care is in this case not a limitation, but, on the contrary, facilitates their potential re-use beyond the IPS scope.
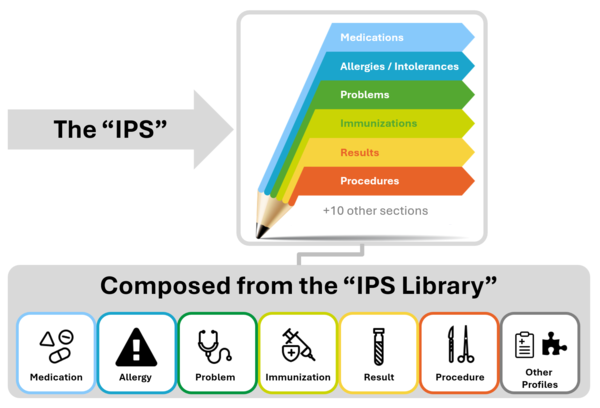
Figure 1: The IPS product and by-products
Purpose
The goal of this Implementation Guide is to specify how to represent in HL7 CDA the International Patient Summary (IPS). An alternative representation as templated HL7 FHIR R2 is also provided ( see the hl7.org site ). The initial focus of the International Patient Summary (IPS) was the unplanned care across national borders. This specification can be used and be useful also in local applications and be supportive of planned care.
Scope
As specified in EN ISO 27269, the IPS dataset is a “minimal, non-exhaustive set of data elements required for the international patient summary”. A Patient Summary is defined by ISO/TR 12773-1:2009 as a “Health record extract comprising a standardized collection of clinical and contextual information (retrospective, concurrent, prospective) that
provides a snapshot in time of a subject of care’s health information and healthcare.”
‘Minimal’ reflects the ideas of ‘summary’ and the need to be concise, but also alludes to the existence of a core set of data elements that all health care professionals can use; it is intended to be a specialty agnostic and condition independent set. It does not imply that all the items in the data set will be used in every summary. It is also possible to refine the extract
from a record such that the content of the summary is more relevant to a particular condition (e.g. asthma) but no asthma-specific elements will be specified in this standard. The IPS Document or IPS can be extended by non-IPS standard condition-specific data. ‘Non-exhaustive’ recognizes that the ideal data set is not closed, and is likely to be extended, not just in terms
of requirement evolution, but also pragmatically in instances of use. [EN ISO 27269].
Furthermore the scope of the IPS is global. Although this is a major challenge, this implementation guide takes various experiences and newer developments into account to address, as far as possible, global feasibility.
The following picture provides an overview of the current IPS content.
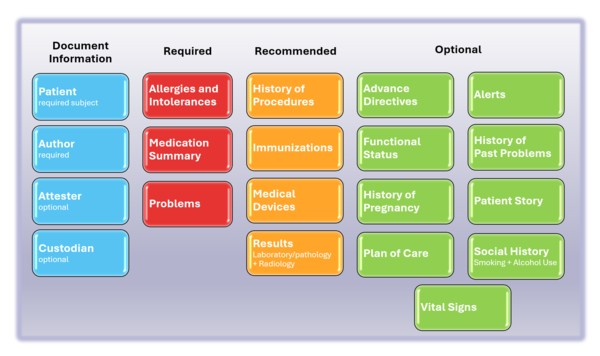
Figure 2: The IPS composition
Project Background and relationships with other projects
Details on the project background and relationships with other projects are available in the IPS Website.
Ballot Status of the Document
This Implementation Guide is STU with the intention to go normative.
Audience
The audience for this Implementation Guide includes:
Public
-
Citizens who want to carry or access their healthcare data for emergency or unplanned care purposes.
Regulatory
-
Policy makers such as healthcare payers or government agencies.
-
Healthcare information governance authorities and regulatory bodies.
Clinical
-
Healthcare providers that offer unscheduled and emergency care.
-
Healthcare providers that populate regional and national patient summaries.
Technical
-
Vendors of EHR systems for unplanned care management, personal health records and mobile health data applications.
-
System integrators.
-
Organizations that manage regional and national patient summaries.
Reading Publication Artifacts
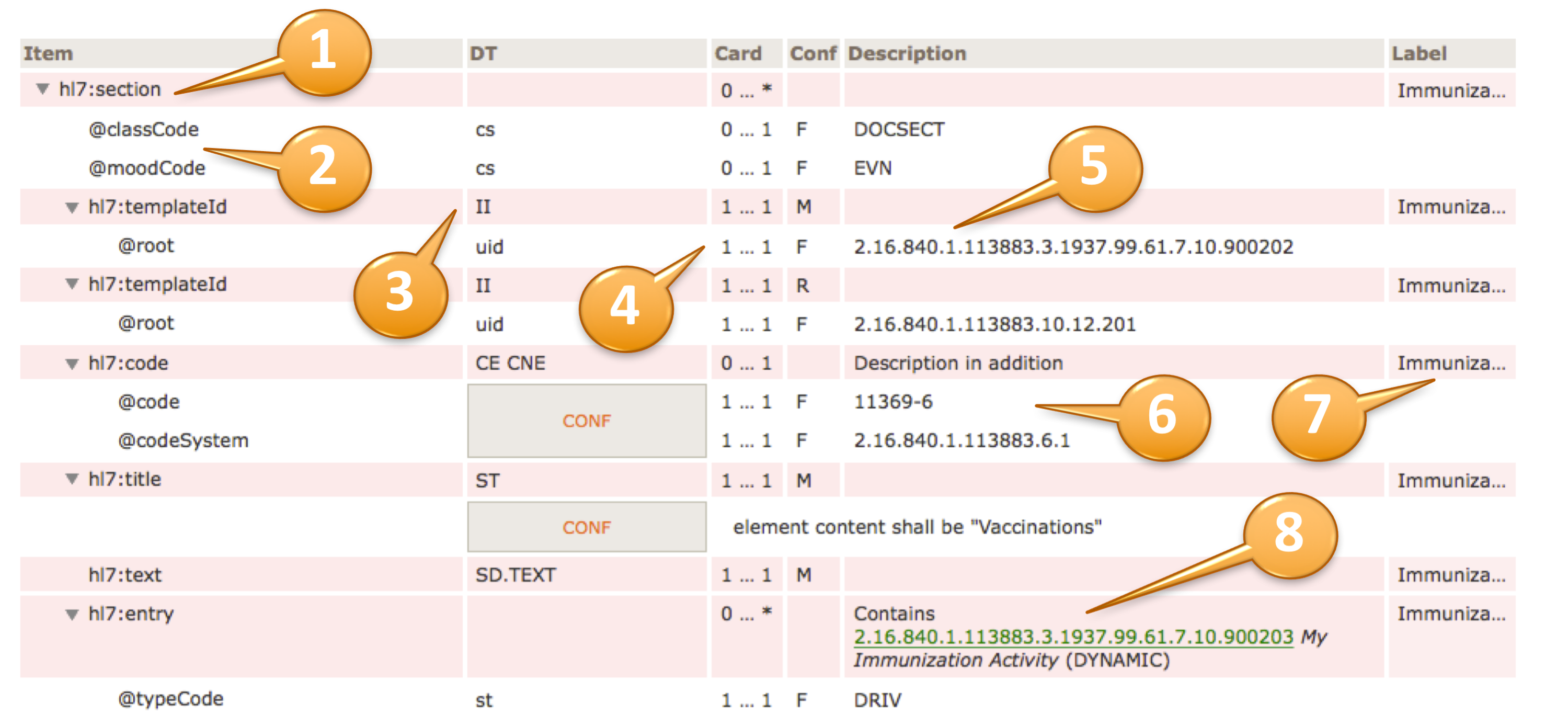
A reading guide is available that explains the formalism used to express the publication artifacts, i.e. template meta data and template design. For convenience the guide is included in the appendix. (see section "How to read the table view for templates")
Technical Background
What is a CDA
CDA R2 is
… a document markup standard that specifies the structure and semantics of clinical documents for the purpose of exchange.
[CDA R2 Standard http://www.hl7.org/implement/standards/product_brief.cfm?product_id=7, Section 1.1]
Clinical documents, according to CDA, have the following characteristics:
-
Persistence
-
Stewardship
-
Potential for authentication
-
Context
-
Wholeness
-
Human readability
CDA defines a header for classification and management and a document body that carries the clinical record. While the header metadata are prescriptive and designed for consistency across all instances, the body is highly generic, leaving the designation of semantic requirements to implementation.
Templated CDA
CDA R2 can be constrained by mechanisms defined in the “Refinement and Localization” section of the HL7 Version 3 Interoperability Standards. The mechanism most commonly used to constrain CDA is referred to as “templated CDA”. This specification created a set of artifacts containing modular CDA templates (and associated value sets) for the purpose of the International Patient
Summary, and the templates can be reused across any number of CDA document types.
There are different kinds of templates that might be created. Among them, the most common ones are:
- CDA Document Level Templates constrain fields in the Clinical Document Architecture (CDA) header, and define containment relationships to CDA sections.
For example, a History-and-Physical document-level template might require that the patient’s name be present, and that the document contain a Physical Exam section.
-
CDA Header Level Templates constrain fields for parts of the CDA header, like the patient (record target), the author, participations or the service event.
-
CDA Section Level Templates constrain fields in the CDA section, and define containment relationships to CDA entries.
For example, a Physical-exam section-level template might require that the section/code be fixed to a particular LOINC code, and that the section contain a Systolic Blood Pressure observation.
-
CDA Entry Level Templates constrain the CDA clinical statement model in accordance with real world observations and acts.
For example, a Systolic-blood-pressure entry-level template defines how the CDA Observation class is constrained (how to populate observation/code, how to populate observation/value, etc.) to represent the notion of a systolic blood pressure.
Open and Closed Templates
Open templates permit anything to be done in the underlying standard that is not explicitly prohibited. This allows templates to be built up over time that extend and go beyond the original use cases for which they were originally designed.
Closed templates only permit what has been defined in the template, and do not permit anything beyond that. There are good reasons to use closed templates, sometimes having to do with local policy. For example, in communicating information from a healthcare provider to an insurance company, some information may need to be omitted to ensure patient privacy laws are followed. Most
templates developed for CDA are of the open sort.
Template versioning
Template versioning is needed to enable template designs to evolve over time.
Template versioning enables template designers to control and shape the conformance statements that make up a template’s design over time tailoring the design to fit the template’s intended purpose.
Each template version is associated with a particular template. The template – as a whole – has a mandatory globally unique, non-semantic, identifier. The identifier serves as the identifier of the original intent of the template and as the identifier of the set of versions that represent the template over time.
Template versions have a mandatory timestamp (date and optional time), called the “effective date”. The date can be seen as the point in time when the template version “came into being”, i.e. was recognized as existent by the governance group. Use of the template prior to this date would be considered an invalid use of the template.
For further information on Templates, Template Versions and related topics refer to the HL7 Templates Standard [HL7 Templates Standard: Specification and Use of Reusable Information Constraint Templates, Release 1 http://www.hl7.org/implement/standards/product_brief.cfm?product_id=377].
Conformance Verbs
The conformance verb keywords SHALL, SHOULD, MAY and SHALL NOT in this document are to be interpreted as described in the HL7 Version 3 Publishing Facilitator's Guide [HL7 Version 3 Publishing Facilitator's Guide http://www.hl7.org/v3ballot/html/help/pfg/pfg.htm].
-
SHALL: an absolute requirement
-
SHALL NOT: an absolute prohibition against inclusion
-
SHOULD: best practice or recommendation. There may be valid reasons to ignore an item, but the full implications must be understood and carefully weighed before choosing a different course
-
MAY: truly optional; can be included or omitted as the author decides with no implications
Identifiers for Templates and Value Sets
This specification uses the following OIDs for the artifacts that are registered at the HL7 OID registry.
The sub branches for templates follow the recommendations of HL7 International and ISO 13582 [ISO/TS 13582:2015 Health informatics -- Sharing of OID registry information]
Terminologies
Note: Much of the description provided in this section is copied and adapted from the §4.2.8 Vocabulary Conformance section of the C-CDA DSTU R2.1 Implementation Guide Volume 1 [HL7 C-CDA Implementation Guide DSTU R2.1
http://www.hl7.org/implement/standards/product_brief.cfm?product_id=379
]
The templates in this document use terms from several code systems. These vocabularies are defined in various supporting specifications and may be maintained by other bodies, as is the case for the LOINC® and SNOMED CT® vocabularies. The primary terminologies identified for this specification are listed in the Design conventions and principles, section "How to use terminologies
(preferred binding)".
Note that value set identifiers (e.g., ValueSet 2.16.840.1.113883.1.11.78 Observation Interpretation (DYNAMIC)) used in the binding definitions of template conformance statements do not appear in the XML instance of a CDA document. The definition of the template must be referenced to determine or validate the vocabulary conformance requirements of the template.
Value set bindings adhere to HL7 Vocabulary Working Group best practices, and include both an indication of stability and of coding strength for the binding. Value set bindings can be STATIC, meaning that they bind to a specified version of a value set, or DYNAMIC, meaning that they bind to the most current version of the value set. If a STATIC binding is specified, a date SHALL
be included to indicate the value set version. If a DYNAMIC binding is specified, the value set authority and link to the base definition of the value set SHALL be included, if available, so implementers can access the current version of the value set. When a vocabulary binding binds to a single code, the stability of the binding is implicitly STATIC.
For example, to convey @code=11450-4, the code’s displayName 'Problem List', the OID of the codeSystem from which the code is drawn '2.16.840.1.113883.6.1', and the codeSystemName 'LOINC', the tabular view used in this guide is presented as shown below.

Figure: Binding to a Single Code (tabular view)
HL7 Data Types Release 1 requires the codeSystem attribute unless the underlying data type is “Coded Simple” or “CS”, in which case it is prohibited. The displayName and the codeSystemName are optional, but recommended, in all cases.
The above example would be properly expressed as follows.
<code code="11450-4" codeSystem="2.16.840.1.113883.6.1"/>
<!-- or -->
<code code="11450-4" codeSystem="2.16.840.1.113883.6.1" displayName="Problem List" codeSystemName=”LOINC”/>
Figure: XML Expression of a Single-Code Binding
A full discussion of the representation of vocabulary is outside the scope of this document; for more information, see the HL7 V3 Normative Edition 2010 [HL7 V3 Normative Edition 2010 http://www.hl7.org/memonly/downloads/v3edition.cfm] sections on Abstract Data Types
and XML Data Types R1.
There is a discrepancy between the HL7 R1 Data Types and this guide in the implementation of translation code versus the original code. The R1 data type requires the original code in the root. The convention agreed upon for this implementation guide is that a code from the required value set is used in the element and other codes not included in the value set are to be
represented in a translation for the element. Note: This discrepancy is resolved in HL7 Data Types R2, but that is not available for use in CDA R2.
In the next example, the conformant code is SNOMED CT code 206525008.
<code code="206525008" codeSystem="2.16.840.1.113883.6.96"
displayName="neonatal necrotizing enterocolitis" codeSystemName="SNOMED CT">
<translation code="NEC-1" codeSystem="2.16.840.1.113883.19"
displayName="necrotizing enterocolitis"/>
</code>
Figure: Translation Code Example
Value set tables are present below a template, or are referenced if they occur elsewhere in the specification, when there are value set bindings in the template. The value set table provides the value set identifier, a description, and a link to the source of the value set when possible. Ellipses in the last row indicate the value set members shown are examples and the true
source must be accessed to see all members.
If a value set binding has a DYNAMIC stability, implementers creating a CDA document must go to the location in the URL to check for the most current version of the value set expansion.
Note: Much of the description provided in the following three sections on value set definitions and expansions and extending value sets is adapted from Core Principles and Properties of HL7 Version 3 Models [Core Principles and Properties of HL7 Version 3 Models http://www.hl7.org/implement/standards/product_brief.cfm?product_id=58].
Value Set Definitions
Two approaches can be used to define the contents of a Value Set:
-
Extensional Definition: Explicitly enumerating each of the Value Set concepts.
-
An Extensional Definition is an enumeration of all of the concepts within the Value Set. A Value Set defined by extension is composed of explicitly enumerated sets of concept representations (with the Code System in which they are valid). The simplest case is when the Value Set consists of only one concept.
-
Intensional Definition: Defining an algorithm that, when executed by a machine (or interpreted by a human being), yields an enumeration of all of the concepts within the Value Set, which is called a Value Set Expansion.
-
An Intensional Definition is a set of rules that can be expanded (ideally computationally) to an exact set of concept representations at a particular point in time.
Many of the value sets used in the IPS specification are defined intensionally. The source of truth for these value sets and their definitions for IPS is ART-DECOR® [IPS Value Sets in ART-DECOR®].
Figure: Intensional value set definition.
Value Set Expansions
To obtain a list of enumerated concepts, Value Sets must be expanded. This means that the Value Set Definition must be converted to a list of concept representations at a point in time. This normally is a list of codes that may be used in populating or validating communicated model instances (but it may alternatively be a list of designations). While this is straightforward for
extensional Value Set Definitions, an intensional Value Set Definition must be resolved to a Value Set Expansion by processing the rules contained in the Value Set Definition. Value Set Expansion can be done as early as the point of Value Set definition or as late as run time. For intensional Value Sets, the set of concepts contained in the expansion will generally change when
the definition is changed (a new version of the Value Set Definition), but also may change with the identical version of the definition if the underlying code systems change, and the changes are part of the Value Set Expansion. This can be controlled if the definition statement refers to specific code system versions, thereby prohibiting the expansion from changing when the
code system changes with a new version release. See Core Principles and Properties of HL7 Version 3 Models for additional details [Core Principles and Properties of HL7 Version 3 Models http://www.hl7.org/implement/standards/product_brief.cfm?product_id=58].
In order to implement the IPS specification, the intensionally defined value sets must be expanded (as noted above). ART-DECOR® provides capabilities for generating (and store) the required value set expansions. Other terminology servers are also expected to provide this service.
How to extend Value Sets
For elements with a binding to a value set that allows extensibility (Extensible/CWE), it may at times be necessary to extend the value set in order to support implementation needs. Coded With Extensibility (Extensible/CWE) means that the set of codes resulting from processing the Value Set Definition is not necessarily complete for its intended use-case. There may be some
concepts that need to be communicated that cannot be represented within the expansion of the specified value set. In these cases, implementers therefore have permission to send local codes or original text within the coded element if an appropriate code cannot be found within the value set and its current expansion. When this does occur, however, there is an expectation that
implementers should feed back these "missing concepts" to the maintainers of the Value Sets or referenced Code System(s) to have the necessary concept added, and then to transition to the new "official" code when one is subsequently added to the value set.
Functional requirements and high-level use cases
The International Patient Summary, i.e. a “specialty-agnostic, condition-independent" summary, was initially designed for cross-border unscheduled care, but its actual adoption is not limited to this usage scenario.
Patient summary initiatives are currently in various stages of development in different parts of the world. A summary of the most recent initiatives and experiences across the world can be found in the https://international-patient-summary.net/implementations-across-the-globe/.
As mentioned there are several options in terms of creation, sharing and usage of an IPS. For example:
-
An IPS can be created when requested and used before, during, or after a care episode; or can be asynchronously generated and made available for future usage (e.g. store and retrieve).
-
The IPS can be retrieved using a document exchange infrastructure; transported by the patient; or shared using cloud-services.
-
The IPS may be subject of a transformation process that may include syntactical conversions, coded concept mappings and coded concept designation or free text translations. This transformation process may be performed in the creation phase, during the transmission, or after the IPS has been received, possibly using an external service.
-
Finally, the received CDA may be used in different ways. For example, displayed using a common CDA stylesheet; display the extracted relevant information; incorporated into the receiver’s EHR. Alternatively, a specialized viewer may be adopted to enable the display of the translated content.
Moreover, for cross-jurisdiction exchange, the IPS could be used as:
-
shared format among jurisdictions (case A), where jurisdictions originate and use IPS conforming documents unaltered.
-
pivot document among existing summaries / data formats (case B). For example, the IPS is used as intermediate format between the US C-CDA CCD (please note that the CCD scope differs from that of the IPS) and the European eHDSI Patient Summary for a Transatlantic Patient Summary exchange.
-
mixed mode (Case C), where either the originator or the consumer is expected to use an IPS conforming document.
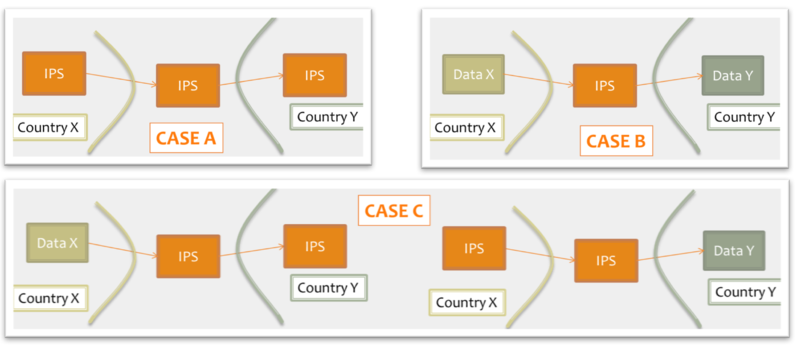
Figure: Examples of IPS usage
An International Patient Summary may:
-
be the result of automatic assembly (assembled IPS) or of a human summarization act (human curated IPS)
-
have one or more EHR sources
-
document information from a single or multiple jurisdictions/organizations
-
aggregate input from a single or multiple encounters.
A clear determination of such contextual information raises the trustworthiness of the received IPS and helps the appropriate usage of its content by the receiver .
Most of these aspects are related to data provenance introduced in
Chapter 5: Provenance.
Finally, there are several technical infrastructures and services that may be designed to support these requirements.
That said, it is out of scope of this standard to:
- give indications about solutions and strategies for the IPS creation, sharing, syntactical and semantic mapping, translation, and use.
- Provide guidance on how to determine the relevance of data for their inclusion in a IPS.
- Define selection or composition rules for facing potential inconsistencies from multiple sources in case of automatic collection.
- Impose constraints on the possible active Patient Summaries. Many jurisdictions require that only one active Patient Summary for unscheduled care is made available, but this is an implementation choice.
Code mappings and multilingual support
The capability of managing locally used coded concepts and reference terminologies, and that of providing receiving providers with human readable information in a language that can be understood by them, are critical aspects to be taken in account in the cross-border sharing of documents. This section summarizes some of the requirements related to these aspects, including also
additional needs derived from the European cross-border services and some lessons learned by the EU/US Trillium Bridge Project.
The European cross-border services (Myhealth@EU), formerly known as eHDSI, use a business to business exchange infrastructure based on a network of country gateways that mediate access to the national infrastructures. The EU Patient Summary (EU PS) is used as a “pivot” document for the cross-border exchange. Local PS using data/document formats are in fact remapped into
the eHDSI PS. The document exchanged is processed each time it passes through one of these gateway applying the needed syntactical transformation, code mappings. and translation of the code designations. Finally, in the current practice the received PSs are displayed using specialized display tools that build a human readable representation of the PS in the target language
using the translated designations reported in the coded entries.
The adoption of translated narratives in the received document has been one of the indications received by the Trillium Bridge Project. This in fact allows extending multilingual support for the cross-border patient summaries to a wider set of potential consumers (EHR-or PHR systems), without requiring specialized viewers as applied in the eHDSI services.
Concept code mapping
In several real world use cases the records used as source for the Patient Summaries may use locally adopted terminologies, which are mapped to the reference value sets when possible, or otherwise used directly to provide uncoded information. This leads to a series of requirements listed below and detailed further in section "Design conventions and principles".
- When the original coded concept is mapped to one of the coded concepts included in the reference value sets (called hereafter reference code/coded concept), both the original and the reference codes SHALL be reported in the IPS instance.
- When the original coded concept is not mapped to one of the coded concepts included in the reference value sets, the original code SHALL be reported in the IPS instance as well as the indication that mapping was not successful.
- When the original record, for a specific coded element, is not able to provide coded but only textual information, this information SHALL be reported in the IPS instance.
This guide also accommodates these situations:
- The original record may support multi-coding. The IPS instance should make clear whether the additional codes belong to the original content or are the result of post hoc concept code mapping.
- The original record may include references to the pieces of text the coding was derived from. If present, the IPS instance should preserve this link between the original code and the referred text.
- Distinct original and reference coded concepts may belong to the same code system. This may be the result of a different level of granularity between the original and the reference value sets, or the result of format transformation - e.g. CCD document is used as input for generating an IPS. The requirement of recording both coded concepts applies also to these cases.
Multilingual support
Multilingual support by IPS can be split in two categories of action:
- The translation of coded concept designations (displayName)
- The translation of the narrative.
These actions may deal with various choices:
- Translation to the language of the receiving care provider: a foreign provider retrieves a translated copy of the IPS from the patient's country of affiliation…
- Translation to a commonly agreed language: an English version of the IPS is prepared.
- Predefined set of translations included in the shared IPS.
This guide does not favor any of these solutions. All of them are supported.
Translation of Designations
The European Cross-border services requires that for “safety and liability reasons” all of the original coded terms (designations, displayName) shall be recorded in the exchanged documents together with at least the English and the receiving country language terms (designations, displayName) associated with the reference codes. The designations translated in the receiving
country language are used to generate the human readable content shown to the receiving provider. No free text translation is applied in this case. In order to accomplish this objective, the IPS should have the capability to record one or more designations, possibly indicating the language used. The solution chosen to fulfill this requirement is specified in section 4.6.
Narrative Translation
Narrative translation covers two kinds of operations:
- Translation of the original narrative text, which can be automated (e.g. using translation services) and/or human curated.
- Creation of new narrative for the target language, based on the coded entries.
The level of quality and liability obtained depends on the solution adopted and on the quality of the translation service used. It is out of the scope of this guide to suggest any of these solutions. In all cases, however:
- the language of the narrative should be identifiable
- the original and the translated narrative should be clearly distinguished
- the translation methodology applied (e.g. derived from the coded entries; translated by a generic service;..) should be noted
Design conventions and principles
How to use terminologies (preferred binding)
As stated in section 1.5, to be universally exchangeable the International Patient Summary must rely on international multilingual reference terminologies. To that effect, each codeable element of the international patient summary template is bound to a Value Set built upon an international reference terminology, such as SNOMED CT, LOINC, UCUM or EDQM Standard Terms. In some
selected cases, in consideration of the availability of other globally usable reference terminologies and for alignment with a future FHIR version of the IPS, FHIR-defined terminologies have been specified. These terminologies have been selected to provide the preferred bindings for the codeable elements of the patient summary. They are the primary
terminologies of this specification.
Nevertheless, it is anticipated that in some situations a system producing an instance of patient summary might not support one or the other of these primary terminologies, supporting only a local interface terminology instead. Similarly, it is also anticipated that the receiving system might in some cases not be able to use a code in a patient summary, either because this code
belongs to a primary terminology that the receiving system does not support or because this code belongs to an interface terminology specific to the country of the producing system.
In order to maximize the international scope and usability of patient summaries, and also to accommodate the exceptional situations listed above, this specification makes these requirements:
- The Primary Code of a codeable element SHOULD be populated.
- If populated, the Primary Code of a codeable element SHALL be chosen from the primary terminology assigned to the value set bound to this element; unless exceptions have been agreed.
- The 'displayName' of the Primary Code SHALL be populated with a term representing this same code in the terminology used, in the language chosen for the current instance of the patient summary.
- When the primary 'code' element is not populated, an appropriate 'nullFlavor' value SHALL be used along with the 'originalText' element (referencing a textual expression in the narrative representing the meaning for the producer) and/or one or more coded 'translation' elements.
- One or more Alternate Codes from a local interface terminology MAY be provided, each with its associated 'displayName'.
- In case the primary code is derived from an alternate terminology the alternate code SHOULD be provided in the translation element.
Primary Code
In the data type for codeable elements (CD constrained by the CD.IPS template), the Primary Code is represented by the attributes @code, @displayName, @codeSystem, @codeSystemName, @codeSystemVersion.
Alternate Code
In the data type for codeable elements (CD constrained by the CD.IPS template), an Alternate Code is carried in a 'translation' sub-element.
Original Text
In the data type for codeable elements (CD constrained by the CD.IPS template), the Original Text is provided in the 'originalText' sub-element.
Representing "known absent" and "not known"
In line with the properties of minimalism and non-exhaustiveness for the IPS (see the IPS definition above), and benefiting from the experience acquired with the European cross-border services, this guide explicitly addresses two general situations:
- condition or activity unknown
- condition or activity known absent.
Other kinds of negations such as: (a) the negation of an allergy to a specific agent; (b) the absence of a particular disease; or (c) the fact that a specific vaccination has not been performed, have been considered beyond the set of essential data for an IPS.
This specification represents this core set of negations (“general condition/activity unknown” and “general condition/activity/known absent” ) using explicit coded elements rather than relying on specific mechanisms of HL7 CDA such as nullFlavor and negationInd attributes or human readable text (possibly not understood by the foreign country receiver).
In contrast to the practice to use negationInd or nullFlavor attributes on a section itself, we prohibit the use of these attributes on section level to express “unknown” or “no information” situations. A section holds the categorized (coded) narrative part of the documented activity and will never carry negationInd or nullFlavor attributes. Instead, we enforce by design, that
“unknown” or “no information” expressions always go to the coded entry with a corresponding act code.
The main reasons for this choice are:
- @negationInd in CDA has been superseded in V3 later by two other negation indicators: @actNegationInd and @valueNegationInd.
- To make clinical content representation less dependent on a particular format or syntax, enabling a more practical path to transforming and exchanging data from one standard format (e.g. CDA R2) to another (e.g. FHIR).
- to provide one single method to express the presence or absence of a particular condition (e.g. an allergy) or activity (e.g. an immunization), or the lack of knowledge regarding this kind of condition or activity, resulting in a more robust and easily implementable specification.
For the other kinds of negations, not explicitly mentioned in this guide, it is suggested to apply – where possible – the same approach. Future versions of this guide may extend the number of cases covered and include new coded concepts for supporting them.
When needed, more specific statements such as the absence of a specific condition or activity, although considered as beyond the set of essential data for IPS, can still be expressed by using the native negationInd attribute of CDA R2.
Uncoded information
An IPS originator may not be able to value a coded element with an appropriate coded concept, but only with textual information. This may happen for two reasons:
- the originator is not able to express the concept in the reference value sets
- the originator is not able to express the concept in any known terminology.
The first case, assuming that the coding strength is Required (aka CNE, coded, no extensions), is represented in this guide with the following assertion:
<code codeSystem="2.16.840.1.113883.6.96" nullFlavor="OTH">
<originalText>
<reference value="#ref1"/>
</originalText>
</code>
That is expressing that there are no codes applicable in the referred code system (in the example, SNOMED CT). Please note that according to this guide the text is documented in the section narrative and only referenced by the coded element.
Note: Data Types R1 doesn't allow specifying that there are no codes applicable in the referred value set, as instead is possible with Data Types R2. Future versions of this guide may consider extending the data type to better support this situation.
The second case, that applies both to Required (aka CNE, coded no extensions) and Extensible (CWE, coded with extensions) coding strengths, is instead here represented valuing the coded element with the most generic nullFlavor “NI” (No Information) and pointing the text in the section narrative:
<value xsi:type="CD" nullFlavor="NI">
<originalText>
<reference value="#ref1"/>
</originalText>
</value>
Note: The most proper NullFlavor code to be used here would be "UNC" (Uncoded). This code is available in the current and other recent versions of the HL7 RIM, but it is not present in version 2.07 of the RIM on which the CDA R2 standard is based. In the absence of "UNC", the most appropriate NullFlavor code to use for representing that the data is unable to be coded is
"NI".
Unmapped Coded Concepts
In several real world situations the records used as source for the Patient Summaries may use locally adopted terminologies mapped into the reference value sets. When the original coded concept cannot be mapped in one of the coded concepts included in the reference value sets, it is recommended to populate the original code in the IPS instance (in a 'translation' sub-element),
with a nullFlavor indicating that the mapping didn’t occur. (See also the "Concept code mapping" in the section "Functional requirements and high-level use cases".). The nullFlavor value depends upon the coding strength of the binding.
Two circumstances are considered here: the case in which the coding strength is Required (CNE) and when it is Extensible (CWE).
In the case of coding strength Required (CNE), use nullFlavor "OTH":
<value xsi:type="CD" codeSystem="2.16.840.1.113883.6.96" nullFlavor="OTH">
[
<originalText>
<reference value="#ref1"/>
</originalText>
]
<translation code="A02.9" codeSystem="2.16.840.1.113883.6.3"
displayName="Infezioni da Salmonella non specificate"/>
</value>
The square brackets [ ] are used to indicate that the originalText element may or may not be present
Note: It may happen that a mapping would be possible in the target code system, but not in the target value set; Data Types R1 does not allow the specification of this difference, that there are no codes applicable in the reference value set, which is possible with Data Types R2.
Future versions of this guide may consider extending the data type to better support this situation.
In the case of Extensible (CWE) coding strength, this guide recommends representing the original code in the <translation> sub-element and using a nullFlavor for the primary code. This highlights the fact that a mapping to the reference value set was attempted, but no suitable target codes were identified.
<value xsi:type="CD" codeSystem="2.16.840.1.113883.6.96" nullFlavor="NI">
[
<originalText>
<reference value="#ref1"/>
</originalText>
]
<translation code="A02.9" codeSystem="2.16.840.1.113883.6.3"
displayName="Infezioni da Salmonella non specificate"/>
</value>
The square brackets [ ] are used to indicate that the originalText element may or may not be present.
Mapped coded concepts
As mentioned above, in several circumstances an original coded concept is mapped into the reference value set. When this occurs, both the original and the reference codes should be reported in the IPS instance.
Functional requirements exposed in "Concept Code Mapping" (multi-coding, link to original text, mapping between codes of the same code system) are also detailed below.
Case 1: Single local code mapped to primary code from the reference value set.
<value xsi:type="CD" code="42338000" codeSystem="2.16.840.1.113883.6.96"
displayName="Salmonella gastroenteritis">
[
<originalText>
<reference value="#ref1"/>
</originalText>
]
<translation code="003.0" codeSystem="2.16.840.1.113883.6.103"
displayName="Gastroenterite da Salmonella"/>
</value>
The square brackets [ ] are used to indicate that the originalText element may or may not be present
Case 2: Multiple local codes mapped together using nested 'translation' elements, and mapped to the primary code from the reference value set.
<value xsi:type="CD" code="422479008" codeSystem="2.16.840.1.113883.6.96"
codeSystemName="SNOMED CT"
displayName="FEMALE BREAST INFILTRATING DUCTAL CARCINOMA, STAGE 2">
[
<originalText>
<reference value="#problem4name"/>
</originalText>
]
<translation code="code-example" codeSystem="1.999.999"
codeSystemName="this is only an example"
displayName="FEMALE BREAST INFILTRATING DUCTAL CARCINOMA, STAGE 2">
<translation code="174.9" codeSystem="2.16.840.1.113883.6.103"
codeSystemName="ICD-9CM"
displayName="Malignant neoplasm of breast (female), unspecified"/>
<translation code="C50.919" codeSystem="2.16.840.1.113883.6.90"
codeSystemName="ICD-10-CM"
displayName="Malignant neoplasm of unspecified site of unspecified female breast"/>
</translation>
</value>
Case 3: Original and the reference coded concepts belong to the same code system (distinct codes). This may be the result of a different level of granularity between the original term and the reference value sets.
<code code="60591-5" codeSystem="2.16.840.1.113883.6.1"
codeSystemName="LOINC" displayName="Patient Summary">
<translation code="60592-3" codeSystem="2.16.840.1.113883.6.1"
displayName="Patient summary unexpected contact"/>
</code>
Note: The R1 data type definition identifies the <translation> as “a set of other concept descriptors that translate this concept descriptor into other code systems." There is however a common understanding that it may be more than one representation in a single code system where code systems allow multiple representations, such as SNOMED CT. Data Types R2 extended
the possibility to provide translations also in the same code system.
Translation of designations
The capability of recording one or more designations, in different languages, for the exchanged Patent Summary is one of the functional requirements requested for “safety and liability reasons” by the European Cross-border services (see "Designations’ Translation" in section "Functional requirements and high-level use cases" for more details).
Given that the base CDA R2 standard which uses datatypes R1 does not offer a native way to convey the language translation of 'displayName', this guide introduces an optional 'ips:designation' extension to the CD datatype for that purpose.
Below are examples of usage of this extension.
No code mapping
<code code="60591-5" codeSystem="2.16.840.1.113883.6.1"
codeSystemName="LOINC" displayName="Patient Summary">
<ips:designation language="it-IT">Profilo Sanitario Sintetico</ips:designation>
<ips:designation language="fr-FR">Patient Summary</ips:designation>
<ips:designation language="en">Patient Summary</ips:designation>
</code>
Including code mapping
<value xsi:type="CD" code="42338000" codeSystem="2.16.840.1.113883.6.96"
displayName="Salmonella-gastroenterit">
<ips:designation language="da-DK">Salmonella-gastroenterit</ips:designation>
<ips:designation language="en">Salmonella gastroenteritis (disorder)</ips:designation>
[
<originalText>
<reference value="#ref1"/>
</originalText>
]
<translation code="003.0" codeSystem="2.16.840.1.113883.6.103"
displayName="Gastroenterite da Salmonella"/>
</value>
Narrative Translations
“Narrative translation” means both the translation of the original narrative text, that can be human curated or automatically performed, and the generation of a translated narrative based on the coded entries.
The functional requirements associated with this process are described in "Designations’ Translation" in section "Functional requirements and high-level use cases", and can be summarized in two main points : (a) language identification and (b) distinguishable original and translated narratives. Moreover, the methodology applied for the narrative translation (e.g. derived from
the coded entries; translated by a generic service;..) should be noted.
Regarding the translation of section narrative <text>, this guide recommends providing this translation on purely textual subordinate sections (one per translation) as specified in the IPS Translation Section (2.16.840.1.113883.10.22.3.15) template.
An example of this is:
<section>
<templateId root="2.16.840.1.113883.3.1937.777.13.10.5"/>
<id root="..." extension="..."/>
<code code="48765-2" codeSystem="2.16.840.1.113883.6.1"
displayName="Allergies and adverse reactions"/>
<title>Allergies and Intolerances</title>
<text>No known Allergies</text>
<!-- omissions -->
<component>
<section>
<!-- subordinate section carrying a translation of the parent section -->
<title>Allergie ed Intolleranze</title>
<text>Nessuna Allergia Nota</text>
<languageCode code="it-IT"/>
</section>
</component>
</section>
Provenance
In the development of this Implementation Guide, consideration was given to the HL7 CDA® Release 2 Implementation Guide: Data Provenance, Release 1 - US Realm Draft Standard for Trial Use (December 2015). That guide provides a matrix offering a thorough and systematic analysis of provenance characteristics of electronic health records. Given the agreed scope principle
(see section "Introduction") that the IPS be minimal and implementable, and the variable maturity and operational methods of existing national patient summaries, the proposal is that this first version should not attempt to require the full detail of that provenance specification.
The approach proposed for this version of the IPS is to:
- Allow optional documentation of section-level provenance.
- Require document-level provenance.
- Define IPS document provenance as one of two types: human-curated or software-assembled, based on the authors recorded in the header.
- The classification is based on whether the IPS document is constructed by a human or an automated process, regardless of whether the IPS contains some content of both kinds.
- Require the IPS source system to identify the IPS provenance type and author.
- The author shall be a human, if the IPS provenance type is "human-curated", or a device or system if the IPS provenance type is "software-assembled".
- In the case of a software-assembled IPS that is then verified by a human, the document provenance type shall be "software-assembled" and the author shall be the device or system that constructed the IPS document, but an additional verifier identity shall name the human who performed this check. For the avoidance of doubt, verifier is not the same as legalAuthenticator (the verifier is represented as a participant with typeCode "VRF"). However, in cases where the verifying person intentionally wishes to sign the document, this shall be recorded as a legalAuthenticator.
- Allow optional section level author, provenance type, verifier, and informant identification, for IPS source systems that can support this.
- Not attempt to implement the US Realm CDA data provenance templates.
Note: Discussions with the EHR work group suggest that a possible future project should be an IPS functional profile, once there is greater clarity and operational experience of using the IPS.
Representation of Names
This specification requires that any Person Name is represented including at least the given and family components and therefore is never documented as a single string.
Even though it is recognized that there is not in all cultures the same concept of “family name”, no evidence has been collected in analyzing the international context (e.g. Japan, Korea; China) that justifies the retirement of this requirement.
Moreover, due to the global scope of the International Patient Summary, the case of non-alphabetic representations of the names has also been considered.
In this case, to facilitate the global use of the IPS, at least one alphabetic representation of the name SHALL be provided.
Determining the Status of Clinical Statement
Note: Most of the description provided in this section is copied from the § 3.3 Determining the Status of Clinical Statement of the C-CDA DSTU R2.1 Implementation Guide Volume 1 [HL7 C-CDA Implementation Guide DSTU R2.1 http://www.hl7.org/implement/standards/product_brief.cfm?product_id=379].
A recipient must be able to determine whether the status of an entry — which can include a problem, a medication administration, etc. — is active, completed, or in some other state. Determination of the exact status is dependent on the interplay between an act’s various components (such as statusCode and effectiveTime). The following principles apply when representing or
interpreting the status of a clinical statement.
- The Act.statusCode of the clinical statement specifies the state of the entry: Per the RIM, the statusCode “reflects the state of the activity. In the case of an Observation, this is the status of the activity of observing, not the status of what is being observed.”
- Act.statusCode and Act.moodCode are inter-related: Generally, an act in EVN (event) mood is a discrete event (a user looks, listens, measures, and records what was done or observed), so generally an act in EVN mood will have a statusCode of “completed.” A prolonged period of observation is an exception, in which a user would potentially have an observation in EVN mood
that is “active.” For an Observation in RQO (request) mood, the statusCode generally remains “active” until the request is complete, at which time the statusCode changes to “completed.” For an Observation in GOL (goal) mood, the statusCode generally remains “active” as long as the observation in question is still an active goal for the patient.
- Act.statusCode and Act.effectiveTime are interrelated: Per the RIM, the effectiveTime, also referred to as the “biologically relevant time,” is the time at which the act holds for the patient. So, whereas the effectiveTime is the biologically relevant time, the statusCode is the state of the activity. For a provider seeing a patient in a clinic and observing a history of
heart attack that occurred 5 years ago, the status of the observation is completed, and the effectiveTime is five years ago.
The IPS Problem Concern Entry and the IPS Allergy and Intolerance Concern templates reflect an ongoing concern on behalf of the provider that placed the concern (e.g. a disease, an allergy, or other conditions) on a patient’s problem or allergy list. The purpose of the concern act is that of supporting the tracking of a problem or a condition (e.g. an allergy). A concern act can
contain one or more discrete observations related to this concern. Each of them reflects the change in the clinical understanding of a condition over time. For instance, a Concern may initially contain a symptom of “chest pain”, later identified as consequence of a gastroesophageal reflux. The later problem observation will have a more recent author time stamp.
There are different kinds of status that could be of clinical interest for a condition:
- The status of the concern (active, inactive,..)
- The status of the condition (e.g. active, inactive, resolved,..)
- The confirmation status [verification status, certainty] (e.g. confirmed, provisional, refuted,…)
Not all of them can be represented in a CDA using the statusCode elements of the concern (ACT) and observation (condition).
Status of the concern and related times
The statusCode of the Problem Concern Act is the definitive indication of the status of the concern. So long as the provider has an ongoing concern — meaning that the provider is monitoring the condition, whether it includes problems that have been resolved or not — the statusCode of the Concern Act is “active.” When the underlying conditions are no longer an active concern, the
statusCode of the Problem Concern Act is set to “completed.”
The Concern Act effectiveTime reflects the time that the underlying condition was considered a concern. It may or may not correspond to the effectiveTime of the condition (e.g., even five years later, the clinician may remain concerned about a prior heart attack). The effectiveTime/low of the Concern Act asserts when the concern became active. This equates to the time the
concern was authored in the patient's chart. (i.e. it should be equal to the earliest author time stamp) The effectiveTime/high asserts when the concern became inactive, and it is present if the statusCode of the concern act is "completed". If this date is not known, then effectiveTime/high will be present with a nullFlavor of "UNK".
Status of the condition and related times
Each Observation contained by a Concern Act is a discrete observation of a condition. Its statusCode is always “completed” since it is the “status of the activity of observing, not the status of what is being observed”. The clinical status of a condition (e.g. a disease, an allergy,..) is instead recorded by specialized subordinated observations (IPS Allergy Status Observation;
IPS Problem Status Observation), documenting whether it is active, in remission, resolved, et cetera. The effectiveTime, also referred to as the "biologically relevant time", is the time at which the observation holds for the patient. For a provider seeing a patient in the clinic today, observing a history of penicillin allergy that developed six months ago, the effectiveTime
is six months ago. The effectiveTime of the Observation gives an indication of whether or not the underlying condition is resolved, but the current status is documented by a subordinated observation. The effectiveTime/low (a.k.a. "onset date") asserts when the allergy/intolerance became biologically active. The effectiveTime/high (a.k.a. "resolution date") asserts when the
allergy/intolerance was biologically resolved. If the date of resolution is not known, then effectiveTime/high will be present with a nullFlavor of "UNK".
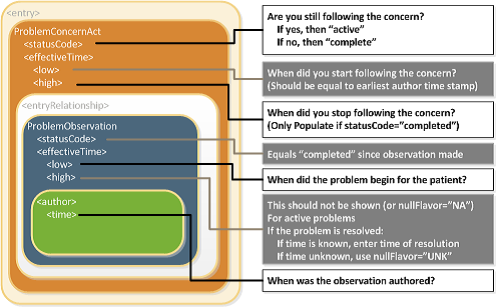
Figure: Problem Concern Act (from C-CDA IG DTSU R2.1 HL7 C-CDA Implementation Guide DSTU R2.1 http://www.hl7.org/implement/standards/product_brief.cfm?product_id=379)
Confirmation status
The confirmation status, also noted as verification status or certainty, indicates the certainty associated with a condition (e.g. confirmed, provisional, refuted,…) providing information about the potential risk, for example, of a reaction to the identified substance. The confirmation status of a condition (e.g. a disease, an allergy,..) is recorded by a specialized
subordinated observation (IPS certainty Observation), documenting whether the condition is confirmed, unconfirmed, provisional, refuted, et cetera.ge
The use of medication statements in the summary
A medication list may strongly vary depending on the context of use (e.g. support for prescription or dispensation, drug reconciliation, etc. ) and on the type of information reported (e.g. patient-reported medication, prescribed, dispensed or administered medications, active or past medications, etc.).
This is still true also for the medication summary in a Patient Summary. It could be, for instance, a list of "Relevant prescribed medicines whose period of time indicated for the treatment has not yet expired whether it has been dispensed or not, or medicines that influence current health status or are relevant to a clinical decision " (European eHN Guideline on the electronic exchange of health data under Cross-Border Directive 2011/24/EU: Patient Summary. Release 3.3.; a list of actually dispensed medications; a list of relevant
medications for the patient (IHE PCC, IHE Patient Care Coordination Technical Framework http://ihe.net/Technical_Frameworks/#pcc); or conversely, it could be a complete history including the full patient's prescription and dispensation history and information about intended drug monitoring
(C-CDA [HL7 C-CDA Implementation Guide DSTU R2.1 http://www.hl7.org/implement/standards/product_brief.cfm?product_id=379]).
Moreover, for the scope of the International Patient Summary, it is important to know what medications are being taken by or have been given to a patient; without necessarily providing all the details about the medication order, supply, administration or monitoring. This information need, in line with the IPS principle of minimum non exhaustive data, is well expressed by the
concept of Medication Statement (see https://www.hl7.org/fhir/medicationstatement.html). The IPS medication summary is therefore typically a list of relevant medication statements, possibly built from either a prescription list or a dispensation list. In fact, in many practical
cases data included in a Patient Summary are derived from the list of the medicines prescribed by a GP and recorded in its EHR-S; or extracted from a regional/national prescribing and/or dispensation systems. In these cases, data obtained from actual dispensation and/or prescription records can be still recorded in the IPS as statements and the original request, supply or
administration records referred by the statement if really needed.
Medicinal Product Identification
The identification of medicinal products is quite easily solved within a single jurisdiction relying on local drug databases. In contrast, it is one of the major open issues for eHealth services across jurisdictions.
The set of ISO standards called IDMP [IDMP standards <a href="https://www.idmp1.com/idmp-standards" rel="noopener noreferrer nofollow">https://www.idmp1.com/idmp-standards</a>] – designed initially for the regulatory scope, but hopefully extensible to other domains – are the most promising solution for solving this known issue, as also highlighted by the <a
href="https://unicom-project.eu/" rel="noopener noreferrer nofollow">European project UNICOM</a>. The completion of the IDMP implementation guides, the deployment of the needed supporting services, and the development of some companion standards that will allow the seamless flow of the IDMP identifiers and attributes from the regulatory space to the clinical world (and
back) are however still in progress. Additional time is needed before these identifiers and attributes will be available in full for practical use.
Following therefore the IPS principles of “implementability”, “openness" and "extensibility”, the solution proposed here does not rely on IDMP identifiers. Nonetheless, it takes into account, however, relevant IDMP identifiers and attributes which are already usable in the IPS, namely the Pharmaceutical Product Identifiers (PhPIDs), the Medicinal Product Identifier (MPID), and
the Medicinal Product Package Identifier (PCID).
Note: IDMP Medicinal Product (MPID) and Medicinal Product Package (PCID) identifiers depend on the market authorization. The “same” product might therefore have different IDs if different authorizations have been received in different countries, while the PhPID should be the same. For the purpose of the IPS, future standards and implementation guides should define global
product identifiers that do not depend on the drug registration process (as the Virtual Medicinal Product in SNOMED CT) and relate to IDMP identifiers.
Thus, in the absence of a global identification system for medicinal products, the solution proposed here is based on the approach initially adopted by the European cross-border services (epSOS and currently by the eHDSI project), reused by the IHE Pharmacy templates and more recently adopted (for specific cases) by the HL7 Pharmacy Medication statement templates. The main idea
is to integrate local drug identifiers (e.g. product codes) with all the relevant identifying and descriptive attributes that may help the receiver to understand the type of product the sender is referring to, e.g.: active ingredients, administrative dose forms, strength, route of administration and package description.
Medication data is usually represented in the CDA Templates using the manufacturedMaterial class, which includes a code and a name to describe any level of the product: packaged product, medicinal product, classes or clusters or products, and so on.
This information is not however sufficient for covering the requirements of the IPS.
</html>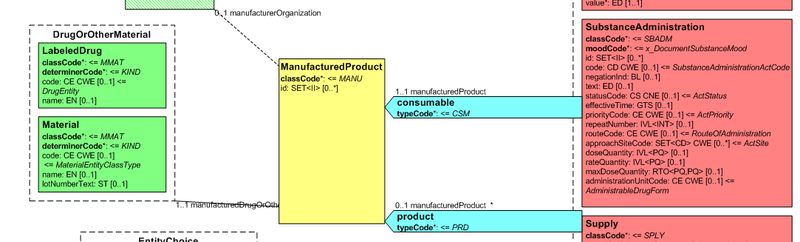
Figure: Representation of medicines in CDA
Hence, in order to describe these attributes the CDA model has been extended enhancing the Manufactured Material class with attributes and relationships derived from the latest published version of the R_Medication CMET based on the HL7 Common Product Model (“The common product model is used to improve the alignment between the different representations of
products used within the body of HL7 Version 3 models. One goal of this effort is to make it possible to have a single representation, which could then be used as a common message element type (CMET) across those models.”) .
The next figure provides an overview of the CDA model extensions, approach adopted and further developed by the HL7 CDA® R2 Implementation Guide: Pharmacy Templates.
Starting from this IG version, the HL7 CDA IPS medication templates have been defined as specialization of the Pharmacy ones, adopting the pharm (urn:hl7-org:pharm) extensions.
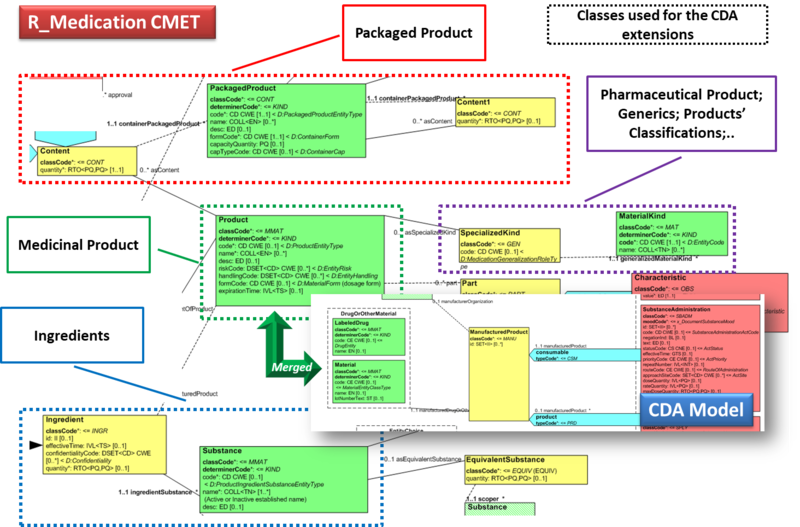
Figure: Extension of the CDA model from the content of the R_Medication CMET.
Conformance clause
This section references the requirements, criteria, or conditions to be satisfied in order that a product (tangible) or a service may claim conformance to this guide, and how other artifacts may claim compliance with it. (Note: The concept of conformance and compliance are used coherently with the HL7 Service-Aware Interoperability Framework: Canonical Definition Specification,
Release 2 [HL7 Service-Aware Interoperability Framework: Canonical Definition Specification, Release 2 http://www.hl7.org/implement/standards/product_brief.cfm?product_id=3].
The fulfilment of these clauses indirectly assures that a product that is the subject of a “conformity assessment” satisfies the business or the design requirements this specification complies to. It should, however, be clear that compliance with the specified business or design requirements, for example with the EN ISO 17269 IPS, does not imply that the compliant
implementations are technically interoperable.
A “conformity assessment” is a process that assesses that any proposition that is true in a given specification is also true in the service or product that implements it. In most real-world cases conformance testing objects are used to technically validate the products. These objects provide a great help in the validation of the instances, even if they are most often not
sufficient to guarantee the functional/ semantic conformity: many real-life examples can be made about instances that are technically valid, but not clinically meaningful or correct.
The "rules" and processes for refining the standard through constraint and extension, including which standard artifacts are subject to constraint or extension; the definition of constraint and localization profiles; the criteria for establishing a conformance statement; and the principles guiding who may define extensions to the standards and under what circumstances they apply
to the CDA standards are defined in §1.3 CDA Conformance of the CDA and detailed in the HL7 V3 Refinement, Constraint and Localization section (see the CDA R2 Standard [CDA R2 Standard]).
This guide does not provide additional requirements regarding the Recipient and the Originator Responsibilities.
The formal representation used in this implementation guide for expressing the conformance statement is described in chapter "How to read the table view for templates" of this guide and makes use of a tabular representation that may include also computable or textual constraints. The template rules are formalized using the computable format defined by the HL7 Templates Standard:
Specification and Use of Reusable Information Constraint Templates, Release 1 [HL7 Templates Standard: Specification and Use of Reusable Information Constraint Templates, Release 1 http://www.hl7.org/implement/standards/product_brief.cfm?product_id=377] in order to facilitate also the automatic generation of consistent testing and validation capabilities.
The HL7 Templates Standard: "Specification and Use of Reusable Information Constraint Templates, Release 1" defines also how derived templates may relate to the templates defined in this guide for example:
- Specialization: “A specialized template is a narrower, more explicit, more constrained template based on a “parent” template.
- Adaptation: “The adapted template is “based on” the original template which means it can be an extension or a specialization (restriction) of the original template design.”
- Equivalency: “two templates have the same purpose and the same design; however, their governance and/or metadata and/or details of their design may be different.”
Based on this the following way to use this guide may be considered :
- IPS as a document: the conformance is asserted at the document level. All the rules defined by this guide, or by a specialized IPS document level template, are fulfilled. Implementers may take advantage of the template openness to better support specific cases - “extended” parts, however, may not be interoperable among them.
- IPS as a library: the conformance is asserted at the section or the entry level. The templates are used as a library to build, for example, new cross-border documents. For example the immunization section may be used to build an electronic implementation of the WHO yellow card for vaccinations; or the IPS section templates are used to communicate to the country of
affiliation minimal and non-exhaustive information about the encounter in which the Patient Summary has been used (cross-border encounter report ). Implementers may take advantage of the template openness to better support specific cases - "extended” parts, however, may not be interoperable among them.
- IPS as a reference: the implementation is conformant with templates that are an adaptation of or equivalent to those defined by this guide. In this case some of the rules defined by this guide are not fulfilled and the conformance cannot be asserted. However, differences may be limited and the effort required to achieve the harmonization may not be not large. Typical
examples are templates in which alternatives vocabularies are used.
Jurisdictions may also decide to impose the closure of the template in order to limit the implementation optionality.
This should be carefully evaluated in terms of the flexibility of the solution.
CDA Document Level Templates
International Patient Summary
| Id | 2.16.840.1.113883.10.22.1.2 | Effective Date | 2024‑08‑02 13:02:39 |
|---|
| Status |  Draft Draft | Version Label | 2024 |
|---|
| Name | HL7-IPS | Display Name | International Patient Summary |
|---|
| Description | The International Patient Summary is a "Minimal and non-exhaustive Patient Summary, specialty-agnostic, condition-independent, but readily usable by all clinicians for the unscheduled (cross-border) care of a patient."
The IPS templates aim to:
Serve for both cross-jurisdictional (through adaptation/extension for multi-language and realm scenarios, including translation) and national (through localization) patient summaries. Support emergency care and unplanned care in any country (home and foreign), regardless of language Define value sets based on international vocabularies that
are usable and understandable in any country
|
|
| Context | Pathname / |
|---|
| Classification | CDA Document Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Uses | | Uses 23 templates | | Used by | as | Name | Version |
|---|
| hl7ips-transaction-3 | Transaction |  IPS Created (2024) IPS Created (2024) | 2024‑08‑02 | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.2.1 | Include |  IPS CDA recordTarget (2021) IPS CDA recordTarget (2021) | DYNAMIC | | 2.16.840.1.113883.10.22.2.2 | Include |  IPS CDA author (STU1) IPS CDA author (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.2.3 | Include |  IPS CDA custodian (2021) IPS CDA custodian (2021) | DYNAMIC | | 2.16.840.1.113883.10.22.2.4 | Include |  IPS CDA legalAuthenticator (STU1) IPS CDA legalAuthenticator (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.2.5 | Include |  IPS Patient Contacts (STU1) IPS Patient Contacts (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.2.6 | Include |  IPS CDA documentationOf (STU1) IPS CDA documentationOf (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.2.7 | Include |  IPS CDA relatedDocument (STU1) IPS CDA relatedDocument (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.3.1 | Containment |  IPS Medication Summary Section (STU1) IPS Medication Summary Section (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.3.2 | Containment |  IPS Allergies and Intolerances Section (STU2) IPS Allergies and Intolerances Section (STU2) | DYNAMIC | | 2.16.840.1.113883.10.22.3.3 | Containment |  IPS Problems Section (STU1) IPS Problems Section (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.3.4 | Containment |  IPS History of Procedures Section (STU1) IPS History of Procedures Section (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.3.5 | Containment |  IPS Immunizations Section (STU1) IPS Immunizations Section (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.3.6 | Containment |  IPS Medical Devices Section (2021) IPS Medical Devices Section (2021) | DYNAMIC | | 2.16.840.1.113883.10.22.3.14 | Containment |  IPS Results Section (STU1) IPS Results Section (STU1) | DYNAMIC | | 1.3.6.1.4.1.19376.1.5.3.1.1.5.3.2 | Containment |  IHE Coded Vital Signs Section (2014) IHE Coded Vital Signs Section (2014) | DYNAMIC | | 2.16.840.1.113883.10.22.3.7 | Containment |  IPS History of Past Illness Section (STU1) IPS History of Past Illness Section (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.3.8 | Containment |  IPS Functional Status Section (TI-2020) IPS Functional Status Section (TI-2020) | DYNAMIC | | 2.16.840.1.113883.10.22.3.9 | Containment |  IPS Plan of Care Section (TI-2020) IPS Plan of Care Section (TI-2020) | DYNAMIC | | 2.16.840.1.113883.10.22.3.10 | Containment |  IPS Social History Section (TI-2020) IPS Social History Section (TI-2020) | DYNAMIC | | 2.16.840.1.113883.10.22.3.11 | Containment |  IPS History of Pregnancy Section (TI-2020) IPS History of Pregnancy Section (TI-2020) | DYNAMIC | | 2.16.840.1.113883.10.22.3.12 | Containment |  IPS Advance Directives Section (TI-2020) IPS Advance Directives Section (TI-2020) | DYNAMIC | | 2.16.840.1.113883.10.22.15 | Containment |  IPS Alerts Section IPS Alerts Section | DYNAMIC | | 2.16.840.1.113883.10.22.16 | Containment |  IPS Patient Story Section IPS Patient Story Section | DYNAMIC |
|
|
|---|
| Relationship | Specialization: template 2.16.840.1.113883.10.22.1.1 International Patient Summary (2020‑07‑14 16:08:21)
Version: template 2.16.840.1.113883.10.22.1.1 International Patient Summary (2020‑05‑08 12:30:59)
Version: template 2.16.840.1.113883.10.22.1.1 International Patient Summary (2017‑04‑11)
Adaptation: template 1.3.6.1.4.1.12559.11.10.1.3.1.1.3 epSOS-Patient Summary (2013‑12‑20) ref epsos-
Adaptation: template 2.16.840.1.113883.10.12.1 CDA ClinicalDocument (2005‑09‑07) ref ad1bbr- |
|---|
| Example | | Example | <ClinicalDocument>
<realmCode code="ES"/> <typeId extension="POCD_HD000040" root="2.16.840.1.113883.1.3"/> <templateId root="2.16.840.1.113883.10.22.1.1"/> <id root="2.16.724.4.8.10.200.10" extension="PSCTD0160f274530a031"/> <code displayName="Patient Summary" code="60591-5" codeSystem="2.16.840.1.113883.6.1">
<title>Patient Summary</title> <effectiveTime value="20111113125600+0200"/> <confidentialityCode code="N" displayName="normal" codeSystem="2.16.840.1.113883.5.25"/> <languageCode code="es-ES"/> <setId root="2.16.724.4.8.10.200.10" extension="PSCTD0160f274530a031S"/> <versionNumber value="2"/> <!-- include template 2.16.840.1.113883.10.22.2.1 'IPS CDA recordTarget' (dynamic) 1..1 M -->
<!-- include template 2.16.840.1.113883.10.22.2.2 'IPS CDA author' (dynamic) 1..* M -->
<!-- include template 2.16.840.1.113883.10.22.2.3 'IPS CDA custodian' (dynamic) 1..1 M -->
<!-- include template 2.16.840.1.113883.10.22.2.4 'IPS CDA legalAuthenticator' (dynamic) 0..1 R -->
<!-- include template 2.16.840.1.113883.10.22.2.5 'IPS Patient Contacts' (dynamic) 0..* O -->
<!-- include template 2.16.840.1.113883.10.22.2.6 'IPS CDA documentationOf ' (dynamic) 1..1 M -->
<!-- include template 2.16.840.1.113883.10.22.2.7 'IPS CDA relatedDocument' (dynamic) 0..* R -->
<component>
<structuredBody classCode="DOCBODY">
<component>
<!-- template 2.16.840.1.113883.10.22.3.1 'IPS Medication Summary Section' (dynamic) -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.3.2 'IPS Allergies and Intolerances Section' (dynamic) -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.3.3 'IPS Problems Section' (dynamic) -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.3.4 'IPS History of Procedures Section' (dynamic) -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.3.5 'IPS Immunizations Section' (dynamic) -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.3.6 'IPS Medical Devices Section' (dynamic) -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.3.7 'IPS History of Past Illness Section' (dynamic) -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.3.14 'IPS Results Section' (dynamic) -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.3.8 'IPS Functional Status Section' (dynamic) -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.3.9 'IPS Plan of Treatment Section' (dynamic) -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.3.10 'IPS Social History Section' (dynamic) -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.3.11 'IPS History of Pregnancy Section' (dynamic) -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.3.12 'IPS Advance Directives Section' (dynamic) -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.15 'IPS Alerts Section' (dynamic) -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.16 'IPS Patient Story Section' (dynamic) -->
</component> </structuredBody> </component> </code></ClinicalDocument> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | | R | CDA header | (HL7-IPS) |  | hl7:realmCode
|
| CS | 0 … 1 | R | | (HL7-IPS) |  | hl7:typeId
|
| II | 1 … 1 | M | The clinical document typeId identifies the constraints imposed by CDA R2 on the content, essentially acting as a version identifier.
| (HL7-IPS) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.1.3 |  |  | @extension
|
| st | 1 … 1 | F | POCD_HD000040 | | | Example | <typeId extension="POCD_HD000040" root="2.16.840.1.113883.1.3"/> |  | hl7:templateId
|
| II | 1 … 1 | M | | (HL7-IPS) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.1.1 |  | hl7:id
|
| II | 1 … 1 | M | Unique identifier of this instance of the Patient Summary. | (HL7-IPS) |  | hl7:code
|
| CE.IPS | 1 … 1 | M | Determines the document type that is the "Patient Summary" document | (HL7-IPS) |  |  | @displayName
|
| | 1 … 1 | R | |  |  | @code
|
| CONF | 1 … 1 | F | 60591-5 |  |  | @codeSystem
|
| 1 … 1 | F | 2.16.840.1.113883.6.1 (LOINC) | | | Example | <code code="60591-5" codeSystem="2.16.840.1.113883.6.1" codeSystemName="LOINC" displayName="Patient Summary"/> |  |  | hl7:translation
|
| CD.IPS | 0 … * | R | This element can be here used either to provide the originally used document code if this IPS is the result of a transformation. | (HL7-IPS) |  | hl7:title
|
| ST | 1 … 1 | M | ClinicalDocument/title is used for display purposes. | (HL7-IPS) |  | hl7:effectiveTime
|
| TS.IPS.TZ | 1 … 1 | M | Time of creation of the Patient Summary | (HL7-IPS) | | | Example | <effectiveTime value="20111113125600+0200"/> |  | hl7:confidentialityCode
|
| CE.IPS | 1 … 1 | R | | (HL7-IPS) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.1.11.16926 HL7 BasicConfidentialityKind (DYNAMIC) |
| | | Example | <confidentialityCode code="N" codeSystem="2.16.840.1.113883.5.25" displayName="normal"/> |  | hl7:languageCode
|
| CS | 1 … 1 | M | Document Language Code | (HL7-IPS) | | | Constraint | The two characters form SHALL be used when available; otherwise the three characters representation SHALL be adopted | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.4.642.3.21 All Languages (DYNAMIC) |
| | | Example | <languageCode code="en-GB"/> | | | Example | <languageCode code="fil-PH"/> | | | Schematron assert | role | error | | | | test | matches(@code,'[a-z]{2,3}-[A-Z]{2,3}') | | | | Message | The language code SHALL be in the form nn-CC or nnn-CCC, in accordance with BCP 47 (e.g. nn is the ISO language code; CC is ISO country code) | |  | hl7:setId
|
| II | 0 … 1 | R | This attribute “represents an identifier that is common across all document revisions”.
In the case the IPS instance is generated as result of one or more transformations (translation/transcoding) the setId is supposed to remain unchanged across all those transformations.
Implementers are recommended to use this attribute.
| (HL7-IPS) |  | hl7:versionNumber
|
| | 0 … 1 | R | | (HL7-IPS) | | Included | 1 … 1 | M | from 2.16.840.1.113883.10.22.2.1 IPS CDA recordTarget (DYNAMIC) |  | hl7:recordTarget
|
| | 1 … 1 | M | | (HL7-IPS) |  |  | @typeCode
|
| cs | 0 … 1 | F | RCT |  |  | @contextControlCode
|
| cs | 0 … 1 | F | OP | | | Example | <recordTarget typeCode="RCT" contextControlCode="OP">
<patientRole classCode="PAT">
<id root="1.2.3.999" extension="__example only__"/> <addr>
<streetAddressLine>HSE M CASSAR STR</streetAddressLine> <city>ISLA</city> <country>MT</country> </addr> <telecom use="HP" value="tel:+356124567891"/> <telecom use="WP" value="mailto:elif@foo.too.mt"/> <patient>
<name>
<family>BORG</family> <given>TANIA</given> </name> <administrativeGenderCode code="F" codeSystem="2.16.840.1.113883.5.1" displayName="Female"/> <birthTime value="19430130"/> <!-- Optional guardian information ; see example below-->
<!-- Optional languageCommunication information see example below -->
</patient> </patientRole></recordTarget> |  |  | hl7:patientRole
|
| | 1 … 1 | M | | (HL7-IPS) | | |  | | hl7ips-dataelement-2.1 |  Patient Attributes Patient Attributes |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | cs | 0 … 1 | F | PAT | | II | 1 … * | R | Patient Identifiers: Primary Patient Identifier (Regional/National Health Id), Secondary Patient Identifier (Social/Insurance Number) | (HL7-IPS) | | |  | | hl7ips-dataelement-202 |  Healthcare related Identifiers Healthcare related Identifiers |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-7 |  Insurance identifier Insurance identifier |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | AD.IPS | 1 … * | R | The patient address. | (HL7-IPS) | | |  | | hl7ips-dataelement-162 |  Address Address |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | Constraint | When used for cross-border exchange the country address part has to be provided. | | Included | | | from 2.16.840.1.113883.10.22.11 IPS Address (DYNAMIC) | | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.10637 PostalAddressUse (2005‑05‑01) |
| | cs | 0 … 1 | F | NI | | | Constraint | SHALL NOT have mixed content except for white space
If there is no information, the nullFlavor attribute shall have a value of 'NI' and no address parts shall be present, otherwise there shall be no nullFlavor attribute, and at least one of the address parts listed below shall be present. | | | Schematron assert | role | error | | | | test | @nullFlavor or hl7:* | | | | Message | If addr is not nullflavored at least one sub element has to be provided | | | ADXP | 0 … * | C | Subject's or Organization's Street Address Line | (HL7-IPS) | | | Schematron assert | role | error | | | | test | hl7:streetAddressLine and (hl7:city or hl7:postalCode) | | | | Message | If the address line is included either the city or the zip code has to be provided | | | ADXP | 0 … 1 | C | Subject's or Organization's City | (HL7-IPS) | | ADXP | 0 … 1 | C | Subject's or Organization's Postal Code | (HL7-IPS) | | ADXP | 0 … 1 | C | Subject's or Organization's State or Province | (HL7-IPS) | | ADXP | 0 … 1 | C | Subject's Country. | (HL7-IPS) | | | Constraint | The content of this element SHALL be selected EITHER from ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300 DYNAMIC OR MAY be selected from ISO Country Alpha-3 2.16.840.1.113883.1.11.171 DYNAMIC, IF the country is not specified in ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300. | | TEL | 1 … * | R | Patient’s telecom information : e.g. telephone number, e-mail address. | (HL7-IPS) | | |  | | hl7ips-dataelement-100 |  Telecoms Telecoms |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.201 TelecommunicationAddressUse (DYNAMIC) |
| | cs | 0 … 1 | F | NI | | | Constraint | If there is no information, the nullFlavor attribute shall have a value of 'NI' and the "value" and "use" attributes shall be omitted, otherwise the nullFlavor attribute shall not be present, and the "value" and "use" attributes shall be present.
| | | Example | <telecom use="HP" value="tel:+356124567891"/> | | | Example | <telecom use="WP" value="mailto:elif@foo.too.mt"/> | | | Example | <telecom nullFlavor="NI"/> | | | 1 … 1 | M | | (HL7-IPS) | | cs | 0 … 1 | F | PSN | | cs | 0 … 1 | F | INSTANCE | | | Example | Japanese example (Person Name) <patient>
<name use="IDE">
<family>木村</family> <given>通男</given> </name> <name use="SYL">
<family>きむら</family> <given>みちお</given> </name> <name use="ABC">
<family>KIMURA</family> <given>MICHIO</given> </name> <administrativeGenderCode code="M" codeSystem="2.16.840.1.113883.5.1" displayName="Male"/> <birthTime nullFlavor="UNK"/></patient> | | PN | 1 … * | M | Patient Name | (HL7-IPS) | | |  | | hl7ips-dataelement-3 |  Patient's name Patient's name |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | Constraint | The Alphabetic representation of the name SHALL be always provided | | | 1 … * | R | Patient's Family Name/Surname | (HL7-IPS) | | | 1 … * | R | Patient's Given Name | (HL7-IPS) |  |  |  |  | hl7:administrativeGenderCode
|
| CE.IPS | 1 … 1 | R | Patient's Gender | (HL7-IPS) | | |  | | hl7ips-dataelement-4 |  Administrative gender Administrative gender |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | cs | 0 … 1 | F | UNK | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.1.11.1 Administrative Gender (HL7 V3) (DYNAMIC) |
| | | Example | <administrativeGenderCode code="F" codeSystem="2.16.840.1.113883.5.1" displayName="Female">
<translation code="2" codeSystem="2.16.840.1.113883.3.129.1.2.21" codeSystemName="Cinsiyet" displayName="Kadın"/></administrativeGenderCode> | | TS | 1 … 1 | R | Patient's Date of Birth. The patient date of birth may be a partial date such as only the year. | (HL7-IPS) | | |  | | hl7ips-dataelement-5 |  Date of birth Date of birth |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | 0 … * | R |
The guardians of a patient.
Other patient contacts are described using the /ClinicalDocument/participant structure. The <associatedEntity> element defines the type of contact.
| (HL7-IPS) | | cs | 1 … 1 | F | GUARD | | | Example | <guardian classCode="GUARD">
<code code="AUNT" displayName="tante" codeSystem="2.16.840.1.113883.5.111"/> <addr nullFlavor="NI"/> <telecom use="MC" value="tel:+33-12345678"/> <guardianPerson>
<name>
<family>Curie</family> <given>Marie</given> </name> </guardianPerson></guardian> | | CD.IPS | 0 … 1 | R | The relationship between the patient and the guardian or other contact may be recorded in the element. | (HL7-IPS) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.1.11.19563 PersonalRelationshipRoleType (DYNAMIC) |
| | AD.IPS | 1 … * | R | | (HL7-IPS) | | | Constraint | If there is no information, the nullFlavor attribute shall have a value of 'NI' and no address parts shall be present, otherwise there shall be no nullFlavor attribute, and at least one of the address parts listed below shall be present.
| | Included | | | from 2.16.840.1.113883.10.22.11 IPS Address (DYNAMIC) | | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.10637 PostalAddressUse (2005‑05‑01) |
| | cs | 0 … 1 | F | NI | | | Constraint | SHALL NOT have mixed content except for white space
If there is no information, the nullFlavor attribute shall have a value of 'NI' and no address parts shall be present, otherwise there shall be no nullFlavor attribute, and at least one of the address parts listed below shall be present. | | | Schematron assert | role | error | | | | test | @nullFlavor or hl7:* | | | | Message | If addr is not nullflavored at least one sub element has to be provided | | | ADXP | 0 … * | C | Subject's or Organization's Street Address Line | (HL7-IPS) | | | Schematron assert | role | error | | | | test | hl7:streetAddressLine and (hl7:city or hl7:postalCode) | | | | Message | If the address line is included either the city or the zip code has to be provided | | | ADXP | 0 … 1 | C | Subject's or Organization's City | (HL7-IPS) | | ADXP | 0 … 1 | C | Subject's or Organization's Postal Code | (HL7-IPS) | | ADXP | 0 … 1 | C | Subject's or Organization's State or Province | (HL7-IPS) | | ADXP | 0 … 1 | C | Subject's Country. | (HL7-IPS) | | | Constraint | The content of this element SHALL be selected EITHER from ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300 DYNAMIC OR MAY be selected from ISO Country Alpha-3 2.16.840.1.113883.1.11.171 DYNAMIC, IF the country is not specified in ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300. | | TEL | 1 … * | R | Guardian’s telecom information: e.g. telephone number; e-mail address. | (HL7-IPS) | | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.201 TelecommunicationAddressUse (DYNAMIC) |
| | cs | 0 … 1 | F | NI | | | Constraint | If there is no information, the nullFlavor attribute shall have a value of 'NI' and the "value" and "use" attributes shall be omitted, otherwise the nullFlavor attribute shall not be present, and the "value" and "use" attributes shall be present.
| | | 1 … 1 | R | | (HL7-IPS) | | PN | 1 … * | R | Patient Guardian's Name | (HL7-IPS) | | ENXP | 1 … * | R | Patient Guardian's Family Name/Surname | (HL7-IPS) | | ENXP | 1 … * | R | Patient Guardian's Given Name | (HL7-IPS) |  |  |  |  | hl7:languageCommunication
|
| | 0 … * | R | | (HL7-IPS) | | CS | 1 … 1 | R | Patient’s language | (HL7-IPS) | | |  | | hl7ips-dataelement-135 |  Patient’s preferred language Patient’s preferred language |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | Constraint | The two characters form SHALL be used when available; otherwise the three characters representation SHALL be adopted | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.4.642.3.21 All Languages (DYNAMIC) |
| | | Example | British English <languageCode code="en-GB"/> | | | Example | Amurdak (Australia) <languageCode code="amg-AU"/> | | | Schematron assert | role | error | | | | test | matches(@code,'[a-z]{2,3}-[A-Z]{2,3}') | | | | Message | The language code SHALL be in the form nn-CC or nnn-CCC, in accordance with BCP 47 (e.g. nn is the ISO language code; CC is ISO country code) | | | Included | 1 … * | M | from 2.16.840.1.113883.10.22.2.2 IPS CDA author (DYNAMIC) |  | hl7:author
|
| | 1 … * | M | | (HL7-IPS) |  |  | @typeCode
|
| cs | 0 … 1 | F | AUT |  |  | @contextControlCode
|
| cs | 0 … 1 | F | OP | | | Example | <author>
<time value="201212290600+0100"/> <assignedAuthor>
<id root="2.16.840.1.113883.2.9.4.3.2" extension="RSSMRA00A01F205F" assigningAuthorityName="Ministero Economia e Finanze"/> <addr use="WP">
<streetAddressLine>Viale della Cristallina 3</streetAddressLine> <city>Bologna</city> <state>BO</state> <postalCode>40121</postalCode> <country>IT</country> </addr> <telecom use="WP" value="tel:+39-051-34343434"/> <assignedPerson>
<name>
<given>Paolo</given> <family>Rossi</family> </name> </assignedPerson> </assignedAuthor> <representedOrganization>
<!-- template 'IPS CDA Organization' (dynamic) -->
</representedOrganization></author> |  |  | hl7:functionCode
|
| CE.IPS | 0 … 1 | R | | (HL7-IPS) |  |  | hl7:time
|
| TS.IPS.TZ | 1 … 1 | R | The author/time element represents the start time of the author’s participation in the creation of the clinical document. | (HL7-IPS) | | | Example | <time value="201212290600+0100"/> |  |  | hl7:assignedAuthor
|
| | 1 … 1 | R | | (HL7-IPS) | | cs | 0 … 1 | F | ASSIGNED | | II | 1 … * | R | Author Identifier(s) | (HL7-IPS) | | cs | 0 … 1 | | | | CE.IPS (extensible) | 0 … 1 | R | A code, which identifies the profession/competence/specialty of the author when it is a person. | (HL7-IPS) | | | CONF | | The value of @code should be drawn from value set 2.16.840.1.113883.11.22.53 IPS Healthcare Professional Roles (DYNAMIC) |
| | | Example | <code code="221" codeSystem="2.16.840.1.113883.2.9.6.2.7" codeSystemName="ISCO" displayName="Medical doctors"/> | | AD.IPS | 1 … * | R | | (HL7-IPS) | | | Example | <addr use="WP">
<streetAddressLine>Viale della Cristallina 3</streetAddressLine> <city>Bologna</city> <state>BO</state> <postalCode>40121</postalCode> <country>IT</country></addr> | | TEL.IPS | 1 … * | R | | (HL7-IPS) | | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.201 TelecommunicationAddressUse (DYNAMIC) |
| | url | 0 … 1 | | | | | Example | <telecom use="WP" value="tel:+39-051-34343434"/> | | | Example | <telecom nullFlavor="NI"/> | | Choice | 1 … 1 | | Elements to choose from:- hl7:assignedPerson
- hl7:assignedAuthoringDevice
| | | 0 … 1 | C | | (HL7-IPS) | | cs | 0 … 1 | F | PSN | | cs | 0 … 1 | F | INSTANCE | | PN | 1 … * | R | Name of the person (e.g. the Healthcare Professional) authoring this document | (HL7-IPS) | | | Example | <name>
<given>John</given> <family>Español Smith</family></name> | | | 1 … * | R | | (HL7-IPS) | | | 1 … * | R | | (HL7-IPS) |  |  |  |  | hl7:assignedAuthoringDevice
|
| | 0 … 1 | C | | (HL7-IPS) | | | Example | <assignedAuthoringDevice classCode="DEV" determinerCode="INSTANCE">
<softwareName displayName="Turriano"/></assignedAuthoringDevice> | | Included | | | from 2.16.840.1.113883.10.22.9.2 IPS CDA Device (DYNAMIC) | | cs | 0 … 1 | F | DEV | | cs | 0 … 1 | F | INSTANCE | | CE | 0 … 1 | | | (HL7-IPS) |  |  |  |  |  | hl7:manufacturerModelName
|
| SC | 0 … 1 | | | (HL7-IPS) | | SC | 0 … 1 | | | (HL7-IPS) |  |  |  | hl7:representedOrganization
|
| | 0 … 1 | R | | (HL7-IPS) | | Included | | | from 2.16.840.1.113883.10.22.9.1 IPS CDA Organization (DYNAMIC) | | cs | 1 … 1 | F | ORG | | cs | 1 … 1 | F | INSTANCE | | II | 1 … * | R | | (HL7-IPS) | | cs | 0 … 1 | | | | ON | 1 … 1 | R | | (HL7-IPS) | | cs | 0 … 1 | | | | TEL | 1 … * | R | | (HL7-IPS) | | set_cs | 1 … 1 | R | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.201 TelecommunicationAddressUse (DYNAMIC) |
| | cs | 0 … 1 | | | | | Constraint | If there is no information, the nullFlavor attribute shall have a value of 'NI' and the "value" and "use" attributes shall be omitted, otherwise the nullFlavor attribute shall not be present, and the "value" and "use" attributes shall be present.
| | AD.IPS | 1 … 1 | R | | (HL7-IPS) | | Included | | | from 2.16.840.1.113883.10.22.11 IPS Address (DYNAMIC) | | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.10637 PostalAddressUse (2005‑05‑01) |
| | cs | 0 … 1 | F | NI | | | Constraint | SHALL NOT have mixed content except for white space
If there is no information, the nullFlavor attribute shall have a value of 'NI' and no address parts shall be present, otherwise there shall be no nullFlavor attribute, and at least one of the address parts listed below shall be present. | | | Schematron assert | role | error | | | | test | @nullFlavor or hl7:* | | | | Message | If addr is not nullflavored at least one sub element has to be provided | | | ADXP | 0 … * | C | Subject's or Organization's Street Address Line | (HL7-IPS) | | | Schematron assert | role | error | | | | test | hl7:streetAddressLine and (hl7:city or hl7:postalCode) | | | | Message | If the address line is included either the city or the zip code has to be provided | | | ADXP | 0 … 1 | C | Subject's or Organization's City | (HL7-IPS) | | ADXP | 0 … 1 | C | Subject's or Organization's Postal Code | (HL7-IPS) | | ADXP | 0 … 1 | C | Subject's or Organization's State or Province | (HL7-IPS) | | ADXP | 0 … 1 | C | Subject's Country. | (HL7-IPS) | | | Constraint | The content of this element SHALL be selected EITHER from ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300 DYNAMIC OR MAY be selected from ISO Country Alpha-3 2.16.840.1.113883.1.11.171 DYNAMIC, IF the country is not specified in ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300. | | Included | 1 … 1 | M | from 2.16.840.1.113883.10.22.2.3 IPS CDA custodian (DYNAMIC) |  | hl7:custodian
|
| | 1 … 1 | M | | (HL7-IPS) |  |  | @typeCode
|
| cs | 0 … 1 | F | CST | | | Example | <custodian typeCode="CST">
<assignedCustodian classCode="ASSIGNED">
<representedCustodianOrganization classCode="ORG" determinerCode="INSTANCE">
<!-- template 'IPS CDA Organization' (dynamic) -->
</representedCustodianOrganization> </assignedCustodian></custodian> |  |  | hl7:assignedCustodian
|
| | 1 … 1 | R | | (HL7-IPS) | | cs | 0 … 1 | F | ASSIGNED |  |  |  | hl7:representedCustodianOrganization
|
| | 1 … 1 | R | | (HL7-IPS) | | cs | 0 … 1 | F | ORG | | cs | 0 … 1 | F | INSTANCE | | II | 1 … * | R | | (HL7-IPS) | | cs | 0 … 1 | | | | ON | 1 … 1 | R | | (HL7-IPS) | | cs | 0 … 1 | | | | TEL | 1 … 1 | R | | (HL7-IPS) | | set_cs | 1 … 1 | R | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.201 TelecommunicationAddressUse (DYNAMIC) |
| | cs | 0 … 1 | | | | | Constraint | If there is no information, the nullFlavor attribute shall have a value of 'NI' and the "value" and "use" attributes shall be omitted, otherwise the nullFlavor attribute shall not be present, and the "value" and "use" attributes shall be present.
| | AD.IPS | 1 … 1 | R | | (HL7-IPS) | | Included | | | from 2.16.840.1.113883.10.22.11 IPS Address (DYNAMIC) | | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.10637 PostalAddressUse (2005‑05‑01) |
| | cs | 0 … 1 | F | NI | | | Constraint | SHALL NOT have mixed content except for white space
If there is no information, the nullFlavor attribute shall have a value of 'NI' and no address parts shall be present, otherwise there shall be no nullFlavor attribute, and at least one of the address parts listed below shall be present. | | | Schematron assert | role | error | | | | test | @nullFlavor or hl7:* | | | | Message | If addr is not nullflavored at least one sub element has to be provided | | | ADXP | 0 … * | C | Subject's or Organization's Street Address Line | (HL7-IPS) | | | Schematron assert | role | error | | | | test | hl7:streetAddressLine and (hl7:city or hl7:postalCode) | | | | Message | If the address line is included either the city or the zip code has to be provided | | | ADXP | 0 … 1 | C | Subject's or Organization's City | (HL7-IPS) | | ADXP | 0 … 1 | C | Subject's or Organization's Postal Code | (HL7-IPS) | | ADXP | 0 … 1 | C | Subject's or Organization's State or Province | (HL7-IPS) | | ADXP | 0 … 1 | C | Subject's Country. | (HL7-IPS) | | | Constraint | The content of this element SHALL be selected EITHER from ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300 DYNAMIC OR MAY be selected from ISO Country Alpha-3 2.16.840.1.113883.1.11.171 DYNAMIC, IF the country is not specified in ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300. | | Included | 0 … 1 | R | from 2.16.840.1.113883.10.22.2.4 IPS CDA legalAuthenticator (DYNAMIC) |  | hl7:legalAuthenticator
|
| | 0 … 1 | R | | (HL7-IPS) | | | Example | <legalAuthenticator>
<time value="20111013150937-0800"/> <signatureCode code="S"/> <assignedEntity>
<id extension="admin" root="2.16.17.710.780.1000.903.1.1.3.3"/> <assignedPerson>
<name>
<given>John</given> <family>Español Smith</family> </name> </assignedPerson> <representedOrganization>
<name>Healthcare Facility's name</name> <addr>
<country>NL</country> <streetName>Duinweg</streetName> <houseNumber>23</houseNumber> <postalCode>7364 RX</postalCode> <city>Amsterdam</city> </addr> </representedOrganization> </assignedEntity></legalAuthenticator> |  |  | hl7:time
|
| TS.IPS.TZ | 1 … 1 | M | Time of signing the document | (HL7-IPS) |  |  | hl7:signatureCode
|
| CS | 0 … 1 | R | Signature code | (HL7-IPS) | | CONF | 0 … 1 | F | S |  |  | hl7:assignedEntity
|
| | 0 … 1 | R | The entity that is responsible for the legal authentication of the CDA document | (HL7-IPS) | | | 1 … * | R | Unique identification of legal authenticator | (HL7-IPS) | | AD.IPS | 1 … * | R | | (HL7-IPS) | | TEL.IPS | 1 … * | R | | (HL7-IPS) | | | 1 … 1 | R | | (HL7-IPS) | | cs | 0 … 1 | F | PSN | | cs | 0 … 1 | F | INSTANCE | | PN | 1 … * | R | Name of the legal authenticator | (HL7-IPS) | | | Example | <name>
<given>John</given> <family>Español Smith</family></name> | | | 1 … * | R | HP Family Name/Surname | (HL7-IPS) | | | 1 … * | R | HP Given Name | (HL7-IPS) |  |  |  | hl7:representedOrganization
|
| | 1 … 1 | M | Organization the legal authenticator is acting for
Contains 2.16.840.1.113883.10.22.9.1 IPS CDA Organization (DYNAMIC) | (HL7-IPS) | | Included | 0 … * | | from 2.16.840.1.113883.10.22.2.5 IPS Patient Contacts (DYNAMIC) |  | hl7:participant
|
| | 0 … * | R | Patient contacts or the Preferred Health Professional to contact in case of emergency. | (HL7-IPS) | | where [hl7:templateId/@root='2.16.840.1.113883.10.22.2.5'] | | | |  | | hl7ips-dataelement-154 |  Patient’s Address Book Patient’s Address Book |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-163 |  Preferred Healthcare providers Preferred Healthcare providers |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | @typeCode
|
| cs | 1 … 1 | F | IND | | | Example | <participant typeCode="IND">
<templateId root="2.16.840.1.113883.10.22.2.5"/> <associatedEntity classCode="NOK">
<addr>
<streetAddressLine>Promenade des Anglais 111</streetAddressLine> <city>Lyon</city> <postalCode>69001</postalCode> <country>FR</country> </addr> <telecom value="tel:(+33)555-20036" use="WP"/> <associatedPerson>
<name>
<given>Martha</given> <family>Mum</family> </name> </associatedPerson> </associatedEntity></participant> |  |  | hl7:templateId
|
| II | 1 … 1 | M | | (HL7-IPS) | | uid | 1 … 1 | F | 2.16.840.1.113883.10.22.2.5 |  |  | hl7:functionCode
|
| | 0 … 1 | C | The <functionCode> element may be used to indicate that this participant is the preferred Health Professional to contact in case of emergency.</functionCode> | (HL7-IPS) | | CONF | 0 … 1 | F | PCP | | 0 … 1 | F | 2.16.840.1.113883.5.88 (Participation Function) |  |  | hl7:associatedEntity
|
| | | R | The <associatedEntity> element identifies the type of contact. </associatedEntity> | (HL7-IPS) | | cs | 1 … 1 | R | | | | CONF | | The value of @classCode shall be drawn from value set 2.16.840.1.113883.11.20.9.33 INDRoleclassCodes (DYNAMIC) |
| | | Example | <associatedEntity classCode="ECON">
<addr>
<streetAddressLine>Karl Strasse</streetAddressLine> <city>Freiberg</city> <postalCode>09599</postalCode> <country>DE</country> </addr> <telecom value="tel:+49-761-11110000" use="WP"/> <associatedPerson>
<name>
<given>Arzt</given> <family>Guter</family> </name> </associatedPerson></associatedEntity> | | CV.IPS | 0 … 1 | R | This element indicates the relationship between the patient and this participant. | (HL7-IPS) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.54 IPS Personal Relationship (DYNAMIC) | | or | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.53 IPS Healthcare Professional Roles (DYNAMIC) |
| | | Example | <code code="AUNT" displayName="θεία" codeSystem="2.16.840.1.113883.5.111"/> | | AD.IPS | 1 … * | R | Patient Contact's / Preferred HP's Address | (HL7-IPS) | | | Schematron assert | role | error | | | | test | @nullFlavor or hl7:* | | | | Message | If addr is not nullflavored at least one sub element has to be provided | | | Included | | | from 2.16.840.1.113883.10.22.11 IPS Address (DYNAMIC) | | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.10637 PostalAddressUse (2005‑05‑01) |
| | cs | 0 … 1 | F | NI | | | Constraint | SHALL NOT have mixed content except for white space
If there is no information, the nullFlavor attribute shall have a value of 'NI' and no address parts shall be present, otherwise there shall be no nullFlavor attribute, and at least one of the address parts listed below shall be present. | | | Schematron assert | role | error | | | | test | @nullFlavor or hl7:* | | | | Message | If addr is not nullflavored at least one sub element has to be provided | | | ADXP | 0 … * | C | Subject's or Organization's Street Address Line | (HL7-IPS) | | | Schematron assert | role | error | | | | test | hl7:streetAddressLine and (hl7:city or hl7:postalCode) | | | | Message | If the address line is included either the city or the zip code has to be provided | | | ADXP | 0 … 1 | C | Subject's or Organization's City | (HL7-IPS) | | ADXP | 0 … 1 | C | Subject's or Organization's Postal Code | (HL7-IPS) | | ADXP | 0 … 1 | C | Subject's or Organization's State or Province | (HL7-IPS) | | ADXP | 0 … 1 | C | Subject's Country. | (HL7-IPS) | | | Constraint | The content of this element SHALL be selected EITHER from ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300 DYNAMIC OR MAY be selected from ISO Country Alpha-3 2.16.840.1.113883.1.11.171 DYNAMIC, IF the country is not specified in ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300. | | TEL | 1 … * | R | Patient Contact's / Preferred HP's/Legal Organization telephone or e-mail <telecom> element is required.</telecom> | (HL7-IPS) | | |  | | hl7ips-dataelement-169 |  Telecoms Telecoms |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-174 |  Telecoms Telecoms |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.201 TelecommunicationAddressUse (DYNAMIC) |
| | cs | 0 … 1 | F | NI | | | Constraint | If there is no information, the nullFlavor attribute shall have a value of 'NI' and the "value" and "use" attributes shall be omitted, otherwise the nullFlavor attribute shall not be present, and the "value" and "use" attributes shall be present
| | | Example | <telecom use="WP" value="tel:+45 20 7025 6161"/><telecom use="HP" value="mailto:jsmith@myprovider.co.uk"/> | | Choice | 1 … 2 | | Elements to choose from:- hl7:associatedPerson
- hl7:scopingOrganization
| | | 0 … 1 | C | Or the associatedPerson, or the scopingOrganization, or both elements shall be provided | (HL7-IPS) | | |  | | hl7ips-dataelement-165 |  Healthcare Provider (person) Healthcare Provider (person) |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | PN | 1 … * | R | Patient Contact's Name / Preferred HP's Name | (HL7-IPS) | | |  | | hl7ips-dataelement-121 |  Name Name |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | Example | <name>
<given>John</given> <family>Español Smith</family></name> | | | 1 … * | R | Patient Contact's Family Name/Surname / Preferred HP's Family Name/Surname | (HL7-IPS) | | | 1 … * | R | Patient Contact's Given Name / Preferred HP's Given Name | (HL7-IPS) | | | 0 … 1 | C | Or the associatedPerson, or the scopingOrganization, or both elements shall be provided | (HL7-IPS) | | |  | | hl7ips-dataelement-166 |  Healthcare Provider (organisation) Healthcare Provider (organisation) |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | ON | 1 … * | R | Organization's Name | (HL7-IPS) | | |  | | hl7ips-dataelement-172 |  Organisation’s Name Organisation’s Name |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | Included | 1 … 1 | M | from 2.16.840.1.113883.10.22.2.6 IPS CDA documentationOf (DYNAMIC) |  | hl7:documentationOf
|
| | 1 … 1 | M | The documentationOf relationship in an International Patient Summary contains the representation of providers who are wholly or partially responsible for the safety and well-being of a subject of care. | (HL7-IPS) |  |  | @typeCode
|
| cs | 0 … 1 | F | DOC | | | Example | <documentationOf>
<serviceEvent classCode="PCPR">
<effectiveTime>
<low nullFlavor="NI"/> <high value="20110308"/> </effectiveTime> <performer typeCode="PRF">
<!-- See example below -->
</performer> </serviceEvent></documentationOf> |  |  | hl7:serviceEvent
|
| | 1 … 1 | R | The main activity being described by a IPS is the provision of healthcare over a period of time. This is shown by setting the value of serviceEvent/@classCode to “PCPR” (care provision) and indicating the duration over which care was provided in serviceEvent/effectiveTime. Additional data from outside this duration may also be included if it is relevant to care provided during that time range (e.g., reviewed during the stated time range).
For example if the IPS is generated by a GP based on information recorded in his/her EHR-S, then the low value should represent the date when the treatment relationship between the patient and the GP started; and the high value the date of the latest care event. | (HL7-IPS) | | cs | 1 … 1 | F | PCPR | | cs | 1 … 1 | F | EVN | | II | 0 … * | R | | (HL7-IPS) | | IVL_TS | 1 … 1 | R | | (HL7-IPS) | | TS | 1 … 1 | R | | (HL7-IPS) | | TS | 1 … 1 | R | | (HL7-IPS) | | | 0 … * | R | It represents the healthcare providers involved in the current or pertinent historical care of the patient. Preferably, the patient’s key healthcare providers would be listed, particularly their primary physician and any active consulting physicians, therapists, and counselors | (HL7-IPS) | | cs | 1 … 1 | R | | | | CONF | | The value of @typeCode shall be drawn from value set 2.16.840.1.113883.1.11.19601 x_ServiceEventPerformer (DYNAMIC) |
| | | Example | <performer typeCode="PRF">
<assignedEntity>
<id assigningAuthorityName="MEF" displayable="false" extension="DVLMMG57R07F205G" root="2.16.840.1.113883.2.9.4.3.2"/> <code code="221" codeSystem="2.16.840.1.113883.2.9.6.2.7" codeSystemName="ISCO" displayName="Medical doctors">
<translation codeSystem="2.16.840.1.113883.2.9.5.1.111" code="MMG" displayName="Medico di Medicina Generale"/> </code> <addr nullFlavor="NI"/> <telecom nullFlavor="NI"/> <assignedPerson>
<name>
<family>DVALUNO</family> <given>MMG</given> </name> </assignedPerson> <representedOrganization>
<id assigningAuthorityName="A.S.L. DELLA PROVINCIA DI LECCO" extension="030305" root="2.16.840.1.113883.2.9.4.1.1"/> <name>A.S.L. DELLA PROVINCIA DI LECCO</name> <telecom nullFlavor="NI"/> <addr>
<state>LECCO</state> <city>LECCO</city> <country>IT</country> <postalCode>23900</postalCode> <streetAddressLine>CORSO CARLO ALBERTO,120</streetAddressLine> </addr> </representedOrganization> </assignedEntity></performer> | | CE.IPS | 0 … 1 | R | | (HL7-IPS) | | IVL_TS.IPS.TZ | 0 … 1 | R | | (HL7-IPS) | | | 1 … 1 | M | | (HL7-IPS) | | II | 1 … * | R | Healthcare provider ID number | (HL7-IPS) | | CE.IPS (extensible) | 0 … 1 | R | It describes the professional role of the healthcare provider involved in the current or pertinent historical care of the patient. | (HL7-IPS) | | | CONF | | The value of @code should be drawn from value set 2.16.840.1.113883.11.22.53 IPS Healthcare Professional Roles (DYNAMIC) |
| | AD.IPS | 1 … * | R | | (HL7-IPS) | | TEL.IPS | 1 … * | R | | (HL7-IPS) | | | 0 … 1 | | | (HL7-IPS) | | Included | | | from 2.16.840.1.113883.10.22.9.3 IPS CDA Person (DYNAMIC) | | cs | 0 … 1 | F | PSN | | cs | 0 … 1 | F | INSTANCE | | PN | 1 … * | R | | (HL7-IPS) |  |  |  |  |  | hl7:representedOrganization
|
| | 0 … 1 | | Contains 2.16.840.1.113883.10.22.9.1 IPS CDA Organization (DYNAMIC) | (HL7-IPS) | | Included | 0 … 2 | R | from 2.16.840.1.113883.10.22.2.7 IPS CDA relatedDocument (DYNAMIC) | | | Constraint | A conformant CDA document can have:
- a single relatedDocument with typeCode "APND"; OR
- a single relatedDocument with typeCode "RPLC"; OR
- a single relatedDocument with typeCode "XFRM"; OR
- a combination of two relatedDocuments with typeCodes "XFRM" and "RPLC"; OR
- a combination of two relatedDocuments with typeCodes "XFRM" and "APND".
No other combinations are allowed.
|  | hl7:relatedDocument
|
| | 0 … 2 | R | | (HL7-IPS) |  |  | @typeCode
|
| cs | 1 … 1 | R | | | | CONF | | The value of @typeCode shall be drawn from value set 2.16.840.1.113883.1.11.11610 x_ActRelationshipDocument (DYNAMIC) |
|  |  | hl7:parentDocument
|
| | 1 … 1 | R | | (HL7-IPS) | | cs | 0 … 1 | F | DOCCLIN | | cs | 0 … 1 | F | EVN | | II | 1 … * | R | | (HL7-IPS) | | CD.IPS | 0 … 1 | R | | (HL7-IPS) | | CONF | 0 … 1 | F | 2.16.840.1.113883.6.1 (LOINC) | | ED | 0 … 1 | R | | (HL7-IPS) | | II | 0 … 1 | R | | (HL7-IPS) | | INT | 0 … 1 | R | | (HL7-IPS) |  | hl7:component
|
| | 1 … 1 | M | | (HL7-IPS) |  |  | hl7:structuredBody
|
| | 1 … 1 | M | Note: the proposed order of the sections hereafter indicated is not mandatory | (HL7-IPS) | | cs | 0 … 1 | F | DOCBODY | | | 1 … 1 | M | Contains 2.16.840.1.113883.10.22.3.1 IPS Medication Summary Section (DYNAMIC) | (HL7-IPS) | | | 1 … 1 | M | Contains 2.16.840.1.113883.10.22.3.2 IPS Allergies and Intolerances Section (DYNAMIC) | (HL7-IPS) | | | 1 … 1 | M | Contains 2.16.840.1.113883.10.22.3.3 IPS Problems Section (DYNAMIC) | (HL7-IPS) | | | 0 … 1 | R | Contains 2.16.840.1.113883.10.22.3.4 IPS History of Procedures Section (DYNAMIC) | (HL7-IPS) | | | 0 … 1 | R | Contains 2.16.840.1.113883.10.22.3.5 IPS Immunizations Section (DYNAMIC) | (HL7-IPS) | | | 0 … 1 | R | Contains 2.16.840.1.113883.10.22.3.6 IPS Medical Devices Section (DYNAMIC) | (HL7-IPS) | | | 0 … 1 | R | Contains 2.16.840.1.113883.10.22.3.14 IPS Results Section (DYNAMIC) | (HL7-IPS) | | | 0 … 1 | R | Contains 1.3.6.1.4.1.19376.1.5.3.1.1.5.3.2 IHE Coded Vital Signs Section (DYNAMIC) | (HL7-IPS) | | | 0 … 1 | | Contains 2.16.840.1.113883.10.22.3.7 IPS History of Past Illness Section (DYNAMIC) | (HL7-IPS) | | | 0 … 1 | | Contains 2.16.840.1.113883.10.22.3.8 IPS Functional Status Section (DYNAMIC) | (HL7-IPS) | | | 0 … 1 | | Contains 2.16.840.1.113883.10.22.3.9 IPS Plan of Care Section (DYNAMIC) | (HL7-IPS) | | | 0 … 1 | | Contains 2.16.840.1.113883.10.22.3.10 IPS Social History Section (DYNAMIC) | (HL7-IPS) | | | 0 … 1 | | Contains 2.16.840.1.113883.10.22.3.11 IPS History of Pregnancy Section (DYNAMIC) | (HL7-IPS) | | | 0 … 1 | | Contains 2.16.840.1.113883.10.22.3.12 IPS Advance Directives Section (DYNAMIC) | (HL7-IPS) | | | 0 … 1 | | Contains 2.16.840.1.113883.10.22.15 IPS Alerts Section (DYNAMIC) | (HL7-IPS) | | | 0 … 1 | | Contains 2.16.840.1.113883.10.22.16 IPS Patient Story Section (DYNAMIC) | (HL7-IPS) |
|
IPS CDA author
| Id | 2.16.840.1.113883.10.22.2.2 | Effective Date | 2017‑04‑11 |
|---|
| Status |  Under pre-publication review Under pre-publication review | Version Label | STU1 |
|---|
| Name | IPSCDAauthor | Display Name | IPS CDA author |
|---|
| Description | A CDA document shall have at least one author. Authors could be either human (ClinicalDocument/author/assignedAuthor/assignedPerson) either devices (ClinicalDocument/author/assignedAuthor/assignedAuthoringDevice). For definition “The author element represents the creator of the clinical document. If the role of the actor is the entry of information
from his or her own knowledge or application of skills, that actor is the author. If one actor provides information to another actor who filters, reasons, or algorithmically creates new information, then that second actor is also an author, having created information from his or her own knowledge or skills.” [From Implementation Guide for CDA Release 2: Imaging Integration – UV Realm, March 2009]. According to this definition, not any device that generates the electronic document has to be considered as an author: - a spider collecting and filtering information from different repositories, according to defined rules and policies, for the scope of creating a Patient Summary is definitely a document
author (and in some cases the only one );
- an application that transforms a Patient Summary record into this CDA format may not be an author;
- For cross-border exchange purposes, a device, which modifies the concepts conveyed (e.g. applying code system mappings), should appear as one of the authors. In this case (document generated through a transformation
process) the authors of the parent (original) patient summary should appear as authors as well.
Further to this, authorship can give information about the nature of Patient Summary : - if there is a person author only, then the Patient Summary is the result of a practitioner clinical act;
- if there are device authors only, the summary was
automatically generated according to well defined rules defined by the responsible organization.
The CDA standard allows to provide detailed information about what was authored by whom in the Patient Summary, allowing the specification of authorship at the whole document level, at the section level and also at the entry level. In any case it is not required to repeat this information for each of the mentioned levels, taking advantage of the context conduction propriety. In fact “context that is specified on an outer tag holds true for all nested tags, unless overridden on a nested tag. Context specified on a tag within the CDA body always overrides context propagated from an outer tag. For instance, the specification of authorship at a document section level overrides all authorship propagated from outer tags.” (HL7 CDA R2 Standard). |
|
| Classification | CDA Header Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Uses | | Uses 2 templates | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.9.2 | Include |  IPS CDA Device (STU1) IPS CDA Device (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.9.1 | Include |  IPS CDA Organization (STU1) IPS CDA Organization (STU1) | DYNAMIC |
|
|
|---|
| Relationship | Adaptation: template 2.16.840.1.113883.10.12.102 CDA author (2005‑09‑07) ref ad1bbr- |
|---|
| Example | | Human Author | <author>
<time value="201212290600+0100"/> <assignedAuthor>
<id root="2.16.840.1.113883.2.9.4.3.2" extension="RSSMRA00A01F205F" assigningAuthorityName="Ministero Economia e Finanze"/> <code code="221" codeSystem="2.16.840.1.113883.2.9.6.2.7" codeSystemName="ISCO" displayName="Medico"/> <addr use="WP">
<streetAddressLine>Viale della Cristallina 3</streetAddressLine> <city>Bologna</city> <state>BO</state> <postalCode>40121</postalCode> <country>IT</country> </addr> <telecom use="WP" value="tel:+39-051-34343434"/> <assignedPerson>
<name>
<given>Paolo</given> <family>Rossi</family> </name> </assignedPerson> <representedOrganization>
<!-- template 'IPS CDA Organization' (dynamic) -->
</representedOrganization> </assignedAuthor></author> |
|
|---|
| Example | | Device Author | <author>
<time value="201212290600+0100"/> <assignedAuthor>
<id root="1.2.3.999" extension="__example only__"/> <addr use="WP">
<state>Castilla-La Mancha</state> <city>Toledo</city> <precinct>Toledo</precinct> <country>ES</country> <postalCode>45071</postalCode> <streetAddressLine>Av. Río Guadiana, 4</streetAddressLine> </addr> <telecom nullFlavor="NI"/> <assignedAuthoringDevice classCode="DEV" determinerCode="INSTANCE">
<softwareName displayName="Turriano"/> </assignedAuthoringDevice> <representedOrganization classCode="ORG" determinerCode="INSTANCE">
<id root="1.2.3.999" extension="__example only__"/> <name>SESCAM</name> <telecom use="WP" value="tel:+34925274100"/> <addr use="WP">
<state>Castilla-La Mancha</state> <city>Toledo</city> <precinct>Toledo</precinct> <country>ES</country> <postalCode>45071</postalCode> <streetAddressLine>Av. Río Guadiana, 4</streetAddressLine> </addr> </representedOrganization> </assignedAuthor></author> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | 1 … * | R | | (IPS...hor) |  | @typeCode
|
| cs | 0 … 1 | F | AUT |  | @contextControlCode
|
| cs | 0 … 1 | F | OP | | | Example | <author>
<time value="201212290600+0100"/> <assignedAuthor>
<id root="2.16.840.1.113883.2.9.4.3.2" extension="RSSMRA00A01F205F" assigningAuthorityName="Ministero Economia e Finanze"/> <addr use="WP">
<streetAddressLine>Viale della Cristallina 3</streetAddressLine> <city>Bologna</city> <state>BO</state> <postalCode>40121</postalCode> <country>IT</country> </addr> <telecom use="WP" value="tel:+39-051-34343434"/> <assignedPerson>
<name>
<given>Paolo</given> <family>Rossi</family> </name> </assignedPerson> </assignedAuthor> <representedOrganization>
<!-- template 'IPS CDA Organization' (dynamic) -->
</representedOrganization></author> |  | hl7:functionCode
|
| CE.IPS | 0 … 1 | R | | (IPS...hor) |  | hl7:time
|
| TS.IPS.TZ | 1 … 1 | R | The author/time element represents the start time of the author’s participation in the creation of the clinical document. | (IPS...hor) | | | Example | <time value="201212290600+0100"/> |  | hl7:assignedAuthor
|
| | 1 … 1 | R | | (IPS...hor) |  |  | @classCode
|
| cs | 0 … 1 | F | ASSIGNED |  |  | hl7:id
|
| II | 1 … * | R | Author Identifier(s) | (IPS...hor) | | cs | 0 … 1 | | |  |  | hl7:code
|
| CE.IPS (extensible) | 0 … 1 | R | A code, which identifies the profession/competence/specialty of the author when it is a person. | (IPS...hor) | | | CONF | | The value of @code should be drawn from value set 2.16.840.1.113883.11.22.53 IPS Healthcare Professional Roles (DYNAMIC) |
| | | Example | <code code="221" codeSystem="2.16.840.1.113883.2.9.6.2.7" codeSystemName="ISCO" displayName="Medical doctors"/> |  |  | hl7:addr
|
| AD.IPS | 1 … * | R | | (IPS...hor) | | | Example | <addr use="WP">
<streetAddressLine>Viale della Cristallina 3</streetAddressLine> <city>Bologna</city> <state>BO</state> <postalCode>40121</postalCode> <country>IT</country></addr> |  |  | hl7:telecom
|
| TEL.IPS | 1 … * | R | | (IPS...hor) | | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.201 TelecommunicationAddressUse (DYNAMIC) |
| | url | 0 … 1 | | | | | Example | <telecom use="WP" value="tel:+39-051-34343434"/> | | | Example | <telecom nullFlavor="NI"/> | | Choice | 1 … 1 | | Elements to choose from:- hl7:assignedPerson
- hl7:assignedAuthoringDevice
| | | 0 … 1 | C | | (IPS...hor) | | cs | 0 … 1 | F | PSN | | cs | 0 … 1 | F | INSTANCE | | PN | 1 … * | R | Name of the person (e.g. the Healthcare Professional) authoring this document | (IPS...hor) | | | Example | <name>
<given>John</given> <family>Español Smith</family></name> | | | 1 … * | R | | (IPS...hor) | | | 1 … * | R | | (IPS...hor) |  |  |  | hl7:assignedAuthoringDevice
|
| | 0 … 1 | C | | (IPS...hor) | | | Example | <assignedAuthoringDevice classCode="DEV" determinerCode="INSTANCE">
<softwareName displayName="Turriano"/></assignedAuthoringDevice> | | Included | | | from 2.16.840.1.113883.10.22.9.2 IPS CDA Device (DYNAMIC) | | cs | 0 … 1 | F | DEV | | cs | 0 … 1 | F | INSTANCE | | CE | 0 … 1 | | | (IPS...hor) |  |  |  |  | hl7:manufacturerModelName
|
| SC | 0 … 1 | | | (IPS...hor) | | SC | 0 … 1 | | | (IPS...hor) |  |  | hl7:representedOrganization
|
| | 0 … 1 | R | | (IPS...hor) | | Included | | | from 2.16.840.1.113883.10.22.9.1 IPS CDA Organization (DYNAMIC) | | cs | 1 … 1 | F | ORG | | cs | 1 … 1 | F | INSTANCE | | II | 1 … * | R | | (IPS...hor) | | cs | 0 … 1 | | | | ON | 1 … 1 | R | | (IPS...hor) | | cs | 0 … 1 | | | | TEL | 1 … * | R | | (IPS...hor) | | set_cs | 1 … 1 | R | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.201 TelecommunicationAddressUse (DYNAMIC) |
| | cs | 0 … 1 | | | | | Constraint | If there is no information, the nullFlavor attribute shall have a value of 'NI' and the "value" and "use" attributes shall be omitted, otherwise the nullFlavor attribute shall not be present, and the "value" and "use" attributes shall be present.
| | AD.IPS | 1 … 1 | R | | (IPS...hor) | | Included | | | from 2.16.840.1.113883.10.22.11 IPS Address (DYNAMIC) | | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.10637 PostalAddressUse (2005‑05‑01) |
| | cs | 0 … 1 | F | NI | | | Constraint | SHALL NOT have mixed content except for white space
If there is no information, the nullFlavor attribute shall have a value of 'NI' and no address parts shall be present, otherwise there shall be no nullFlavor attribute, and at least one of the address parts listed below shall be present. | | | Schematron assert | role | error | | | | test | @nullFlavor or hl7:* | | | | Message | If addr is not nullflavored at least one sub element has to be provided | | | ADXP | 0 … * | C | Subject's or Organization's Street Address Line | (IPS...hor) | | | Schematron assert | role | error | | | | test | hl7:streetAddressLine and (hl7:city or hl7:postalCode) | | | | Message | If the address line is included either the city or the zip code has to be provided | | | ADXP | 0 … 1 | C | Subject's or Organization's City | (IPS...hor) | | ADXP | 0 … 1 | C | Subject's or Organization's Postal Code | (IPS...hor) | | ADXP | 0 … 1 | C | Subject's or Organization's State or Province | (IPS...hor) | | ADXP | 0 … 1 | C | Subject's Country. | (IPS...hor) | | | Constraint | The content of this element SHALL be selected EITHER from ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300 DYNAMIC OR MAY be selected from ISO Country Alpha-3 2.16.840.1.113883.1.11.171 DYNAMIC, IF the country is not specified in ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300. |
|
IPS CDA custodian
| Id | 2.16.840.1.113883.10.22.2.3 | Effective Date | 2021‑08‑04 12:13:07Other versions this id:  IPSCDAcustodian as of 2017‑04‑11 IPSCDAcustodian as of 2017‑04‑11
|
|---|
| Status |  Draft Draft | Version Label | 2021 |
|---|
| Name | IPSCDAcustodian | Display Name | IPS CDA custodian |
|---|
| Description |
The custodian element represents the organization that is in charge of maintaining and is entrusted with the care of the document.
This information is required by the CDA R2 standard and shall be recorded in the ClinicalDocument/custodian/assignedCustodian/ representedCustodianOrganization element.
There is only one custodian per CDA document. Allowing that a CDA document may not represent the original form of the authenticated document, the custodian represents the steward of the original source document. The custodian may be the document originator, a health information exchange, or other responsible party.
|
|
| Classification | CDA Header Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Uses | | Uses 1 template | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.11 | Include |  IPS Address (STU1) IPS Address (STU1) | DYNAMIC |
|
|
|---|
| Relationship | Version: template 2.16.840.1.113883.10.22.2.3 IPS CDA custodian (2017‑04‑11)
Adaptation: template 2.16.840.1.113883.10.12.104 CDA custodian (2005‑09‑07) ref ad1bbr- |
|---|
| Example | | Example | <custodian typeCode="CST">
<assignedCustodian classCode="ASSIGNED">
<representedCustodianOrganization classCode="ORG" determinerCode="INSTANCE">
<!-- template 'IPS CDA Organization' (dynamic) -->
</representedCustodianOrganization> </assignedCustodian></custodian> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | 1 … 1 | R | | (IPS...ian) |  | @typeCode
|
| cs | 0 … 1 | F | CST | | | Example | <custodian typeCode="CST">
<assignedCustodian classCode="ASSIGNED">
<representedCustodianOrganization classCode="ORG" determinerCode="INSTANCE">
<!-- template 'IPS CDA Organization' (dynamic) -->
</representedCustodianOrganization> </assignedCustodian></custodian> |  | hl7:assignedCustodian
|
| | 1 … 1 | R | | (IPS...ian) |  |  | @classCode
|
| cs | 0 … 1 | F | ASSIGNED |  |  | hl7:representedCustodianOrganization
|
| | 1 … 1 | R | | (IPS...ian) | | cs | 0 … 1 | F | ORG | | cs | 0 … 1 | F | INSTANCE | | II | 1 … * | R | | (IPS...ian) | | cs | 0 … 1 | | | | ON | 1 … 1 | R | | (IPS...ian) | | cs | 0 … 1 | | | | TEL | 1 … 1 | R | | (IPS...ian) | | set_cs | 1 … 1 | R | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.201 TelecommunicationAddressUse (DYNAMIC) |
| | cs | 0 … 1 | | | | | Constraint | If there is no information, the nullFlavor attribute shall have a value of 'NI' and the "value" and "use" attributes shall be omitted, otherwise the nullFlavor attribute shall not be present, and the "value" and "use" attributes shall be present.
| | AD.IPS | 1 … 1 | R | | (IPS...ian) | | Included | | | from 2.16.840.1.113883.10.22.11 IPS Address (DYNAMIC) | | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.10637 PostalAddressUse (2005‑05‑01) |
| | cs | 0 … 1 | F | NI | | | Constraint | SHALL NOT have mixed content except for white space
If there is no information, the nullFlavor attribute shall have a value of 'NI' and no address parts shall be present, otherwise there shall be no nullFlavor attribute, and at least one of the address parts listed below shall be present. | | | Schematron assert | role | error | | | | test | @nullFlavor or hl7:* | | | | Message | If addr is not nullflavored at least one sub element has to be provided | | | ADXP | 0 … * | C | Subject's or Organization's Street Address Line | (IPS...ian) | | | Schematron assert | role | error | | | | test | hl7:streetAddressLine and (hl7:city or hl7:postalCode) | | | | Message | If the address line is included either the city or the zip code has to be provided | | | ADXP | 0 … 1 | C | Subject's or Organization's City | (IPS...ian) | | ADXP | 0 … 1 | C | Subject's or Organization's Postal Code | (IPS...ian) | | ADXP | 0 … 1 | C | Subject's or Organization's State or Province | (IPS...ian) | | ADXP | 0 … 1 | C | Subject's Country. | (IPS...ian) | | | Constraint | The content of this element SHALL be selected EITHER from ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300 DYNAMIC OR MAY be selected from ISO Country Alpha-3 2.16.840.1.113883.1.11.171 DYNAMIC, IF the country is not specified in ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300. |
|
IPS CDA documentationOf
IPS CDA legalAuthenticator
IPS CDA Organization
IPS CDA Person
| Id | 2.16.840.1.113883.10.22.9.3 | Effective Date | 2017‑04‑12 |
|---|
| Status |  Under pre-publication review Under pre-publication review | Version Label | STU1 |
|---|
| Name | IPSCDAPerson | Display Name | IPS CDA Person |
|---|
| Description | Person name |
|---|
| Classification | CDA Header Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Relationship | Adaptation: template 2.16.840.1.113883.10.12.152 CDA Person (2005‑09‑07) ref ad1bbr- |
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| cs | 0 … 1 | F | PSN | | cs | 0 … 1 | F | INSTANCE | | PN | 1 … * | R | | (IPS...son) |
|
IPS CDA recordTarget
| Id | 2.16.840.1.113883.10.22.2.1 | Effective Date | 2021‑09‑02 12:10:24Other versions this id:  IPSCDArecordTarget as of 2020‑07‑14 16:56:08 IPSCDArecordTarget as of 2020‑07‑14 16:56:08 IPSCDArecordTarget as of 2017‑04‑11 IPSCDArecordTarget as of 2017‑04‑11
|
|---|
| Status |  Draft Draft | Version Label | 2021 |
|---|
| Name | IPSCDArecordTarget | Display Name | IPS CDA recordTarget |
|---|
| Description | The recordTarget records the administrative and demographic data of the patient whose health information is described by the clinical document; each recordTarget must contain at least one patientRole element. |
|
| Classification | CDA Header Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Associated with | | Associated with 9 concepts | | Id | Name | Data Set |
|---|
| hl7ips-dataelement-100 |  Telecoms Telecoms |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-135 |  Patient’s preferred language Patient’s preferred language |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-162 |  Address Address |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-2.1 |  Patient Attributes Patient Attributes |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-202 |  Healthcare related Identifiers Healthcare related Identifiers |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-3 |  Patient's name Patient's name |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-4 |  Administrative gender Administrative gender |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-5 |  Date of birth Date of birth |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-7 |  Insurance identifier Insurance identifier |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|
|
|---|
| Uses | | Uses 1 template | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.11 | Include |  IPS Address (STU1) IPS Address (STU1) | DYNAMIC |
|
|
|---|
| Relationship | Version: template 2.16.840.1.113883.10.22.2.1 IPS CDA recordTarget (2020‑07‑14 16:56:08)
Adaptation: template 2.16.840.1.113883.3.1937.777.11.10.100 epSOS CDA recordTarget (2013‑12‑20) ref epsos-
Adaptation: template 2.16.840.1.113883.10.12.101 CDA recordTarget (2005‑09‑07) ref ad1bbr- |
|---|
| Example | | Example | <recordTarget typeCode="RCT" contextControlCode="OP">
<patientRole classCode="PAT">
<id root="1.2.3.999" extension="__example only__"/> <addr>
<streetAddressLine>HSE M CASSAR STR</streetAddressLine> <city>ISLA</city> <country>MT</country> </addr> <telecom use="HP" value="tel:+356124567891"/> <telecom use="WP" value="mailto:elif@foo.too.mt"/> <patient>
<name>
<family>BORG</family> <given>TANIA</given> </name> <administrativeGenderCode code="F" codeSystem="2.16.840.1.113883.5.1" displayName="Female"/> <birthTime value="19430130"/> <!-- Optional guardian information ; see example below-->
<!-- Optional languageCommunication information see example below -->
</patient> </patientRole></recordTarget> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | 1 … * | R | | (IPS...get) |  | @typeCode
|
| cs | 0 … 1 | F | RCT |  | @contextControlCode
|
| cs | 0 … 1 | F | OP | | | Example | <recordTarget typeCode="RCT" contextControlCode="OP">
<patientRole classCode="PAT">
<id root="1.2.3.999" extension="__example only__"/> <addr>
<streetAddressLine>HSE M CASSAR STR</streetAddressLine> <city>ISLA</city> <country>MT</country> </addr> <telecom use="HP" value="tel:+356124567891"/> <telecom use="WP" value="mailto:elif@foo.too.mt"/> <patient>
<name>
<family>BORG</family> <given>TANIA</given> </name> <administrativeGenderCode code="F" codeSystem="2.16.840.1.113883.5.1" displayName="Female"/> <birthTime value="19430130"/> <!-- Optional guardian information ; see example below-->
<!-- Optional languageCommunication information see example below -->
</patient> </patientRole></recordTarget> |  | hl7:patientRole
|
| | 1 … 1 | M | | (IPS...get) | | |  | | hl7ips-dataelement-2.1 |  Patient Attributes Patient Attributes |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | @classCode
|
| cs | 0 … 1 | F | PAT |  |  | hl7:id
|
| II | 1 … * | R | Patient Identifiers: Primary Patient Identifier (Regional/National Health Id), Secondary Patient Identifier (Social/Insurance Number) | (IPS...get) | | |  | | hl7ips-dataelement-202 |  Healthcare related Identifiers Healthcare related Identifiers |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-7 |  Insurance identifier Insurance identifier |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | hl7:addr
|
| AD.IPS | 1 … * | R | The patient address. | (IPS...get) | | |  | | hl7ips-dataelement-162 |  Address Address |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | Constraint | When used for cross-border exchange the country address part has to be provided. | | Included | | | from 2.16.840.1.113883.10.22.11 IPS Address (DYNAMIC) | | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.10637 PostalAddressUse (2005‑05‑01) |
| | cs | 0 … 1 | F | NI | | | Constraint | SHALL NOT have mixed content except for white space
If there is no information, the nullFlavor attribute shall have a value of 'NI' and no address parts shall be present, otherwise there shall be no nullFlavor attribute, and at least one of the address parts listed below shall be present. | | | Schematron assert | role | error | | | | test | @nullFlavor or hl7:* | | | | Message | If addr is not nullflavored at least one sub element has to be provided | | | ADXP | 0 … * | C | Subject's or Organization's Street Address Line | (IPS...get) | | | Schematron assert | role | error | | | | test | hl7:streetAddressLine and (hl7:city or hl7:postalCode) | | | | Message | If the address line is included either the city or the zip code has to be provided | | | ADXP | 0 … 1 | C | Subject's or Organization's City | (IPS...get) | | ADXP | 0 … 1 | C | Subject's or Organization's Postal Code | (IPS...get) | | ADXP | 0 … 1 | C | Subject's or Organization's State or Province | (IPS...get) | | ADXP | 0 … 1 | C | Subject's Country. | (IPS...get) | | | Constraint | The content of this element SHALL be selected EITHER from ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300 DYNAMIC OR MAY be selected from ISO Country Alpha-3 2.16.840.1.113883.1.11.171 DYNAMIC, IF the country is not specified in ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300. |  |  | hl7:telecom
|
| TEL | 1 … * | R | Patient’s telecom information : e.g. telephone number, e-mail address. | (IPS...get) | | |  | | hl7ips-dataelement-100 |  Telecoms Telecoms |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.201 TelecommunicationAddressUse (DYNAMIC) |
| | cs | 0 … 1 | F | NI | | | Constraint | If there is no information, the nullFlavor attribute shall have a value of 'NI' and the "value" and "use" attributes shall be omitted, otherwise the nullFlavor attribute shall not be present, and the "value" and "use" attributes shall be present.
| | | Example | <telecom use="HP" value="tel:+356124567891"/> | | | Example | <telecom use="WP" value="mailto:elif@foo.too.mt"/> | | | Example | <telecom nullFlavor="NI"/> |  |  | hl7:patient
|
| | 1 … 1 | M | | (IPS...get) | | cs | 0 … 1 | F | PSN | | cs | 0 … 1 | F | INSTANCE | | | Example | Japanese example (Person Name) <patient>
<name use="IDE">
<family>木村</family> <given>通男</given> </name> <name use="SYL">
<family>きむら</family> <given>みちお</given> </name> <name use="ABC">
<family>KIMURA</family> <given>MICHIO</given> </name> <administrativeGenderCode code="M" codeSystem="2.16.840.1.113883.5.1" displayName="Male"/> <birthTime nullFlavor="UNK"/></patient> | | PN | 1 … * | M | Patient Name | (IPS...get) | | |  | | hl7ips-dataelement-3 |  Patient's name Patient's name |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | Constraint | The Alphabetic representation of the name SHALL be always provided | | | 1 … * | R | Patient's Family Name/Surname | (IPS...get) | | | 1 … * | R | Patient's Given Name | (IPS...get) |  |  |  | hl7:administrativeGenderCode
|
| CE.IPS | 1 … 1 | R | Patient's Gender | (IPS...get) | | |  | | hl7ips-dataelement-4 |  Administrative gender Administrative gender |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | cs | 0 … 1 | F | UNK | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.1.11.1 Administrative Gender (HL7 V3) (DYNAMIC) |
| | | Example | <administrativeGenderCode code="F" codeSystem="2.16.840.1.113883.5.1" displayName="Female">
<translation code="2" codeSystem="2.16.840.1.113883.3.129.1.2.21" codeSystemName="Cinsiyet" displayName="Kadın"/></administrativeGenderCode> | | TS | 1 … 1 | R | Patient's Date of Birth. The patient date of birth may be a partial date such as only the year. | (IPS...get) | | |  | | hl7ips-dataelement-5 |  Date of birth Date of birth |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | 0 … * | R |
The guardians of a patient.
Other patient contacts are described using the /ClinicalDocument/participant structure. The <associatedEntity> element defines the type of contact.
| (IPS...get) | | cs | 1 … 1 | F | GUARD | | | Example | <guardian classCode="GUARD">
<code code="AUNT" displayName="tante" codeSystem="2.16.840.1.113883.5.111"/> <addr nullFlavor="NI"/> <telecom use="MC" value="tel:+33-12345678"/> <guardianPerson>
<name>
<family>Curie</family> <given>Marie</given> </name> </guardianPerson></guardian> | | CD.IPS | 0 … 1 | R | The relationship between the patient and the guardian or other contact may be recorded in the element. | (IPS...get) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.1.11.19563 PersonalRelationshipRoleType (DYNAMIC) |
| | AD.IPS | 1 … * | R | | (IPS...get) | | | Constraint | If there is no information, the nullFlavor attribute shall have a value of 'NI' and no address parts shall be present, otherwise there shall be no nullFlavor attribute, and at least one of the address parts listed below shall be present.
| | Included | | | from 2.16.840.1.113883.10.22.11 IPS Address (DYNAMIC) | | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.10637 PostalAddressUse (2005‑05‑01) |
| | cs | 0 … 1 | F | NI | | | Constraint | SHALL NOT have mixed content except for white space
If there is no information, the nullFlavor attribute shall have a value of 'NI' and no address parts shall be present, otherwise there shall be no nullFlavor attribute, and at least one of the address parts listed below shall be present. | | | Schematron assert | role | error | | | | test | @nullFlavor or hl7:* | | | | Message | If addr is not nullflavored at least one sub element has to be provided | | | ADXP | 0 … * | C | Subject's or Organization's Street Address Line | (IPS...get) | | | Schematron assert | role | error | | | | test | hl7:streetAddressLine and (hl7:city or hl7:postalCode) | | | | Message | If the address line is included either the city or the zip code has to be provided | | | ADXP | 0 … 1 | C | Subject's or Organization's City | (IPS...get) | | ADXP | 0 … 1 | C | Subject's or Organization's Postal Code | (IPS...get) | | ADXP | 0 … 1 | C | Subject's or Organization's State or Province | (IPS...get) | | ADXP | 0 … 1 | C | Subject's Country. | (IPS...get) | | | Constraint | The content of this element SHALL be selected EITHER from ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300 DYNAMIC OR MAY be selected from ISO Country Alpha-3 2.16.840.1.113883.1.11.171 DYNAMIC, IF the country is not specified in ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300. | | TEL | 1 … * | R | Guardian’s telecom information: e.g. telephone number; e-mail address. | (IPS...get) | | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.201 TelecommunicationAddressUse (DYNAMIC) |
| | cs | 0 … 1 | F | NI | | | Constraint | If there is no information, the nullFlavor attribute shall have a value of 'NI' and the "value" and "use" attributes shall be omitted, otherwise the nullFlavor attribute shall not be present, and the "value" and "use" attributes shall be present.
| | | 1 … 1 | R | | (IPS...get) | | PN | 1 … * | R | Patient Guardian's Name | (IPS...get) | | ENXP | 1 … * | R | Patient Guardian's Family Name/Surname | (IPS...get) | | ENXP | 1 … * | R | Patient Guardian's Given Name | (IPS...get) |  |  |  | hl7:languageCommunication
|
| | 0 … * | R | | (IPS...get) | | CS | 1 … 1 | R | Patient’s language | (IPS...get) | | |  | | hl7ips-dataelement-135 |  Patient’s preferred language Patient’s preferred language |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | Constraint | The two characters form SHALL be used when available; otherwise the three characters representation SHALL be adopted | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.4.642.3.21 All Languages (DYNAMIC) |
| | | Example | British English <languageCode code="en-GB"/> | | | Example | Amurdak (Australia) <languageCode code="amg-AU"/> | | | Schematron assert | role | error | | | | test | matches(@code,'[a-z]{2,3}-[A-Z]{2,3}') | | | | Message | The language code SHALL be in the form nn-CC or nnn-CCC, in accordance with BCP 47 (e.g. nn is the ISO language code; CC is ISO country code) | |
|
IPS CDA relatedDocument
IPS Patient Contacts
| Id | 2.16.840.1.113883.10.22.2.5 | Effective Date | 2017‑04‑12 |
|---|
| Status |  Under pre-publication review Under pre-publication review | Version Label | STU1 |
|---|
| Name | IPSCDAContacts | Display Name | IPS Patient Contacts |
|---|
| Description | The IPS may record several kinds of patient contacts, including parents, relatives, caregivers, and others related in some way to the patient. A patient contact may be an individual or an organization with a relationship to the patient, including health provider (person or organization) to be contacted in case of emergency.
|
|
| Classification | CDA Header Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Associated with | | Associated with 8 concepts | | Id | Name | Data Set |
|---|
| hl7ips-dataelement-121 |  Name Name |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-154 |  Patient’s Address Book Patient’s Address Book |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-163 |  Preferred Healthcare providers Preferred Healthcare providers |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-165 |  Healthcare Provider (person) Healthcare Provider (person) |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-166 |  Healthcare Provider (organisation) Healthcare Provider (organisation) |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-169 |  Telecoms Telecoms |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-172 |  Organisation’s Name Organisation’s Name |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-174 |  Telecoms Telecoms |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|
|
|---|
| Uses | | Uses 1 template | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.11 | Include |  IPS Address (STU1) IPS Address (STU1) | DYNAMIC |
|
|
|---|
| Relationship | Adaptation: template 2.16.840.1.113883.10.12.108 CDA participant (DYNAMIC) ref ad1bbr-
Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.2.4 IHE Patient Contacts (DYNAMIC) ref IHE-PCC- |
|---|
| Example | | Contact person | <participant typeCode="IND">
<templateId root="2.16.840.1.113883.10.22.2.5"/> <associatedEntity classCode="NOK">
<addr>
<streetAddressLine>Promenade des Anglais 111</streetAddressLine> <city>Lyon</city> <postalCode>69001</postalCode> <country>FR</country> </addr> <telecom value="tel:(+33)555-20036" use="WP"/> <associatedPerson>
<name>
<given>Martha</given> <family>Mum</family> </name> </associatedPerson> </associatedEntity></participant> |
|
|---|
| Example | | Preferred Health Professional for emergency contact | <participant typeCode="IND">
<templateId root="2.16.840.1.113883.10.22.2.5"/> <functionCode code="PCP" codeSystem="2.16.840.1.113883.5.88"/> <time value="20070213"/> <associatedEntity classCode="ECON">
<addr>
<streetAddressLine>Karl Strasse</streetAddressLine> <city>Freiberg</city> <postalCode>09599</postalCode> <country>DE</country> </addr> <telecom value="tel:(+49)761-11110000" use="WP"/> <associatedPerson>
<name>
<given>Arzt</given> <family>Guter</family> </name> </associatedPerson> </associatedEntity></participant> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | | R | Patient contacts or the Preferred Health Professional to contact in case of emergency. | (IPS...cts) | | where [hl7:templateId/@root='2.16.840.1.113883.10.22.2.5'] | | | |  | | hl7ips-dataelement-154 |  Patient’s Address Book Patient’s Address Book |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-163 |  Preferred Healthcare providers Preferred Healthcare providers |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  | @typeCode
|
| cs | 1 … 1 | F | IND | | | Example | <participant typeCode="IND">
<templateId root="2.16.840.1.113883.10.22.2.5"/> <associatedEntity classCode="NOK">
<addr>
<streetAddressLine>Promenade des Anglais 111</streetAddressLine> <city>Lyon</city> <postalCode>69001</postalCode> <country>FR</country> </addr> <telecom value="tel:(+33)555-20036" use="WP"/> <associatedPerson>
<name>
<given>Martha</given> <family>Mum</family> </name> </associatedPerson> </associatedEntity></participant> |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...cts) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.2.5 |  | hl7:functionCode
|
| | 0 … 1 | C | The <functionCode> element may be used to indicate that this participant is the preferred Health Professional to contact in case of emergency.</functionCode> | (IPS...cts) |  |  | @code
|
| CONF | 0 … 1 | F | PCP |  |  | @codeSystem
|
| 0 … 1 | F | 2.16.840.1.113883.5.88 (Participation Function) |  | hl7:associatedEntity
|
| | | R | The <associatedEntity> element identifies the type of contact. </associatedEntity> | (IPS...cts) |  |  | @classCode
|
| cs | 1 … 1 | R | | | | CONF | | The value of @classCode shall be drawn from value set 2.16.840.1.113883.11.20.9.33 INDRoleclassCodes (DYNAMIC) |
| | | Example | <associatedEntity classCode="ECON">
<addr>
<streetAddressLine>Karl Strasse</streetAddressLine> <city>Freiberg</city> <postalCode>09599</postalCode> <country>DE</country> </addr> <telecom value="tel:+49-761-11110000" use="WP"/> <associatedPerson>
<name>
<given>Arzt</given> <family>Guter</family> </name> </associatedPerson></associatedEntity> |  |  | hl7:code
|
| CV.IPS | 0 … 1 | R | This element indicates the relationship between the patient and this participant. | (IPS...cts) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.54 IPS Personal Relationship (DYNAMIC) | | or | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.53 IPS Healthcare Professional Roles (DYNAMIC) |
| | | Example | <code code="AUNT" displayName="θεία" codeSystem="2.16.840.1.113883.5.111"/> |  |  | hl7:addr
|
| AD.IPS | 1 … * | R | Patient Contact's / Preferred HP's Address | (IPS...cts) | | | Schematron assert | role | error | | | | test | @nullFlavor or hl7:* | | | | Message | If addr is not nullflavored at least one sub element has to be provided | | | Included | | | from 2.16.840.1.113883.10.22.11 IPS Address (DYNAMIC) | | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.10637 PostalAddressUse (2005‑05‑01) |
| | cs | 0 … 1 | F | NI | | | Constraint | SHALL NOT have mixed content except for white space
If there is no information, the nullFlavor attribute shall have a value of 'NI' and no address parts shall be present, otherwise there shall be no nullFlavor attribute, and at least one of the address parts listed below shall be present. | | | Schematron assert | role | error | | | | test | @nullFlavor or hl7:* | | | | Message | If addr is not nullflavored at least one sub element has to be provided | | | ADXP | 0 … * | C | Subject's or Organization's Street Address Line | (IPS...cts) | | | Schematron assert | role | error | | | | test | hl7:streetAddressLine and (hl7:city or hl7:postalCode) | | | | Message | If the address line is included either the city or the zip code has to be provided | | | ADXP | 0 … 1 | C | Subject's or Organization's City | (IPS...cts) | | ADXP | 0 … 1 | C | Subject's or Organization's Postal Code | (IPS...cts) | | ADXP | 0 … 1 | C | Subject's or Organization's State or Province | (IPS...cts) | | ADXP | 0 … 1 | C | Subject's Country. | (IPS...cts) | | | Constraint | The content of this element SHALL be selected EITHER from ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300 DYNAMIC OR MAY be selected from ISO Country Alpha-3 2.16.840.1.113883.1.11.171 DYNAMIC, IF the country is not specified in ValueSet ISO Country Alpha-2 urn:oid:2.16.840.1.113883.1.11.20300. |  |  | hl7:telecom
|
| TEL | 1 … * | R | Patient Contact's / Preferred HP's/Legal Organization telephone or e-mail <telecom> element is required.</telecom> | (IPS...cts) | | |  | | hl7ips-dataelement-169 |  Telecoms Telecoms |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-174 |  Telecoms Telecoms |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | set_cs | 0 … 1 | | | | | CONF | | The value of @use shall be drawn from value set 2.16.840.1.113883.1.11.201 TelecommunicationAddressUse (DYNAMIC) |
| | cs | 0 … 1 | F | NI | | | Constraint | If there is no information, the nullFlavor attribute shall have a value of 'NI' and the "value" and "use" attributes shall be omitted, otherwise the nullFlavor attribute shall not be present, and the "value" and "use" attributes shall be present
| | | Example | <telecom use="WP" value="tel:+45 20 7025 6161"/><telecom use="HP" value="mailto:jsmith@myprovider.co.uk"/> | | Choice | 1 … 2 | | Elements to choose from:- hl7:associatedPerson
- hl7:scopingOrganization
| | | 0 … 1 | C | Or the associatedPerson, or the scopingOrganization, or both elements shall be provided | (IPS...cts) | | |  | | hl7ips-dataelement-165 |  Healthcare Provider (person) Healthcare Provider (person) |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | PN | 1 … * | R | Patient Contact's Name / Preferred HP's Name | (IPS...cts) | | |  | | hl7ips-dataelement-121 |  Name Name |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | Example | <name>
<given>John</given> <family>Español Smith</family></name> | | | 1 … * | R | Patient Contact's Family Name/Surname / Preferred HP's Family Name/Surname | (IPS...cts) | | | 1 … * | R | Patient Contact's Given Name / Preferred HP's Given Name | (IPS...cts) | | | 0 … 1 | C | Or the associatedPerson, or the scopingOrganization, or both elements shall be provided | (IPS...cts) | | |  | | hl7ips-dataelement-166 |  Healthcare Provider (organisation) Healthcare Provider (organisation) |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | ON | 1 … * | R | Organization's Name | (IPS...cts) | | |  | | hl7ips-dataelement-172 |  Organisation’s Name Organisation’s Name |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|
|
CDA Section Level Templates
IHE Coded Vital Signs Section
1.3.6.1.4.1.19376.1.5.3.1.1.5.3.2 (not listed here)
IPS Advance Directives Section
IPS Alerts Section
| Id | 2.16.840.1.113883.10.22.15 | Effective Date | 2024‑06‑12 08:51:01 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSAlertsSection | Display Name | IPS Alerts Section |
|---|
| Description |
The ‘Alert’ Section brings the most important healthcare information to the fore.
This section highlights extreme problem(s). Previously, ‘alerts’ were just reported as ‘problems’, which carried the risk of losing the immediacy and importance of information to the attending clinician.
An alert in meant to be "Information used to warn or call to a state of preparedness. Information flagged (or intended) to raise awareness of a potential danger to/from the subject of the IPS or to/from another individual or awareness of a potential obstacle to care."
|
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.15 |
|---|
| Classification | CDA Section Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Uses | | Uses 2 templates | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.4.31 | Containment |  IPS Internal Reference (STU1) IPS Internal Reference (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.3.15 | Containment |  IPS Translation Section (2021) IPS Translation Section (2021) | DYNAMIC |
|
|
|---|
| Relationship | Adaptation: template 2.16.840.1.113883.10.12.201 CDA Section (2005‑09‑07) ref ad1bbr- |
|---|
|
IPS Allergies and Intolerances Section
IPS Functional Status Section
IPS History of Past Illness Section
IPS History of Pregnancy Section
IPS History of Procedures Section
IPS Immunizations Section
IPS Medical Devices Section
IPS Medication Summary Section
IPS Patient Story Section
| Id | 2.16.840.1.113883.10.22.16 | Effective Date | 2024‑06‑12 09:26:02 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSPatientStorySection | Display Name | IPS Patient Story Section |
|---|
| Description |
A concise narrative from the patient’s perspective about their present health state. This is a record of the things that a person feels it is important to communicate about their needs, strengths, values, concerns and preferences to others providing support and care.
Patients unable to communicate can be supported by a proxy author. The patient’s story, provided here, may be told by the patient or by a proxy.
|
|
| Classification | CDA Section Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Uses | | Uses 3 templates | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.4.14 | Include |  IPS Body Author (STU1) IPS Body Author (STU1) | DYNAMIC | | 2.16.840.1.113883.10.12.319 | Containment |  CDA Informant (Body) CDA Informant (Body) | DYNAMIC | | 2.16.840.1.113883.10.22.3.15 | Containment |  IPS Translation Section (2021) IPS Translation Section (2021) | DYNAMIC |
|
|
|---|
|
IPS Plan of Care Section
IPS Problems Section
IPS Results Section
| Id | 2.16.840.1.113883.10.22.3.14 | Effective Date | 2017‑04‑30 |
|---|
| Status |  Under pre-publication review Under pre-publication review | Version Label | STU1 |
|---|
| Name | IPSResultsSection | Display Name | IPS Results Section |
|---|
| Description | This section assembles relevant observation results collected on the patient or produced on in-vitro biologic specimens collected from the patient. Some of these results may be laboratory results, others may be anatomic pathology results, others, radiology results, and others, clinical results. The structured, machine-processable content of this section is sorted out between as many Result Organizer entries as needed. One Result Organizer entry groups results, which have a common context of production: - common specialty (imaging, bacteriology, serology, chemistry, surgical pathology, clinical, radiology ...),
- common overall interpretation, (which interprets the set of results of the Organizer),
- common
biologic specimen for in vitro diagnostic observations,
- common associated illustrative image.
The optional author and informant elements of the section are used when necessary to convey the provenance and authoring of the section content in case it is different from what is announced in the CDA header.
In case this section assembles results from multiple authors (e.g.; results authored by a clinical laboratory, and results produced by a radiology center), the authors are listed in the section, and each Result Organizer of the section indicates its own author(s). |
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.3.14 |
|---|
| Classification | CDA Section Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Associated with | | Associated with 3 concepts | | Id | Name | Data Set |
|---|
| hl7ips-dataelement-126 |  Observations results list Observations results list |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-142 |  Result Description Result Description |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-19 |  Results Results |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|
|
|---|
| Uses | | Uses 4 templates | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.4.14 | Include |  IPS Body Author (STU1) IPS Body Author (STU1) | DYNAMIC | | 2.16.840.1.113883.10.12.319 | Containment |  CDA Informant (Body) CDA Informant (Body) | DYNAMIC | | 2.16.840.1.113883.10.22.4.9 | Containment |  IPS Result Organizer (STU1) IPS Result Organizer (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.3.15 | Containment |  IPS Translation Section (2021) IPS Translation Section (2021) | DYNAMIC |
|
|
|---|
| Relationship | Adaptation: template 2.16.840.1.113883.10.12.201 CDA Section (2005‑09‑07) ref ad1bbr- |
|---|
| Example | | Example | <section classCode="DOCSECT" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.3.14"/> <id root="1.2.3.999" extension="__example only__"/> <code code="30954-2" codeSystem="2.16.840.1.113883.6.1" displayName="Relevant diagnostic tests/laboratory data Narrative"/> <title>MOST SIGNIFICANT RESULTS</title> <text>
<!-- some clinical laboratory results and some surgical pathology results presented to the human reader -->
</text> <author>
<!-- template 2.16.840.1.113883.10.12.318 'CDA Author (Body)' - 1st author: a clinical lab director -->
</author> <author>
<!-- template 2.16.840.1.113883.10.12.318 'CDA Author (Body)' - 2nd author: a pathologist -->
</author> <entry typeCode="COMP" contextConductionInd="true">
<!--- 1st Organizer: chemistry observations on blood serum specimen produced and interpreted by a clinical laboratory -->
<organizer classCode="BATTERY" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.9"/> <code code="18719-5" displayName="Chemistry studies (set)" codeSystemName="LOINC" codeSystem="2.16.840.1.113883.6.1"/> <satusCode code="completed"/> <author>
<!-- template 2.16.840.1.113883.10.12.318 'CDA Author (Body)' - 1st author: a clinical lab director -->
</author> <component>
<!-- template 2.16.840.1.113883.10.22.4.13 'IPS Laboratory Result Observation' -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.13 'IPS Laboratory Result Observation' -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.30 'IPS Specimen Collection' - common blood serum specimen -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.22 'IPS Comment Activity' - interpretation of chemistry results -->
</component> </organizer> </entry> <entry>
<organizer classCode="BATTERY" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.9"/> <code code="18723-7" displayName="Hematology studies (set)" codeSystemName="LOINC" codeSystem="2.16.840.1.113883.6.1"/> <satusCode code="completed"/> <author>
<!-- template 2.16.840.1.113883.10.12.318 'CDA Author (Body)' - 1st author: a clinical lab director -->
</author> <component>
<!-- template 2.16.840.1.113883.10.22.4.13 'IPS Laboratory Result Observation' -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.13 'IPS Laboratory Result Observation' -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.30 'IPS Specimen Collection' - venous blood total specimen -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.22 'IPS Comment Activity' - interpretation of hematology results -->
</component> </organizer> </entry> <entry>
<organizer classCode="BATTERY" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.9"/> <code code="11529-5" displayName="Surgical pathology studies (set)" codeSystemName="LOINC" codeSystem="2.16.840.1.113883.6.1"/> <satusCode code="completed"/> <author>
<!-- template 2.16.840.1.113883.10.12.318 'CDA Author (Body)' - 2nd author: a pathologist -->
</author> <component>
<!-- template 2.16.840.1.113883.10.22.4.11 'IPS Pathology Result Observation' -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.30 'IPS Specimen Collection' - excised tissue specimen -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.22 'IPS Comment Activity' - pathologist's interpretation -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.23 'IPS ObservationMedia' - an illustrative slide image -->
</component> </organizer> </entry></section> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | | | | (IPS...ion) | | |  | | hl7ips-dataelement-19 |  Results Results |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  | @classCode
|
| cs | 0 … 1 | F | DOCSECT |  | @moodCode
|
| cs | 0 … 1 | F | EVN |  | hl7:templateId
|
| II | 1 … 1 | R | | (IPS...ion) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.3.14 |  | hl7:id
|
| II | 0 … * | R | | (IPS...ion) |  | hl7:code
|
| CE.IPS | 1 … 1 | M | | (IPS...ion) |  |  | @code
|
| CONF | 1 … 1 | F | 30954-2 |  |  | @codeSystem
|
| 1 … 1 | F | 2.16.840.1.113883.6.1 (LOINC) |  | hl7:title
|
| ST | 1 … 1 | M | | (IPS...ion) |  | hl7:text
|
| SD.TEXT | 1 … 1 | M | | (IPS...ion) | | |  | | hl7ips-dataelement-142 |  Result Description Result Description |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | Included | 0 … * | | from 2.16.840.1.113883.10.22.4.14 IPS Body Author (DYNAMIC) |  | hl7:author
|
| | 0 … * | | | (IPS...ion) |  |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...ion) | | uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.14 |  |  | hl7:time
|
| TS.IPS.TZ | 1 … 1 | R | | (IPS...ion) |  |  | hl7:assignedAuthor
|
| | 1 … 1 | M | | (IPS...ion) | | II | 1 … * | R | | (IPS...ion) | | | 0 … 1 | R | | (IPS...ion) | | Choice | 0 … 1 | | Elements to choose from:- hl7:assignedPerson
- hl7:assignedAuthoringDevice
| | | 0 … 1 | C | | (IPS...ion) | | cs | 0 … 1 | F | PSN | | cs | 0 … 1 | F | INSTANCE | | PN | 1 … * | R | Name of the person (e.g. the Healthcare Professional) authoring this document | (IPS...ion) | | | Example | <name>
<given>John</given> <family>Español Smith</family></name> | | | 1 … * | R | | (IPS...ion) | | | 1 … * | R | | (IPS...ion) |  |  |  |  | hl7:assignedAuthoringDevice
|
| | 0 … 1 | C | | (IPS...ion) | | | Example | <assignedAuthoringDevice classCode="DEV" determinerCode="INSTANCE">
<softwareName displayName="Turriano"/></assignedAuthoringDevice> | | Included | | | from 2.16.840.1.113883.10.22.9.2 IPS CDA Device (DYNAMIC) | | cs | 0 … 1 | F | DEV | | cs | 0 … 1 | F | INSTANCE | | CE | 0 … 1 | | | (IPS...ion) |  |  |  |  |  | hl7:manufacturerModelName
|
| SC | 0 … 1 | | | (IPS...ion) | | SC | 0 … 1 | | | (IPS...ion) |  |  |  | hl7:representedOrganization
|
| | 0 … 1 | | | (IPS...ion) | | II | 0 … * | | | (IPS...ion) | | | 0 … * | | | (IPS...ion) | | TEL | 0 … * | | | (IPS...ion) | | AD | 0 … * | | | (IPS...ion) |  | hl7:informant
|
| | 0 … * | | Contains 2.16.840.1.113883.10.12.319 CDA Informant (Body) (DYNAMIC) | (IPS...ion) |  | hl7:entry
|
| | 1 … * | M | Contains 2.16.840.1.113883.10.22.4.9 IPS Result Organizer (DYNAMIC) | (IPS...ion) | | |  | | hl7ips-dataelement-126 |  Observations results list Observations results list |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | @typeCode
|
| cs | 1 … 1 | R | | | | CONF | | The value of @typeCode shall be drawn from value set 2.16.840.1.113883.1.11.19446 x_ActRelationshipEntry (DYNAMIC) |
|  |  | @contextConductionInd
|
| bl | 0 … 1 | F | true |  | hl7:component
|
| | 0 … * | | Contains 2.16.840.1.113883.10.22.3.15 IPS Translation Section (DYNAMIC) | (IPS...ion) |
|
IPS Social History Section
IPS Translation Section
CDA Entry Level Templates
CDA Author (Body)
CDA Device
| Id | 2.16.840.1.113883.10.12.315 ref ad1bbr- | Effective Date | 2005‑09‑07 |
|---|
| Status |  Active Active | Version Label | |
|---|
| Name | CDADevice | Display Name | CDA Device |
|---|
| Description | Template CDA Device (prototype, directly derived from POCD_RM000040 MIF) |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | 0 … 1 | F | DEV | | | 0 … 1 | F | INSTANCE | | CE | 0 … 1 | | | (CDA...ice) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.1.11.16040 EntityCode (DYNAMIC) |
| hl7:manufacturerModelName
|
| SC | 0 … 1 | | | (CDA...ice) | | SC | 0 … 1 | | | (CDA...ice) |
|
CDA Informant (Body)
CDA Performer (Body)
CDA RelatedEntity
| Id | 2.16.840.1.113883.10.12.316 ref ad1bbr- | Effective Date | 2005‑09‑07 |
|---|
| Status |  Active Active | Version Label | |
|---|
| Name | CDARelatedEntity | Display Name | CDA RelatedEntity |
|---|
| Description | Template CDA RelatedEntity (prototype, directly derived from POCD_RM000040 MIF) |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Uses | | Uses 1 template | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.12.152 | Containment |  CDA Person CDA Person | DYNAMIC |
|
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| cs | 1 … 1 | R | | | | CONF | | The value of @classCode shall be drawn from value set 2.16.840.1.113883.1.11.19316 RoleClassMutualRelationship (DYNAMIC) |
| | CE | 0 … 1 | | | (CDA...ity) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.1.11.19563 PersonalRelationshipRoleType (DYNAMIC) |
| | AD | 0 … * | | | (CDA...ity) | | TEL | 0 … * | | | (CDA...ity) | | IVL_TS | 0 … 1 | | | (CDA...ity) | | | 0 … 1 | | Contains 2.16.840.1.113883.10.12.152 CDA Person (DYNAMIC) | (CDA...ity) |
|
IHE Vital Signs Observation
1.3.6.1.4.1.19376.1.5.3.1.4.13.2/dynamic
IPS Advance Directive Observation
IPS Advance Directive Organizer
IPS Allergy and Intolerance Concern
| Id | 2.16.840.1.113883.10.22.4.5 | Effective Date | 2024‑08‑04 10:09:24Other versions this id:  IPSAllergyAndIntoleranceConcern as of 2016‑11‑11 IPSAllergyAndIntoleranceConcern as of 2016‑11‑11
|
|---|
| Status |  Draft Draft | Version Label | STU2 |
|---|
| Name | IPSAllergyAndIntoleranceConcern | Display Name | IPS Allergy and Intolerance Concern |
|---|
| Description | This template reflects an ongoing concern on behalf of the person that placed the allergy on a patient’s allergy list. A concern may refer to one or more allergies or intolerances. There are different kinds of status that could be related to an allergy, or more in general to a condition:
- The status of the concern (active, inactive,..)
- The status of the condition (e.g. active, inactive, resolved,..)
- The confirmation status [clinical workflow status, certainty] (e.g. confirmed, likely, unlikely,…)
Not all of them can be represented in a CDA using the statusCode elements of the concern (ACT) and observation (condition).
As long as the underlying condition is of concern to the author (i.e., as long as the allergy, whether active or resolved, is of ongoing concern and interest to the author), the statusCode is “active”.
In case the clinician deems that there is no longer any need to track the underlying conditions then the concern is inactive and the statusCode is set to "completed".
The effectiveTime/low of the Allergy Concern Act asserts when the concern became active. This equates to the time the concern was authored in the patient's chart.
The effectiveTime/high asserts when the concern became inactive, and it is present if the statusCode of the concern act is "completed"
|
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.5 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Uses | | Uses 1 template | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.4.1 | Containment |  IPS Allergy or Intolerance (STU2) IPS Allergy or Intolerance (STU2) | DYNAMIC |
|
|
|---|
| Relationship | Specialization: template 2.16.840.1.113883.10.22.4.5 IPS Allergy and Intolerance Concern (2016‑11‑11)
Adaptation: template 2.16.840.1.113883.10.12.301 CDA Act (2005‑09‑07) ref ad1bbr-
Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.4.5.3 IHE Allergy and Intolerance Concern Entry (2013‑12‑20) ref IHE-PCC- |
|---|
| Example | | Example | <act classCode="ACT" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.5"/> <id root="1.2.3.999" extension="__example only__"/> <code code="CONC" codeSystem="2.16.840.1.113883.5.6">
<statusCode code="active"/> <effectiveTime>
<low value="..."/> <high value="..."/> </effectiveTime> <entryRelationship typeCode="SUBJ" inversionInd="false">
<!-- template 2.16.840.1.113883.10.22.4.1 'IPS Allergy or Intolerance' (dynamic) -->
</entryRelationship> </code></act> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | 0 … * | R | | (IPS...ern) |  | @classCode
|
| cs | 1 … 1 | F | ACT |  | @moodCode
|
| cs | 1 … 1 | F | EVN |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...ern) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.5 |  | hl7:id
|
| II | 0 … * | R | | (IPS...ern) |  | hl7:code
|
| CD | 1 … 1 | M | | (IPS...ern) |  |  | @code
|
| CONF | 1 … 1 | F | CONC |  |  | @codeSystem
|
| 1 … 1 | F | 2.16.840.1.113883.5.6 (HL7ActClass) |  | hl7:statusCode
|
| CS | 1 … 1 | R | As long as the underlying conditions are of concern to the author (i.e., as long as allergies, whether active or resolved, is of ongoing concern and interest to the author), the statusCode is “ active ”. The concern is tracked by the author.
Only when the underlying allergies are no longer of concern then the statusCode is set to “ completed ”. The author is no more tracking this concern and no further actions are expected. | (IPS...ern) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.1.11.19890 x_ActStatusActiveComplete (DYNAMIC) |
|  | hl7:effectiveTime
|
| IVL_TS | 1 … 1 | R | | (IPS...ern) |  |  | hl7:low
|
| IVXB_TS | 1 … 1 | R | This element asserts when the concern became active. This equates to the time the concern was authored in the patient's chart and the author started tracking this concern. | (IPS...ern) |  |  | hl7:high
|
| IVXB_TS | 0 … 1 | C | This element asserts when the clinician deemed there is no longer any need to track the underlying conditions. | (IPS...ern) | | | Constraint | If statusCode/@code="completed" Completed, then effectiveTime *SHALL* contain [1..1] high | | | Schematron assert | role | error | | | | test | not(../hl7:statusCode[@code='completed']) or hl7:high | | | | Message | If statusCode/@code="completed" Completed, then effectiveTime *SHALL* contain [1..1] high | |  | hl7:entryRelationship
|
| | 1 … * | R | Contains 2.16.840.1.113883.10.22.4.1 IPS Allergy or Intolerance (DYNAMIC) | (IPS...ern) |  |  | @typeCode
|
| cs | 1 … 1 | F | SUBJ |  |  | @inversionInd
|
| bl | 0 … 1 | F | false |
|
IPS Allergy Certainty Observation
IPS Allergy or Intolerance
| Id | 2.16.840.1.113883.10.22.4.1 | Effective Date | 2024‑08‑04 09:48:50Other versions this id:  IPSEntryAllergyOrIntolerance as of 2016‑11‑10 IPSEntryAllergyOrIntolerance as of 2016‑11‑10
|
|---|
| Status |  Draft Draft | Version Label | STU2 |
|---|
| Name | IPSEntryAllergyOrIntolerance | Display Name | IPS Allergy or Intolerance |
|---|
| Description | This template reflects a discrete observation about a patient's allergy or intolerance. Because it is a discrete observation, it will have a statusCode of "completed".
The effectiveTime, also referred to as the "biologically relevant time" is the time at which the observation holds for the patient.
For a provider seeing a patient in the clinic today, observing a history of penicillin allergy that developed five years ago, the effectiveTime is five years ago.
The effectiveTime of the Allergy - Intolerance Observation gives an indication of whether or not the underlying allergy/intolerance is resolved. If known to be resolved, then an effectiveTime/high would be present.
If the date of resolution is not known, then effectiveTime/high will be present with a nullFlavor of "UNK".
It is recommended that the agent responsible for an allergy or adverse reaction would be used for describing the allergy, however the possibility that pre-coordinate codes (e.g. "allergy to nuts") will be used has been here also considered.
The agent responsible for an allergy or adverse reaction it is not always a manufactured material (for example, food allergies), nor is it necessarily consumed; however the playingEntity classCode = "MMAT" for all agents, manufactured or not is expected to be used. This choice depends on thecharacteristics of the base CDA R2 specification.
|
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.1 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Associated with | | Associated with 12 concepts | | Id | Name | Data Set |
|---|
| hl7ips-dataelement-173 |  Agent Agent |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-182 |  Allergies/Intolerances Content Status Allergies/Intolerances Content Status |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-184 |  Allergy/Intolerance Allergy/Intolerance |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-185 |  Diagnosis Diagnosis |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-186 |  Clinical Status Clinical Status |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-187 |  Onset date Onset date |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-188 |  End Date End Date |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-189 |  Criticality Criticality |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-190 |  Certainty Certainty |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-191 |  Type of propensity Type of propensity |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-192 |  Reaction Reaction |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-196 |  Agent code Agent code |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|
|
|---|
| Uses | | Uses 4 templates | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.4.6 | Containment |  IPS Reaction Manifestation (STU2) IPS Reaction Manifestation (STU2) | DYNAMIC | | 2.16.840.1.113883.10.22.4.18 | Containment |  IPS Criticality Observation (STU1) IPS Criticality Observation (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.10 | Containment |  IPS Allergy Certainty Observation (STU1) IPS Allergy Certainty Observation (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.4.21 | Containment |  IPS Allergy Status Observation (STU1) IPS Allergy Status Observation (STU1) | DYNAMIC |
|
|
|---|
| Relationship | Specialization: template 2.16.840.1.113883.10.22.4.1 IPS Allergy or Intolerance (2016‑11‑10)
Adaptation: template 2.16.840.1.113883.10.20.1.18 Alert observation (DYNAMIC) ref ccd1-
Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.4.5 IHE Problem Entry (DYNAMIC) ref ch-pcc-
Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.4.6 eHDSI Allergies And Intolerances (DYNAMIC) ref epsos- |
|---|
| Example | | Example | <observation classCode="OBS" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.1"/> <id root=" " extension=" "/> <!-- This is the code that shows what kind of allergy or intolerance -->
<code code="OINT" displayName="Allergy or Intolerance" codeSystem="2.16.840.1.113883.5.4">
<text>
<reference value="#ref1"/> </text> <statusCode code="completed"/> <effectiveTime>
<low value="20170701"/> </effectiveTime> <!-- This is the allergen - the substance that caused the allergy -->
<participant typeCode="CSM">
<participantRole classCode="MANU">
<playingEntity classCode="MMAT">
<code code=" " codeSystem=" ">
<originalText>
<reference value="#substance"/> </originalText> </code> <name>...</name> </playingEntity> </participantRole> </participant> <!-- This is how the allergy manifests itself -->
<entryRelationship typeCode="MFST" inversionInd="true">
<!-- template 2.16.840.1.113883.10.22.4.6 'IPS Reaction Manifestation' (dynamic) -->
</entryRelationship> <entryRelationship typeCode="SUBJ" inversionInd="true">
<!-- template 2.16.840.1.113883.10.22.4.18 'IPS Criticality Observation' (dynamic) -->
</entryRelationship> <entryRelationship typeCode="SUBJ" inversionInd="true">
<!-- template 2.16.840.1.113883.10.22.4.19 'IPS Certainty Observation' (dynamic) -->
</entryRelationship> <entryRelationship typeCode="REFR" inversionInd="false">
<!-- template 2.16.840.1.113883.10.22.4.21 'IPS Allergy Status Observation' (dynamic) -->
</entryRelationship> </code></observation> |
|
|---|
| Example | | No Known Allergies | <observation classCode="OBS" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.1"/> <code code="allergy" displayName="Allergy" codeSystem="2.16.840.1.113883.4.642.1.122">
<statusCode code="completed"/> <effectiveTime>
<low nullFlavor="UNK"/> </effectiveTime> <value code="X-NoKnownAllergy" displayName="No known allergy" codeSystem="2.16.840.1.113883.5.1150.2"/> </code></observation> |
|
|---|
| Example | | No Information available about allergies or intolerances | <observation classCode="OBS" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.1"/> <code code="OINT" displayName="Allergy or Intolerance" codeSystem="2.16.840.1.113883.5.4">
<statusCode code="completed"/> <effectiveTime>
<low nullFlavor="NA"/> </effectiveTime> <value code="no-allergy-info" displayName="No information about allergies" codeSystem="2.16.840.1.113883.5.1150.1"/> </code></observation> |
|
|---|
| Example | | Minimum Set (active propensity; agent known) | <observation classCode="OBS" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.1"/> <code code="OINT" displayName="Allergy or Intolerance" codeSystem="2.16.840.1.113883.5.4">
<statusCode code="completed"/> <effectiveTime>
<low nullFlavor="UNK"/> </effectiveTime> <participant typeCode="CSM">
<participantRole classCode="MANU">
<playingEntity classCode="MMAT">
<code code="13577000" codeSystem="2.16.840.1.113883.6.96" displayName="Nut"/> </playingEntity> </participantRole> </participant> </code></observation> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | | R | | (IPS...nce) | | |  | | hl7ips-dataelement-184 |  Allergy/Intolerance Allergy/Intolerance |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  | @classCode
|
| cs | 1 … 1 | F | OBS |  | @moodCode
|
| cs | 1 … 1 | F | EVN |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...nce) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.1 |  | hl7:id
|
| II | 0 … * | R | | (IPS...nce) |  | hl7:code
|
| CD.IPS | 1 … 1 | M | This element describes whether this condition refers to an allergy, non-allergy intolerance, or unknown class of intolerance (not known to be allergy or intolerance). | (IPS...nce) | | |  | | hl7ips-dataelement-191 |  Type of propensity Type of propensity |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.2 Allergy or Intolerance Type (DYNAMIC) |
|  | hl7:text
|
| ED | 0 … 1 | R | The text element if present points to the text describing the problem being recorded; including any dates, comments, et cetera. The <reference> contains a URI in value attribute. This URI points to the free text description of the problem in the document that is being described.</reference>
| (IPS...nce) |  |  | hl7:reference
|
| TEL | 1 … 1 | M | | (IPS...nce) | | url | 1 … 1 | R | When used it shall refer to the narrative, typically #{label}-{generated-id}, e.g. #xxx-1 |  | hl7:statusCode
|
| CS | 1 … 1 | M | A clinical document normally records only those condition observation events that have been completed, not observations that are in any other state. Therefore, the <statusCode> shall always have code='completed'.</statusCode>
| (IPS...nce) |  |  | @code
|
| cs | 1 … 1 | F | completed |  | hl7:effectiveTime
|
| IVL_TS | 1 … 1 | M | The effectiveTime, also referred to as the "biologically relevant time" is the time at which the observation holds for the patient. For a provider seeing a patient in the clinic today, observing a history of penicillin allergy that developed five years ago, the effectiveTime is five years ago. The effectiveTime of the Allergy - Intolerance Observation may give an indication of whether or not the underlying allergy/intolerance is resolved.
The <low> and <high> values should be no more precise than known, but as precise as possible. | (IPS...nce) |  |  | hl7:low
|
| IVXB_TS | 1 … 1 | R | The effectiveTime/low (a.k.a. "onset date") asserts when the allergy/intolerance became biologically active. | (IPS...nce) | | |  | | hl7ips-dataelement-187 |  Onset date Onset date |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | hl7:high
|
| IVXB_TS | 0 … 1 | C | The effectiveTime/high (a.k.a. "resolution date") asserts when the allergy/intolerance became biologically resolved.
If the date of resolution is not known, then effectiveTime/high will be present with a nullFlavor of "UNK". | (IPS...nce) | | |  | | hl7ips-dataelement-188 |  End Date End Date |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | Constraint | If this condition is known to be resolved, then the effectiveTime/high would be present. |  | hl7:value
|
| CD.IPS (preferred) | 0 … 1 | | | (IPS...nce) | | |  | | hl7ips-dataelement-185 |  Diagnosis Diagnosis |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | CONF | | The value of @code comes preferably from value set 2.16.840.1.113883.11.22.10 IPS Allergy or Intolerance Conditions (DYNAMIC) |
|  | hl7:participant
|
| | 0 … 1 | C | The substance that causes the allergy or intolerance should be specified in the <participant> structure.
This is the preferred way an allergy is supposed to be expressed.
However it is recognized that in some contexts a controlled vocabulary is used for describing the allergy to a substance; or for asserting known absence or unavailability of information.
In this case the <participant> structure shall be omitted.
The agent responsible for an allergy or adverse reaction is not always a manufactured material (for example, food allergies), nor is it necessarily consumed. The following constraints reflect limitations in the base CDA R2 specification, and should be used to represent any type of responsible agent, i.e., use playingEntity classCode = "MMAT" for all agents, manufactured or not. | (IPS...nce) | | |  | | hl7ips-dataelement-173 |  Agent Agent |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | @typeCode
|
| cs | 1 … 1 | F | CSM | | | Constraint | IF the observation/value element is present and valued with a code derived form the 2.16.840.1.113883.11.22.9 Absent or Unknown Allergies value set THEN the observation/participant element used to describe the agent SHALL be omitted. |  |  | hl7:participantRole
|
| | 1 … 1 | R | | (IPS...nce) | | cs | 1 … 1 | F | MANU | | | 1 … 1 | R | | (IPS...nce) | | cs | 1 … 1 | F | MMAT | | CD.IPS (preferred) | 1 … 1 | R | Code for the substance causing the allergy or intolerance. | (IPS...nce) | | |  | | hl7ips-dataelement-196 |  Agent code Agent code |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | CONF | | The value of @code comes preferably from value set 2.16.840.1.113883.11.22.65 IPS Allergy or Intolerance Substances (DYNAMIC) |
|  | hl7:entryRelationship
|
| | 0 … * | R | The contained entry describes the reactions that are manifestations (typeCode='MFST') of the reported allergy or intolerance.
Contains 2.16.840.1.113883.10.22.4.6 IPS Reaction Manifestation (DYNAMIC) | (IPS...nce) | | |  | | hl7ips-dataelement-192 |  Reaction Reaction |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | @typeCode
|
| cs | 1 … 1 | F | MFST |  |  | @inversionInd
|
| bl | 1 … 1 | F | true |  | hl7:entryRelationship
|
| | 0 … 1 | R | Criticality
The contained entry describes the gravity of the potential risk for future life-threatening adverse reactions when exposed to a substance known to cause an adverse reaction in that individual.
Contains 2.16.840.1.113883.10.22.4.18 IPS Criticality Observation (DYNAMIC) | (IPS...nce) | | |  | | hl7ips-dataelement-189 |  Criticality Criticality |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | @typeCode
|
| cs | 1 … 1 | F | SUBJ |  |  | @inversionInd
|
| bl | 1 … 1 | F | true |  | hl7:entryRelationship
|
| | 0 … 1 | R | Certainty or Verification Status
The contained entry describes the certainty associated with a propensity, or potential risk, of a reaction to the identified substance.
Contains 2.16.840.1.113883.10.22.10 IPS Allergy Certainty Observation (DYNAMIC) | (IPS...nce) | | |  | | hl7ips-dataelement-190 |  Certainty Certainty |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | @typeCode
|
| cs | 1 … 1 | F | SUBJ |  |  | @inversionInd
|
| bl | 1 … 1 | F | true |  | hl7:entryRelationship
|
| | 0 … 1 | R | Status of the Allergy or Intolerance
The contained entry describes the current status of the allergy or intolerance, for example, whether it is active, in remission, resolved, and so on ...
Contains 2.16.840.1.113883.10.22.4.21 IPS Allergy Status Observation (DYNAMIC) | (IPS...nce) | | |  | | hl7ips-dataelement-186 |  Clinical Status Clinical Status |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | @typeCode
|
| cs | 1 … 1 | F | REFR |  |  | @inversionInd
|
| bl | 1 … 1 | F | false |
|
IPS Allergy Status Observation
| Id | 2.16.840.1.113883.10.22.4.21 | Effective Date | 2017‑05‑24 |
|---|
| Status |  Under pre-publication review Under pre-publication review | Version Label | STU1 |
|---|
| Name | IPSAllergyStatusObservation | Display Name | IPS Allergy Status Observation |
|---|
| Description | This subordinated observation used by the allergy observation records information about the current status of an allergy or intolerance, for example, whether it is active, in remission, resolved, et cetera. |
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.21 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Associated with | | Associated with 1 concept | | Id | Name | Data Set |
|---|
| hl7ips-dataelement-186 |  Clinical Status Clinical Status |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|
|
|---|
| Relationship | Specialization: template 2.16.840.1.113883.10.22.4.20 IPS Problem Status Observation (DYNAMIC)
Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.4.1.1 IHE Problem Status Observation (2013‑12‑20) ref IHE-PCC- |
|---|
| Example | | Example | <observation classCode="OBS" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.21"/> <templateId root="2.16.840.1.113883.10.22.4.20"/> <code code="33999-4" codeSystem="2.16.840.1.113883.6.1" displayName="Status"/> <text>
<reference value="#cstatus-2"/> </text> <statusCode code="completed"/> <value code="active" displayName="Active" codeSystem="2.16.840.1.113883.4.642.3.155"/></observation> |
|
|---|
|
IPS Body Author
IPS CDA Device
| Id | 2.16.840.1.113883.10.22.9.2 | Effective Date | 2017‑04‑12 |
|---|
| Status |  Under pre-publication review Under pre-publication review | Version Label | STU1 |
|---|
| Name | IPSCDADevice | Display Name | IPS CDA Device |
|---|
| Description | This template provides basic information about a device |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Relationship | Adaptation: template 2.16.840.1.113883.10.12.315 CDA Device (2005‑09‑07) ref ad1bbr- |
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| cs | 0 … 1 | F | DEV | | cs | 0 … 1 | F | INSTANCE | | CE | 0 … 1 | | | (IPS...ice) | hl7:manufacturerModelName
|
| SC | 0 … 1 | | | (IPS...ice) | | SC | 0 … 1 | | | (IPS...ice) |
|
IPS Certainty Observation
IPS Criticality Observation
IPS Immunization
IPS Immunization Medication Information
IPS Internal Reference
| Id | 2.16.840.1.113883.10.22.4.31 | Effective Date | 2017‑05‑02 |
|---|
| Status |  Under pre-publication review Under pre-publication review | Version Label | STU1 |
|---|
| Name | IPSEntryInternalReference | Display Name | IPS Internal Reference |
|---|
| Description | This template is used to reference (point to) information contained in other entries within the same document. |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Relationship | Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.4.4.1 IHE Internal Reference Entry (2013‑12‑20) ref IHE-PCC- |
|---|
| Example | | Reference to an uncoded element | <act classCode="ACT" moodCode="cs">
<templateId root="2.16.840.1.113883.10.22.4.31"/> <id root="1.2.3.999" extension="__example only__"/> <code nullFlavor="NA"/></act> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | | R | | (IPS...nce) |  | @classCode
|
| cs | 1 … 1 | F | ACT |  | @moodCode
|
| cs | 1 … 1 | R | The @moodCode of the reference SHALL match the @moodCode of the referenced element | | | Variable let | Name | refMoodCode | | | | Value | @moodCode | | | | Variable let | Name | refID | | | | Value | concat(hl7:id[1]/@root,'#',hl7:id[1]/@extension) | | | | Variable let | Name | refCode | | | | Value | concat(hl7:code[1]/@code,'#',hl7:code[1]/@codeSystem) | | | | Variable let | Name | reffedObject | | | | Value | (ancestor::hl7:ClinicalDocument//*:id[concat(@root,'#',@extension)=$refID][not(preceding-sibling::hl7:templateId/@root='1.3.6.1.4.1.19376.1.5.3.1.4.4.1')]/parent::*)[1] | | | | Schematron assert | role | error | | | | test | not(exists($reffedObject)) or $reffedObject[@moodCode=$refMoodCode] | | | | Message | The @moodCode of the reference SHALL match the @moodCode of the referenced element | | | | Schematron assert | role | error | | | | test | exists($reffedObject) | | | | Message | The root and extension attributes SHALL identify an element defined elsewhere in the same document. | | | | Schematron assert | role | error | | | | test | not(exists($reffedObject)) or ($reffedObject[not(*:code/@code)] and hl7:code[@nullFlavor='NA']) or $reffedObject/*:code[concat(@code,'#',@codeSystem)=$refCode] | | | | Message | The code of the reference SHALL match the code of the referenced element | |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...nce) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.31 |  | hl7:id
|
| II | 1 … 1 | R | This element shall be present. The root and extension attributes shall identify an element defined elsewhere in the same document. | (IPS...nce) |  | hl7:code
|
| CD | 1 … 1 | R | This element shall be present. It shall be valued when the internal reference is to element that has a <code> element, and shall have the same attributes as the <code> element in the act it references. If the element it references does not have a <code> element, then the nullFlavor attribute should be set to "NA". | (IPS...nce) |  |  | @nullFlavor
|
| cs | 0 … 1 | F | NA |
|
IPS Laboratory Result Observation
| Id | 2.16.840.1.113883.10.22.4.13 | Effective Date | 2024‑08‑04 11:10:20Other versions this id:  IPSLaboratoryResultObservation as of 2017‑03‑21 IPSLaboratoryResultObservation as of 2017‑03‑21
|
|---|
| Status |  Draft Draft | Version Label | STU2 |
|---|
| Name | IPSLaboratoryResultObservation | Display Name | IPS Laboratory Result Observation |
|---|
| Description | This template constrains the results of a clinical laboratory observation. The result observation includes a statusCode to allow recording the status of an observation. “Pending” results (e.g., a test has been run but results have not been reported yet) should be represented as “active” ActStatus. |
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.13 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Uses | | Uses 2 templates | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.4.14 | Include |  IPS Body Author (STU1) IPS Body Author (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.4.22 | Containment |  IPS Comment Activity (STU1) IPS Comment Activity (STU1) | DYNAMIC |
|
|
|---|
| Relationship | Specialization: template 2.16.840.1.113883.10.22.4.13 IPS Laboratory Result Observation (2017‑03‑21)
Adaptation: template 2.16.840.1.113883.10.12.303 CDA Observation (2005‑09‑07) ref ad1bbr-
Adaptation: template 2.16.840.1.113883.10.20.22.4.2 Result Observation (V3) (2015‑08‑01) ref ccda-
Adaptation: template 2.16.840.1.113883.10.22.4.10 IPS Result Observation (2017‑03‑02) |
|---|
| Example | | Example | <observation classCode="OBS" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.13"/> <id root="1.2.3.999" extension="--example only--"/> <code codeSystem="2.16.840.1.113883.6.1" code="41995-2" displayName="Hemoglobin A1c [Mass/volume] in Blood">
<statusCode code="completed"/> <effectiveTime>
<low value="20171113173215"/> </effectiveTime> <value xsi:type="PQ" value="4.8" unit="%"/> <interpretationCode code="H" displayName="High" codeSystem="2.16.840.1.113883.5.83"/> <author>
<!-- template 2.16.840.1.113883.10.22.4.14 'IPS Body Author' (dynamic) -->
</author> <referenceRange>
<observationRange>
<value xsi:type="IVL_PQ">
<low value="1.5" unit="%"/> <high value="4.5" unit="%"/> </value> <interpretationCode code="N" codeSystem="2.16.840.1.113883.5.83"/> </observationRange> </referenceRange> <entryRelationship typeCode="COMP">
<!-- template 2.16.840.1.113883.10.22.4.22 'IPS Comment Activity' (dynamic) -->
</entryRelationship> </code></observation> |
|
|---|
|
IPS Manufactured Material
| Id | 2.16.840.1.113883.10.22.4.3 | Effective Date | 2024‑08‑04 10:47:30Other versions this id:  IPSMedMaterial as of 2021‑08‑02 16:52:27 IPSMedMaterial as of 2021‑08‑02 16:52:27 IPSMedMaterial as of 2016‑11‑10 IPSMedMaterial as of 2016‑11‑10
|
|---|
| Status |  Draft Draft | Version Label | STU2 |
|---|
| Name | IPSMedMaterial | Display Name | IPS Manufactured Material |
|---|
| Description | This entry provides details about the medicinal product.
Due to the current absence of global product identifiers the product is described through a set of identification and descriptive attributes (e.g. active substances, strength, unit of presentation,...) that may be used to integrate jurisdictional product codes.
This shortage will be likely overcome when the ISO IDMP identifiers will be available for concrete usage in the next years, as well as the globally used value sets for products attributes agreed by the ISO IDMP implemention guides. (e.g. GInAs for substances).
Even though there is a quite common consensus about the attributes that should be provided in order to describe a medicine in the context of the international patient summary (e.g. the list of active substances, the strength(s); the administrable pharmaceutical forms;..), this template doesn't require any of them, recommending, above all for cross-borders services, to provide
all the available information that could be helpful for the identification of medications
Jurisdictions could specialize this template making some ofthese attributes required.
It is also recognized that in many contexts structured information about the product, might not be available, and only textual information for describing products (e.g. the product scientific name "amoxicillin 400mg/5mL suspension”) or some of their attributes (e.g. textual strength "875 mg + 125 mg" ; "amoxycillin and clavulanic acid") could be used.
This template attempts to provide a solution that takes in account this current complexity being also ready for including the future IDMP-based solution as soon as they will become available for concrete use.
Since the CDA R2.0 model support only a very limited set of information about the products, extensions based on the R_ProductList (Common Product Model) CMET have been used for conveying such information, aiming to align this solution with that that will be likely used for the IDMP implementation Guide.
|
|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Associated with | | Associated with 4 concepts | | Id | Name | Data Set |
|---|
| hl7ips-dataelement-105 |  Product Code Product Code |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-117 |  Brand Name Brand Name |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-171 |  Product Common Name (and Strength) Product Common Name (and Strength) |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-227 |  Pharmaceutical dose form Pharmaceutical dose form |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|
|
|---|
| Relationship | Specialization: template 2.16.840.1.113883.10.22.4.3 IPS Manufactured Material (2021‑08‑02 16:52:27)
Adaptation: template 2.16.840.1.113883.10.12.311 CDA Material (2005‑09‑07) ref ad1bbr-
Adaptation: template 2.16.840.1.113883.3.1937.777.11.10.147 OpenMed Material (2016‑05‑10 22:43:03) ref epsos- |
|---|
| Example | | Example | <manufacturedMaterial>
<!-- Example with all the IDMP Levels (PhPID,MPID, PCID) and other product attributes (e.g. ingredients, ATC Code, strengths) -->
<templateId root="2.16.840.1.113883.10.22.4.3"/> <code codeSystem="" code="MPID" displayName="" CodeSystemName="MP EMA">
<name>Medicinal Product Name</name> <formCode codeSystem="0.4.0.127.0.16.1.1.2.1" code="10219000" displayName="tablet" CodeSystemName="EDQM"/> <asContent>
<!-- Packaged Medicinal Product (PC) -->
<containerPackagedProduct>
<!-- PC ID-->
<code codeSystem=" " code="PCID" displayName=" ">
<name>...</name> <formCode codeSystem="0.4.0.127.0.16.1.1.2.1" code="" displayName="" CodeSystemName="EDQM"/> </code> </containerPackagedProduct> </asContent> <asSpecializedKind classCode="GRIC">
<!-- Pharmaceutical Substance (ATC Code)-->
<generalizedMaterialKind classCode="MMAT">
<!-- Pharmaceutical Substance (ATC Code)-->
<code code=" " codeSystem="2.16.840.1.113883.6.73" displayName=" " codeSystemName="WHO ATC"/> </generalizedMaterialKind> </asSpecializedKind> <asSpecializedKind>
<!-- Pharmaceutical Product (PhP)-->
<generalizedMaterialKind classCode="MMAT">
<code code="PhPID" codeSystem=" " displayName=" " codeSystemName="PhP EMA">
<name>....</name> </code> </generalizedMaterialKind> </asSpecializedKind> <!-- list of active ingredients -->
<ingredient classCode="ACTI" determinerCode="KIND">
<quantity>
<!-- strength -->
<numerator type="PQ" value="20" unit="mg"/> <denominator type="PQ" value="1" unit="{tablet}"/> </quantity> <ingredientSubstance>
<code codeSystem=" " code="SubstanceID" displayName=" " CodeSystemName="G-SRS">
<name>...</name> </code> </ingredientSubstance> </ingredient> </code></manufacturedMaterial> |
|
|---|
| Example | | Example | <manufacturedMaterial classCode="MMAT" determinerCode="KIND">
<templateId root="2.16.840.1.113883.10.22.4.3"/> <code code="..." codeSystem="1.2.3.999">
<name>name</name> <formCode code="10101000" displayName="Oral drops, solution" codeSystem="0.4.0.127.0.16.1.1.2.1"/> <asContent classCode="CONT">
<containerPackagedProduct classCode="CONT" determinerCode="KIND">
<code>
<name/> <formCode code="..." displayName="..." codeSystem="0.4.0.127.0.16.1.1.2.1"/> <capacityQuantity value="..." unit="..."/> <asContent classCode="CONT">
<containerPackagedProduct classCode="CONT" determinerCode="KIND">
<code>
<name/> <formCode code="..." displayName="..." codeSystem="0.4.0.127.0.16.1.1.2.1"/> <asContent classCode="CONT">
<containerPackagedProduct classCode="CONT" determinerCode="KIND">
<code>
<name/> <formCode code="..." displayName="..." codeSystem="0.4.0.127.0.16.1.1.2.1"/> </code> </containerPackagedProduct> </asContent> </code> </containerPackagedProduct> </asContent> </code> </containerPackagedProduct> </asContent> <asSpecializedKind classCode="GRIC">
<generalizedMaterialKind classCode="MMAT">
<code code="..." codeSystem="2.16.840.1.113883.6.73">
<name/> </code> </generalizedMaterialKind> </asSpecializedKind> <asSpecializedKind classCode="GRIC">
<generalizedMaterialKind classCode="MMAT">
<code>
<name/> </code> </generalizedMaterialKind> </asSpecializedKind> <ingredient classCode="ACTI" determinerCode="KIND">
<quantity>
<numerator value="20" unit="mg"/> <denominator value="100" unit="mL"/> </quantity> <ingredientSubstance>
<code>
<name/> </code> </ingredientSubstance> </ingredient> </code></manufacturedMaterial> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
hl7:manufacturedMaterial
|
| | 0 … * | R | | (IPS...ial) |  | @classCode
|
| cs | 0 … 1 | F | MMAT |  | @determinerCode
|
| cs | 0 … 1 | F | KIND |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...ial) |  |  | @root
|
| oid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.3 | | Choice | 0 … 1 | | Elements to choose from:- hl7:code
- hl7:code[@codeSystem='2.16.840.1.113883.6.96']
|  |  | hl7:code
|
| CE.IPS | 0 … 1 | R | This element is generally used to identify a medicinal product.
When the IDMP identifiers will be concretely available for usage this element will be used for conveying the Medicinal Product Identifier (MPID).
For the time being, it could be optionally used for conveying jurisdictional or agreed cross jurisdictional medicinal product code. | (IPS...ial) | | |  | | hl7ips-dataelement-105 |  Product Code Product Code |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | hl7:code
|
| CE.IPS | 0 … 1 | R | Non IDMP codes from SNOMED CT value set | (IPS...ial) | | where [@codeSystem='2.16.840.1.113883.6.96'] | | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.71 IPS Medications Products (DYNAMIC) |
|  | hl7:name
|
| EN | 0 … 1 | R | This element is supposed to be valorized with the complete Medicinal Product Name as approved by the Medicines Regulatory Agency in a jurisdiction.
The name may be applicable in one or more country/language combinations.
| (IPS...ial) | | |  | | hl7ips-dataelement-117 |  Brand Name Brand Name |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-171 |  Product Common Name (and Strength) Product Common Name (and Strength) |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  | pharm:formCode
|
| CE.IPS | 0 … 1 | R | Administrable Pharmaceutical Dose Form.
This code represents the form of the medication (e.g. tablet, capsule, liquid)
Since the EDQM Standards Terms, together with UCUM, is one of the IDMP terminologies actually available for usage, this code system has been selected as referecne terminology for representing Pharmaceutical Dose forms;Pakages and Route of Administration.
It is known that also alternative jurisdictional and international terminologies are known to be used for this concept domain, as NCI or SNOMED CT.
| (IPS...ial) | | |  | | hl7ips-dataelement-227 |  Pharmaceutical dose form Pharmaceutical dose form |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.25 IPS Medicine Doseform (DYNAMIC) |
| | | Example | <formCode code="10211000" codeSystem="0.4.0.127.0.16.1.1.2.1" codeSystemName="EDQM" displayName="Capsule, soft">...</formCode> |  | pharm:asContent
|
| | 0 … * | | This structure describes the packaging of the medication.
The <pharm:formCode> element provides the code for the particular package.
If the package has a brand name, it can be described in the <pharm:name> element.
The <pharm:capacityQuantity> element describes the capacity of the packaging, while the <pharm:quantity> the actual quantity of inner packaged items in the outer packaging container.
The product might have a single (30 pills bottle) or multiple (5 vials 10 ml; box with 2 blisters of 20 tablets) layers of packaging.
In the latter case, the most inner (nested) item represents the most outer package item.
For example the case
\--Box
\-----2 blisters
\--------20 tablets
is described as "20 tablets" contained by "a blister"; "2 blisters" contained by one box.
The most inner package represents the Packaged Medicinal Product.
When the IDMP Packaged Medicinal Product ID (PCID) will become actually available for usage, the most inner package <code> element will be used to convey the IDMP PCID.
| (IPS...ial) |  |  | @classCode
|
| cs | 1 … 1 | F | CONT | | | Example | Packaged Medicinal Product with multiple layers packaging <asContent>
<containerPackagedProduct>
<!-- Inner Package -->
<code codeSystem="..." code="..." displayName="...">
<asContent>
<containerPackagedProduct>
<!-- Intermediate Package -->
<asContent>
<containerPackagedProduct>
<!-- Outer Package / Packaged Medicinal Product -->
</containerPackagedProduct> </asContent> </containerPackagedProduct> </asContent> </code> </containerPackagedProduct></asContent> | | | Example | Packaged Medicinal Product with formCode <asContent>
<containerPackagedProduct>
<!-- Packaged Medicinal Product -->
<code codeSystem="1.999.999" code="PC_ID" displayName="Packaged Product Name">
<name>100 MIRACLE PILLS(TM)</name> <formCode codeSystem="0.4.0.127.0.16.1.1.2.1" code="30009000" displayName="Box" CodeSystemName="EDQM"/> </code> </containerPackagedProduct></asContent> |  |  | pharm:quantity
|
| PQ | 0 … 1 | | The quantity which specified how many inner packaged content entities are in an outer packaging container entity.
| (IPS...ial) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | Example | <quantity value="20" unit="{tablet}"/> | | | Example | <quantity value="10" unit="mL"/> |  |  | pharm:containerPackagedProduct
|
| | 1 … 1 | R | It represents the most inner Package Item or the Packaged Medicinal Product. | (IPS...ial) | | cs | 1 … 1 | F | CONT | | cs | 1 … 1 | F | KIND | | | 0 … 1 | | If this is also the most outer <pharm:containerPackagedProduct> than the <code> element can be used to convey the (IDMP) Packaged Medicinal Product ID (e.g. the IDMP PCID when it will become actually available for usage).
The presence of the PCID indicates that that element represents the "Packaged Medicinal Product".
| (IPS...ial) | | ST | 0 … 1 | | It represents the Name of the Package Item or of the Packaged Medicinal Product.
If this is also the most outer <pharm:containerPackagedProduct> than this element can be used for the brand name.
| (IPS...ial) | | | Example | <name>AMOXIFEN(R) 20 compresse 20 mg</name> | | CE.IPS | 0 … 1 | R | This element encodes the type of the most inner package item or of the or the Packaged Medicinal Product.
Since the EDQM Standards Terms, together with UCUM, is one of the IDMP terminologies actually available for usage, this code system has been selected as referecne terminology for representing Pharmaceutical Dose forms; Packages and Route of Administration. | (IPS...ial) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.27 Medicine Package (DYNAMIC) |
| | | Example | <formCode code="30007000" codeSystem="0.4.0.127.0.16.1.1.2.1" codeSystemName="EDQM" codeSystemVersion="2010" displayName="Blister">...</formCode> | | PQ | 0 … 1 | | It represents the functional capacity of the container: e.g. bottle containing up to 20 tablets or ampule of 10 ml. | (IPS...ial) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | 0 … * | R | In case of multiple layers of packaging (5 vials 10 ml; box with 2 blisters of 20 tablets) this element can be used for describing the intermediate Packaged Medicinal Product Item or the Packaged Medicinal Product.
For example in the case
\--Box
\-----2 blisters
\--------20 tablets
it describes the "2 blisters"
In the case of
\--Box
\-----5 vials
it represents the Packaged Medicinal Product.
| (IPS...ial) | | cs | 1 … 1 | F | CONT | | PQ | 0 … 1 | R | The quantity which specified how many inner packaged content entities are in an outer packaging container entity.
| (IPS...ial) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | Example | <quantity value="20" unit="{tablet}"/> | | | Example | <quantity value="10" unit="mL"/> |  |  |  |  | pharm:containerPackagedProduct
|
| | 1 … 1 | R | It represents the intermediate Package Item or the Packaged Medicinal Product | (IPS...ial) | | cs | 1 … 1 | F | CONT | | cs | 1 … 1 | F | KIND | | CD.IPS | 0 … 1 | | If this is also the most inner <pharm:containerPackagedProduct> than the <code> element can be used to convey the (IDMP) Packaged Medicinal Product ID (e.g. the IDMP PCID when it will become actually available for usage).
The presence of the PCID indicates that that element represents the "Packaged Medicinal Product".
| (IPS...ial) | | ST | 0 … 1 | R | It represents the Name of the Package Item or of the Packaged Medicinal Product
If this is also the most inner <pharm:containerPackagedProduct> than this element can be used for the brand name.
| (IPS...ial) | | | Example | <name>...</name> | | CE.IPS | 1 … 1 | R | This element encodes the type of the most inner package item or of the or the Packaged Medicinal Product.
Since the EDQM Standards Terms, together with UCUM, is one of the IDMP terminologies actually available for usage, this code system has been selected as reference terminology for representing Pharmaceutical Dose forms; Packages and Route of Administration. | (IPS...ial) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.27 Medicine Package (DYNAMIC) |
| | PQ | 0 … 1 | | It represents the functional capacity of the container: e.g. bottle containing up to 20 tablets or ampule of 10 ml. | (IPS...ial) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | 0 … * | R | In case of multiple layers of packaging (box with 2 blisters of 20 tablets) this element is used for describing the most outer Packaged Medicinal Product Item or the Packaged Medicinal Product.
For example in the case
\--Box
\-----2 blisters
\--------20 tablets
it describes the Packaged Medicinal Product.
| (IPS...ial) | | cs | 1 … 1 | F | CONT | | PQ | 0 … 1 | R | The quantity which specified how many inner packaged content entities are in an outer packaging container entity.
| (IPS...ial) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | Example | <quantity value="20" unit="{tablet}"/> | | | Example | <quantity value="10" unit="mL"/> |  |  |  |  |  |  | pharm:containerPackagedProduct
|
| | 1 … 1 | R | When present, it represents the Packaged Medicinal Product | (IPS...ial) | | cs | 1 … 1 | F | CONT | | cs | 1 … 1 | F | KIND | | CD.IPS | 0 … 1 | | When present, it can be used to convey the (IDMP) Packaged Medicinal Product ID (e.g. the IDMP PCID when it will become actually available for usage). | (IPS...ial) | | ST | 0 … 1 | R | When present, it can be used for the representing the brand name. | (IPS...ial) | | CE.IPS | 1 … 1 | R | When present, it encodes the type of the outer package.
Since the EDQM Standards Terms, together with UCUM, is one of the IDMP terminologies actually available for usage, this code system has been selected as referecne terminology for representing Pharmaceutical Dose forms; Packages and Route of Administration. | (IPS...ial) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.27 Medicine Package (DYNAMIC) |
| | PQ | 0 … 1 | | It represents the functional capacity of the container: e.g. bottle containing up to 20 tablets or ampule of 10 ml. | (IPS...ial) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | |  | pharm:asSpecializedKind
|
| | 0 … 1 | R | This module is used for representing the classification of the Substance according to the WHO Anatomical Therapeutic Chemical (ATC) Classification System.
The classCode of "GRIC" identifies this structure as the representation of a generic equivalent of the medication described in the current Medicine entry.
| (IPS...ial) | | where [generalizedMaterialKind/code/@codeSystem='2.16.840.1.113883.6.73'] | |  |  | @classCode
|
| cs | 1 … 1 | F | GRIC | | | Example | <asSpecializedKind classCode="GRIC">
<generalizedMaterialKind classCode="MMAT">
<!-- Pharmaceutical Substance (ATC Code)-->
<code code=" " codeSystem="2.16.840.1.113883.6.73" displayName=" " codeSystemName="WHO ATC"/> </generalizedMaterialKind></asSpecializedKind> |  |  | pharm:generalizedMaterialKind
|
| | 1 … 1 | M | | (IPS...ial) | | cs | 1 … 1 | F | MMAT | | CD.IPS | 1 … 1 | R | The <code> element contains the ATC code of this medicine. | (IPS...ial) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.29 IPS WHO ATC (DYNAMIC) |
| | | Example | <code codeSystem="2.16.840.1.113883.6.73" code=" " displayName=" " codeSystemName="WHO ATC"/> | | | 0 … * | | | (IPS...ial) |  | pharm:asSpecializedKind
|
| | 0 … * | R | The Medicinal Product can be classified according to various classification systems, which may be jurisdictional or international.
The classification system itself is specified using an appropriate identification system; the controlled term and the controlled term identifier shall be specified.
When the IDMP Pharmaceutical Product Identifier(s) (PhPID Set) will become actually available for use, the PhPID will be represented by the generalizedMaterialKind/code element. | (IPS...ial) |  |  | @classCode
|
| cs | 1 … 1 | F | GRIC | | | Example | <asSpecializedKind classCode="GRIC">
<generalizedMaterialKind classCode="MMAT">
<code code="PhPID_Lvl1" codeSystem="1.999.999" displayName="Pharmaceutical Product Name" codeSystemName="PhPID Level 1">
<name/> </code> </generalizedMaterialKind></asSpecializedKind> |  |  | pharm:generalizedMaterialKind
|
| | | R | | (IPS...ial) | | cs | 1 … 1 | F | MMAT | | CD.IPS | 1 … 1 | R | When the IDMP Pharmaceutical Product Identifier(s) (PhPID Set) will become actually available for use, this element will be used for representing the IDMP PhP Id.
The level and the stratum of the PhPID will be distiguished by the OID of the code system.
| (IPS...ial) | | | 0 … * | R | | (IPS...ial) |  | pharm:ingredient
|
| | 0 … * | R | This module provides the list of the ingredients (substances with a role) used for this product; one or more ingredients may be present.
The classCode of "ACTI" indicates that this is an active ingredient.
| (IPS...ial) |  |  | @classCode
|
| cs | 1 … 1 | R | | | | CONF | | The value of @classCode shall be drawn from value set 2.16.840.1.113883.1.11.10430 RoleClassIngredientEntity (DYNAMIC) |
|  |  | pharm:quantity
|
| | 1 … 1 | M | The medication strength is represented as the ratio of the active ingredient(s) to a unit of medication. The <quantity> element contains the numerator and denominator of the strength ratio.</quantity>
| (IPS...ial) | | | Example | <quantity>...</quantity> | | PQ | 1 … 1 | R | | (IPS...ial) | | cs | 1 … 1 | R | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.30 Medicine Strength Numerator (DYNAMIC) |
| | | 1 … 1 | R | | | PQ | 1 … 1 | R | | (IPS...ial) | | cs | 1 … 1 | R | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.31 Medicine Strength Denominator (DYNAMIC) |
| | | 1 … 1 | R | |  |  | pharm:ingredientSubstance
|
| | 1 … 1 | R | | (IPS...ial) | | cs | 1 … 1 | F | MMAT | | cs | 1 … 1 | F | KIND | | CD.IPS (extensible) | 0 … 1 | C | The IDMP ISO 11238 standard addresses the identification and exchange of regulated information on substances.
The Global Ingredient Archival System (GInAS) will provide a common global identifier for all of the substances used in medicinal products, providing a definition of substances globally consistent with this standard.
Those identifiers however are yet available for concrete usage, therefore in this version of the template, SNOMED CT has been chosen as reference terminology also for the active substances.
This choice will be revised based on the availability and the maturity of GInAS. | (IPS...ial) | | | CONF | | The value of @code should be drawn from value set 2.16.840.1.113883.11.22.32 IPS Medicine Active Substances (DYNAMIC) |
| | ED | 0 … * | | | (IPS...ial) | | TEL | 0 … * | | | (IPS...ial) | | CD | 0 … * | | This element can be used to provide alternative identifications for the described substance. | (IPS...ial) | | | 0 … 1 | C | Name of the substance | (IPS...ial) | | | Schematron assert | role | error | | | | test | pharm:code or pharm:name | | | | Message | Either the name or the code of the substance (or both) shall be provided | |
|
IPS Medical Device
| Id | 2.16.840.1.113883.10.22.4.26 | Effective Date | 2017‑04‑11 |
|---|
| Status |  Under pre-publication review Under pre-publication review | Version Label | STU1 |
|---|
| Name | IPSMedicalDevice | Display Name | IPS Medical Device |
|---|
| Description | The medical devices entry content module describes the kind of device that is, or has been used by the patient |
|---|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.26 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Associated with | | Associated with 6 concepts | | Id | Name | Data Set |
|---|
| hl7ips-dataelement-150 |  Use end date Use end date |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-218 |  Device content Status Device content Status |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-57 |  Device Device |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-58 |  Device Type Device Type |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-59 |  Use start date Use start date |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-60 |  Device Identifier Device Identifier |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|
|
|---|
| Relationship | Adaptation: template 1.3.6.1.4.1.12559.11.10.1.3.1.3.5 Medical Devices (2013‑12‑20) ref epsos- |
|---|
| Example | | Example | <supply moodCode="EVN" classCode="SPLY">
<templateId root="2.16.840.1.113883.10.22.4.26"/> <id root="2.16.840.1.113883.19.811.3"/> <text>
<reference value="#dev_1"/> </text> <effectiveTime xsi:type="IVL_TS">
<low value="20070728"/> </effectiveTime> <participant typeCode="DEV">
<participantRole classCode="MANU">
<id/> <playingDevice classCode="DEV" determinerCode="INSTANCE">
<code code="304184000" displayName="Ankle joint implant" codeSystem="2.16.840.1.113883.6.96"/> </playingDevice> </participantRole> </participant></supply> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | | R | The <supply> element shall be present. The moodCode attribute shall be EVN to reflect that a medical device has been provided.</supply> | (IPS...ice) |  | @classCode
|
| cs | 1 … 1 | F | SPLY |  | @moodCode
|
| cs | 1 … 1 | F | EVN |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...ice) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.26 |  | hl7:id
|
| II | 0 … * | R | This optional element identifies the provision of the device (e.g. implant procedure) | (IPS...ice) |  | hl7:text
|
| ED | 0 … 1 | R | | (IPS...ice) |  |  | hl7:reference
|
| TEL | 1 … 1 | M | | (IPS...ice) | | url | 1 … 1 | R | Reference pointing to the narrative, typically #{label}-{generated-id}, e.g. #xxx-1 |  | hl7:effectiveTime
|
| IVL_TS | 1 … 1 | R |
This element provides the interval of time corresponding to the device usage by/presence in the patient.
| (IPS...ice) |  |  | @xsi:type
|
| st | 1 … 1 | F | IVL_TS |  |  | hl7:low
|
| TS | 1 … 1 | R | The lower bound of the interval represents the start date/time. | (IPS...ice) | | |  | | hl7ips-dataelement-59 |  Use start date Use start date |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | hl7:high
|
| TS | 0 … 1 | C | The upper bound represents the end date/time. If it is not present, the device is still used by or present in the patient. | (IPS...ice) | | |  | | hl7ips-dataelement-150 |  Use end date Use end date |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  | hl7:participant
|
| | 1 … * | R | The device is represented as a participant in the supply structure. The following descriptions apply to the device structure. | (IPS...ice) | | |  | | hl7ips-dataelement-57 |  Device Device |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | @typeCode
|
| cs | 1 … 1 | F | DEV | | | Example | <participant typeCode="DEV">
<participantRole classCode="MANU">
<id root="1.2.3.999" extension="__example_only__"/> <playingDevice classCode="DEV" determinerCode="INSTANCE">
<code code="" codeSystem=""/> <!-- ... -->
</playingDevice> </participantRole></participant> | | | Example | Presence of implanted device not known (situation) <participant typeCode="DEV">
<participantRole classCode="MANU">
<playingDevice>
<code code="000000" codeSystem="2.16.840.1.113883.6.96" displayName="Presence of implanted device not known (situation)"/> </playingDevice> <scopingEntity>
<id root="2.16.840.1.113883.3.3719"/> </scopingEntity> </participantRole></participant> | | | Example | No implant in situ (situation) <participant typeCode="DEV">
<participantRole classCode="MANU">
<playingDevice>
<code code="000000" codeSystem="2.16.840.1.113883.6.96" displayName="No implant in situ (situation)"/> </playingDevice> <scopingEntity>
<id root="2.16.840.1.113883.3.3719"/> </scopingEntity> </participantRole></participant> |  |  | hl7:participantRole
|
| | 1 … 1 | R | | (IPS...ice) | | cs | 1 … 1 | F | MANU | | II | 0 … * | R |
The device ID, e.g. using UDI, is represented by the id element of the participant role. This element is optional, as not all production identifiers (e.g., serial number, lot/batch number, distinct identification number) may be known to the provider or patient.
| (IPS...ice) | | |  | | hl7ips-dataelement-60 |  Device Identifier Device Identifier |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | Example | UDI GS1: DeviceIdentifier 00844588003288, Serial# 10987654d321, Lot# 7654321D <id root="2.16.840.1.113883.3.3719" extension="{01}00844588003288{17}141120{10}7654321D{21}10987654d321"/> | | | Example | UDI ICCBBA: DeviceIdentifier 00844588003288 <id root="2.16.840.1.113883.3.3719" extension="A9999XYZ100T0474"/> | | | Example | UDI HIBCC: Serial# XYZ456789012345678, Lot# LOT123456789012345 <id root="2.16.840.1.113883.3.3719" extension="+H123PARTNO1234567890120/$$420020216LOT123456789012345/SXYZ456789012345678/16D20130202C"/> | | | 1 … 1 | R | The playingDevice element describes the device instance. | (IPS...ice) | | cs | 1 … 1 | F | DEV | | cs | 1 … 1 | F | INSTANCE | | CE.IPS (preferred) | 1 … 1 | R | The device code describes the type of device (e.g. arm prosthesis, arterial stent). | (IPS...ice) | | |  | | hl7ips-dataelement-218 |  Device content Status Device content Status |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-58 |  Device Type Device Type |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | CONF | | The value of @code comes preferably from value set 2.16.840.1.113883.11.22.23 IPS Medical Devices (DYNAMIC) | | or | | The value of @code comes preferably from value set 2.16.840.1.113883.11.22.61 Absent or Unknown Devices (DYNAMIC) |
|
|
IPS Medication Information (detail)
| Id | 2.16.840.1.113883.10.22.4.2 | Effective Date | 2024‑08‑04 10:45:46Other versions this id:  IPSManufacturedProduct as of 2016‑11‑10 IPSManufacturedProduct as of 2016‑11‑10
|
|---|
| Status |  Draft Draft | Version Label | STU2 |
|---|
| Name | IPSManufacturedProduct | Display Name | IPS Medication Information (detail) |
|---|
| Description | This entry describes the consumable subject of the medication statement.
All the information about the medication is provided in the included IPS Manufactured Material template. |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Associated with | | Associated with 1 concept | | Id | Name | Data Set |
|---|
| hl7ips-dataelement-2 |  Medicinal Product Medicinal Product |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|
|
|---|
| Uses | | Uses 1 template | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.4.3 | Include |  IPS Manufactured Material (STU2) IPS Manufactured Material (STU2) | DYNAMIC |
|
|
|---|
| Relationship | Specialization: template 2.16.840.1.113883.10.22.4.2 IPS Medication Information (detail) (2016‑11‑10)
Adaptation: template 2.16.840.1.113883.10.12.312 CDA ManufacturedProduct (2005‑09‑07) ref ad1bbr-
Specialization: template 2.16.840.1.113883.10.21.4.11 UV Medication Information (detail) (DYNAMIC) ref pharmcda- |
|---|
| Example | | Example | <manufacturedProduct classCode="MANU">
<templateId root="2.16.840.1.113883.10.22.4.2"/> <!-- include template 2.16.840.1.113883.10.22.4.3 'IPS Manufactured Material' (dynamic) 1..1 R -->
</manufacturedProduct> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | 0 … * | R | | (IPS...uct) | | |  | | hl7ips-dataelement-2 |  Medicinal Product Medicinal Product |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  | @classCode
|
| cs | 0 … 1 | F | MANU |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...uct) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.2 | | Included | 1 … 1 | R | from 2.16.840.1.113883.10.22.4.3 IPS Manufactured Material (DYNAMIC) |  | hl7:manufacturedMaterial
|
| | 1 … 1 | R | | (IPS...uct) |  |  | @classCode
|
| cs | 0 … 1 | F | MMAT |  |  | @determinerCode
|
| cs | 0 … 1 | F | KIND |  |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...uct) | | oid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.3 | | Choice | 0 … 1 | | Elements to choose from:- hl7:code
- hl7:code[@codeSystem='2.16.840.1.113883.6.96']
| | CE.IPS | 0 … 1 | R | This element is generally used to identify a medicinal product.
When the IDMP identifiers will be concretely available for usage this element will be used for conveying the Medicinal Product Identifier (MPID).
For the time being, it could be optionally used for conveying jurisdictional or agreed cross jurisdictional medicinal product code. | (IPS...uct) | | |  | | hl7ips-dataelement-105 |  Product Code Product Code |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | CE.IPS | 0 … 1 | R | Non IDMP codes from SNOMED CT value set | (IPS...uct) | | where [@codeSystem='2.16.840.1.113883.6.96'] | | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.71 IPS Medications Products (DYNAMIC) |
|  |  | hl7:name
|
| EN | 0 … 1 | R | This element is supposed to be valorized with the complete Medicinal Product Name as approved by the Medicines Regulatory Agency in a jurisdiction.
The name may be applicable in one or more country/language combinations.
| (IPS...uct) | | |  | | hl7ips-dataelement-117 |  Brand Name Brand Name |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-171 |  Product Common Name (and Strength) Product Common Name (and Strength) |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | pharm:formCode
|
| CE.IPS | 0 … 1 | R | Administrable Pharmaceutical Dose Form.
This code represents the form of the medication (e.g. tablet, capsule, liquid)
Since the EDQM Standards Terms, together with UCUM, is one of the IDMP terminologies actually available for usage, this code system has been selected as referecne terminology for representing Pharmaceutical Dose forms;Pakages and Route of Administration.
It is known that also alternative jurisdictional and international terminologies are known to be used for this concept domain, as NCI or SNOMED CT.
| (IPS...uct) | | |  | | hl7ips-dataelement-227 |  Pharmaceutical dose form Pharmaceutical dose form |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.25 IPS Medicine Doseform (DYNAMIC) |
| | | Example | <formCode code="10211000" codeSystem="0.4.0.127.0.16.1.1.2.1" codeSystemName="EDQM" displayName="Capsule, soft">...</formCode> |  |  | pharm:asContent
|
| | 0 … * | | This structure describes the packaging of the medication.
The <pharm:formCode> element provides the code for the particular package.
If the package has a brand name, it can be described in the <pharm:name> element.
The <pharm:capacityQuantity> element describes the capacity of the packaging, while the <pharm:quantity> the actual quantity of inner packaged items in the outer packaging container.
The product might have a single (30 pills bottle) or multiple (5 vials 10 ml; box with 2 blisters of 20 tablets) layers of packaging.
In the latter case, the most inner (nested) item represents the most outer package item.
For example the case
\--Box
\-----2 blisters
\--------20 tablets
is described as "20 tablets" contained by "a blister"; "2 blisters" contained by one box.
The most inner package represents the Packaged Medicinal Product.
When the IDMP Packaged Medicinal Product ID (PCID) will become actually available for usage, the most inner package <code> element will be used to convey the IDMP PCID.
| (IPS...uct) | | cs | 1 … 1 | F | CONT | | | Example | Packaged Medicinal Product with multiple layers packaging <asContent>
<containerPackagedProduct>
<!-- Inner Package -->
<code codeSystem="..." code="..." displayName="...">
<asContent>
<containerPackagedProduct>
<!-- Intermediate Package -->
<asContent>
<containerPackagedProduct>
<!-- Outer Package / Packaged Medicinal Product -->
</containerPackagedProduct> </asContent> </containerPackagedProduct> </asContent> </code> </containerPackagedProduct></asContent> | | | Example | Packaged Medicinal Product with formCode <asContent>
<containerPackagedProduct>
<!-- Packaged Medicinal Product -->
<code codeSystem="1.999.999" code="PC_ID" displayName="Packaged Product Name">
<name>100 MIRACLE PILLS(TM)</name> <formCode codeSystem="0.4.0.127.0.16.1.1.2.1" code="30009000" displayName="Box" CodeSystemName="EDQM"/> </code> </containerPackagedProduct></asContent> | | PQ | 0 … 1 | | The quantity which specified how many inner packaged content entities are in an outer packaging container entity.
| (IPS...uct) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | Example | <quantity value="20" unit="{tablet}"/> | | | Example | <quantity value="10" unit="mL"/> |  |  |  | pharm:containerPackagedProduct
|
| | 1 … 1 | R | It represents the most inner Package Item or the Packaged Medicinal Product. | (IPS...uct) | | cs | 1 … 1 | F | CONT | | cs | 1 … 1 | F | KIND | | | 0 … 1 | | If this is also the most outer <pharm:containerPackagedProduct> than the <code> element can be used to convey the (IDMP) Packaged Medicinal Product ID (e.g. the IDMP PCID when it will become actually available for usage).
The presence of the PCID indicates that that element represents the "Packaged Medicinal Product".
| (IPS...uct) | | ST | 0 … 1 | | It represents the Name of the Package Item or of the Packaged Medicinal Product.
If this is also the most outer <pharm:containerPackagedProduct> than this element can be used for the brand name.
| (IPS...uct) | | | Example | <name>AMOXIFEN(R) 20 compresse 20 mg</name> | | CE.IPS | 0 … 1 | R | This element encodes the type of the most inner package item or of the or the Packaged Medicinal Product.
Since the EDQM Standards Terms, together with UCUM, is one of the IDMP terminologies actually available for usage, this code system has been selected as referecne terminology for representing Pharmaceutical Dose forms; Packages and Route of Administration. | (IPS...uct) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.27 Medicine Package (DYNAMIC) |
| | | Example | <formCode code="30007000" codeSystem="0.4.0.127.0.16.1.1.2.1" codeSystemName="EDQM" codeSystemVersion="2010" displayName="Blister">...</formCode> | | PQ | 0 … 1 | | It represents the functional capacity of the container: e.g. bottle containing up to 20 tablets or ampule of 10 ml. | (IPS...uct) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | 0 … * | R | In case of multiple layers of packaging (5 vials 10 ml; box with 2 blisters of 20 tablets) this element can be used for describing the intermediate Packaged Medicinal Product Item or the Packaged Medicinal Product.
For example in the case
\--Box
\-----2 blisters
\--------20 tablets
it describes the "2 blisters"
In the case of
\--Box
\-----5 vials
it represents the Packaged Medicinal Product.
| (IPS...uct) | | cs | 1 … 1 | F | CONT | | PQ | 0 … 1 | R | The quantity which specified how many inner packaged content entities are in an outer packaging container entity.
| (IPS...uct) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | Example | <quantity value="20" unit="{tablet}"/> | | | Example | <quantity value="10" unit="mL"/> |  |  |  |  |  | pharm:containerPackagedProduct
|
| | 1 … 1 | R | It represents the intermediate Package Item or the Packaged Medicinal Product | (IPS...uct) | | cs | 1 … 1 | F | CONT | | cs | 1 … 1 | F | KIND | | CD.IPS | 0 … 1 | | If this is also the most inner <pharm:containerPackagedProduct> than the <code> element can be used to convey the (IDMP) Packaged Medicinal Product ID (e.g. the IDMP PCID when it will become actually available for usage).
The presence of the PCID indicates that that element represents the "Packaged Medicinal Product".
| (IPS...uct) | | ST | 0 … 1 | R | It represents the Name of the Package Item or of the Packaged Medicinal Product
If this is also the most inner <pharm:containerPackagedProduct> than this element can be used for the brand name.
| (IPS...uct) | | | Example | <name>...</name> | | CE.IPS | 1 … 1 | R | This element encodes the type of the most inner package item or of the or the Packaged Medicinal Product.
Since the EDQM Standards Terms, together with UCUM, is one of the IDMP terminologies actually available for usage, this code system has been selected as reference terminology for representing Pharmaceutical Dose forms; Packages and Route of Administration. | (IPS...uct) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.27 Medicine Package (DYNAMIC) |
| | PQ | 0 … 1 | | It represents the functional capacity of the container: e.g. bottle containing up to 20 tablets or ampule of 10 ml. | (IPS...uct) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | 0 … * | R | In case of multiple layers of packaging (box with 2 blisters of 20 tablets) this element is used for describing the most outer Packaged Medicinal Product Item or the Packaged Medicinal Product.
For example in the case
\--Box
\-----2 blisters
\--------20 tablets
it describes the Packaged Medicinal Product.
| (IPS...uct) | | cs | 1 … 1 | F | CONT | | PQ | 0 … 1 | R | The quantity which specified how many inner packaged content entities are in an outer packaging container entity.
| (IPS...uct) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | Example | <quantity value="20" unit="{tablet}"/> | | | Example | <quantity value="10" unit="mL"/> |  |  |  |  |  |  |  | pharm:containerPackagedProduct
|
| | 1 … 1 | R | When present, it represents the Packaged Medicinal Product | (IPS...uct) | | cs | 1 … 1 | F | CONT | | cs | 1 … 1 | F | KIND | | CD.IPS | 0 … 1 | | When present, it can be used to convey the (IDMP) Packaged Medicinal Product ID (e.g. the IDMP PCID when it will become actually available for usage). | (IPS...uct) | | ST | 0 … 1 | R | When present, it can be used for the representing the brand name. | (IPS...uct) | | CE.IPS | 1 … 1 | R | When present, it encodes the type of the outer package.
Since the EDQM Standards Terms, together with UCUM, is one of the IDMP terminologies actually available for usage, this code system has been selected as referecne terminology for representing Pharmaceutical Dose forms; Packages and Route of Administration. | (IPS...uct) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.27 Medicine Package (DYNAMIC) |
| | PQ | 0 … 1 | | It represents the functional capacity of the container: e.g. bottle containing up to 20 tablets or ampule of 10 ml. | (IPS...uct) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | |  |  | pharm:asSpecializedKind
|
| | 0 … 1 | R | This module is used for representing the classification of the Substance according to the WHO Anatomical Therapeutic Chemical (ATC) Classification System.
The classCode of "GRIC" identifies this structure as the representation of a generic equivalent of the medication described in the current Medicine entry.
| (IPS...uct) | | where [generalizedMaterialKind/code/@codeSystem='2.16.840.1.113883.6.73'] | | | cs | 1 … 1 | F | GRIC | | | Example | <asSpecializedKind classCode="GRIC">
<generalizedMaterialKind classCode="MMAT">
<!-- Pharmaceutical Substance (ATC Code)-->
<code code=" " codeSystem="2.16.840.1.113883.6.73" displayName=" " codeSystemName="WHO ATC"/> </generalizedMaterialKind></asSpecializedKind> |  |  |  | pharm:generalizedMaterialKind
|
| | 1 … 1 | M | | (IPS...uct) | | cs | 1 … 1 | F | MMAT | | CD.IPS | 1 … 1 | R | The <code> element contains the ATC code of this medicine. | (IPS...uct) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.29 IPS WHO ATC (DYNAMIC) |
| | | Example | <code codeSystem="2.16.840.1.113883.6.73" code=" " displayName=" " codeSystemName="WHO ATC"/> | | | 0 … * | | | (IPS...uct) |  |  | pharm:asSpecializedKind
|
| | 0 … * | R | The Medicinal Product can be classified according to various classification systems, which may be jurisdictional or international.
The classification system itself is specified using an appropriate identification system; the controlled term and the controlled term identifier shall be specified.
When the IDMP Pharmaceutical Product Identifier(s) (PhPID Set) will become actually available for use, the PhPID will be represented by the generalizedMaterialKind/code element. | (IPS...uct) | | cs | 1 … 1 | F | GRIC | | | Example | <asSpecializedKind classCode="GRIC">
<generalizedMaterialKind classCode="MMAT">
<code code="PhPID_Lvl1" codeSystem="1.999.999" displayName="Pharmaceutical Product Name" codeSystemName="PhPID Level 1">
<name/> </code> </generalizedMaterialKind></asSpecializedKind> |  |  |  | pharm:generalizedMaterialKind
|
| | | R | | (IPS...uct) | | cs | 1 … 1 | F | MMAT | | CD.IPS | 1 … 1 | R | When the IDMP Pharmaceutical Product Identifier(s) (PhPID Set) will become actually available for use, this element will be used for representing the IDMP PhP Id.
The level and the stratum of the PhPID will be distiguished by the OID of the code system.
| (IPS...uct) | | | 0 … * | R | | (IPS...uct) |  |  | pharm:ingredient
|
| | 0 … * | R | This module provides the list of the ingredients (substances with a role) used for this product; one or more ingredients may be present.
The classCode of "ACTI" indicates that this is an active ingredient.
| (IPS...uct) | | cs | 1 … 1 | R | | | | CONF | | The value of @classCode shall be drawn from value set 2.16.840.1.113883.1.11.10430 RoleClassIngredientEntity (DYNAMIC) |
| | | 1 … 1 | M | The medication strength is represented as the ratio of the active ingredient(s) to a unit of medication. The <quantity> element contains the numerator and denominator of the strength ratio.</quantity>
| (IPS...uct) | | | Example | <quantity>...</quantity> | | PQ | 1 … 1 | R | | (IPS...uct) | | cs | 1 … 1 | R | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.30 Medicine Strength Numerator (DYNAMIC) |
| | | 1 … 1 | R | | | PQ | 1 … 1 | R | | (IPS...uct) | | cs | 1 … 1 | R | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.31 Medicine Strength Denominator (DYNAMIC) |
| | | 1 … 1 | R | |  |  |  | pharm:ingredientSubstance
|
| | 1 … 1 | R | | (IPS...uct) | | cs | 1 … 1 | F | MMAT | | cs | 1 … 1 | F | KIND | | CD.IPS (extensible) | 0 … 1 | C | The IDMP ISO 11238 standard addresses the identification and exchange of regulated information on substances.
The Global Ingredient Archival System (GInAS) will provide a common global identifier for all of the substances used in medicinal products, providing a definition of substances globally consistent with this standard.
Those identifiers however are yet available for concrete usage, therefore in this version of the template, SNOMED CT has been chosen as reference terminology also for the active substances.
This choice will be revised based on the availability and the maturity of GInAS. | (IPS...uct) | | | CONF | | The value of @code should be drawn from value set 2.16.840.1.113883.11.22.32 IPS Medicine Active Substances (DYNAMIC) |
| | ED | 0 … * | | | (IPS...uct) | | TEL | 0 … * | | | (IPS...uct) | | CD | 0 … * | | This element can be used to provide alternative identifications for the described substance. | (IPS...uct) | | | 0 … 1 | C | Name of the substance | (IPS...uct) | | | Schematron assert | role | error | | | | test | pharm:code or pharm:name | | | | Message | Either the name or the code of the substance (or both) shall be provided | |
|
IPS Medication Statement
| Id | 2.16.840.1.113883.10.22.4.4 | Effective Date | 2024‑08‑04 10:41:54Other versions this id:  IPSMedicationStatement as of 2021‑09‑02 12:17:54 IPSMedicationStatement as of 2021‑09‑02 12:17:54 IPSMedicationStatement as of 2016‑11‑11 IPSMedicationStatement as of 2016‑11‑11
|
|---|
| Status |  Draft Draft | Version Label | STU2 |
|---|
| Name | IPSMedicationStatement | Display Name | IPS Medication Statement |
|---|
| Description | An IPS Medication entry describes a medication statement, that is a substance administration that has actually occurred (e.g., pills ingested or injections given) or are intended to occur (e.g., "take 2 tablets twice a day for the next 10 days"). Medication activities in "INT" mood are reflections of what a clinician
intends a patient to be taking. For example, a clinician may intend that a patient to be administered Lisinopril 20 mg PO for blood pressure control. If what was actually administered was Lisinopril 10 mg., then the Medication activities in the "EVN" mood would reflect actual use.
The source of this information can be the patient, significant other (such as a family member or spouse), or a clinician. A common scenario where this information is captured is during the history taking process during a patient visit or stay, but it could be derived from the medications information recorded into a GP's EHR-system, in form of prescribed medication, or
administration statements.
The medication information may come from sources such as the patient's memory, from a prescription bottle, or from a list of medications the patient, clinician or other party maintains.
A medication statement is usually less specific than an a prescription or a medication administration record.
This entry is composed by a main substanceAdministration act and a subordinate substanceAdministration act, unless it is asserted that there are no medications data.
The first conveys information as the product, the period of administration and the route of administration; the latter is used to provide dosage information as the frequency of intakes or the amount of the medication given.
|
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.4 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Associated with | | Associated with 3 concepts | | Id | Name | Data Set |
|---|
| hl7ips-dataelement-102 |  Route of administration Route of administration |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-104 |  Medication Medication |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-220 |  Medication Summary content status Medication Summary content status |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|
|
|---|
| Uses | | Uses 4 templates | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.21.9.1 | Include |  UV Use Period (2023) UV Use Period (2023) | DYNAMIC | | 2.16.840.1.113883.10.22.4.2 | Include |  IPS Medication Information (detail) (STU2) IPS Medication Information (detail) (STU2) | DYNAMIC | | 2.16.840.1.113883.10.22.4.14 | Include |  IPS Body Author (STU1) IPS Body Author (STU1) | DYNAMIC | | 2.16.840.1.113883.10.21.4.6 | Containment |  UV Subordinate Substance Administration (2023) UV Subordinate Substance Administration (2023) | DYNAMIC |
|
|
|---|
| Relationship | Specialization: template 2.16.840.1.113883.10.22.4.4 IPS Medication Statement (2021‑09‑02 12:17:54)
Version: template 2.16.840.1.113883.10.22.4.4 IPS Medication Statement (2016‑11‑11)
Adaptation: template 1.3.6.1.4.1.12559.11.10.1.3.1.3.4 Medication Item (2013‑12‑20) ref epsos-
Specialization: template 2.16.840.1.113883.10.21.4.7 UV Medication Statement (DYNAMIC) ref pharmcda- |
|---|
| Example | | Example | <substanceAdministration classCode="SBADM" moodCode="INT">
<templateId root="2.16.840.1.113883.10.22.4.4"/> <code code="DRUG" codeSystem="2.16.840.1.113883.5.4" displayName="Drug">
<statusCode code="active"/> <effectiveTime>
<width value="2" unit="wk"/> </effectiveTime> <consumable typeCode="CSM">
<!-- template 'IPS ManufacturedProduct' (dynamic) -->
</consumable> <entryRelationship typeCode="COMP">
<substanceAdministration classCode="SBADM" moodCode="EVN">
<statusCode code="active"/> <effectiveTime xsi:type="PIVL_TS" institutionSpecified="true">
<period value="12" unit="h"/> </effectiveTime> <doseQuantity value="2" unit="{puff}"/> <consumable>
<manufacturedProduct>
<manufacturedMaterial nullFlavor="NA"/> </manufacturedProduct> </consumable> </substanceAdministration> </entryRelationship> </code></substanceAdministration> |
|
|---|
| Example | | No medication infos | <substanceAdministration classCode="SBADM" moodCode="INT">
<templateId root="2.16.840.1.113883.10.22.4.4"/> <code code="no-medication-info" codeSystem="2.16.840.1.113883.5.1150.1" displayName="No information about medications">
<statusCode code="completed"/> <effectiveTime nullFlavor="NA" xsi:type="IVL_TS"/> <consumable>
<manufacturedProduct>
<manufacturedMaterial nullFlavor="NA"/> </manufacturedProduct> </consumable> </code></substanceAdministration> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
hl7:substanceAdministration
|
| | | R | | (IPS...ent) | | |  | | hl7ips-dataelement-104 |  Medication Medication |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  | @classCode
|
| cs | 1 … 1 | F | SBADM |  | @moodCode
|
| cs | 1 … 1 | R | If the statement refers to a prescribed medication then a <substanceAdministration> intent (moodCode='INT') is used; otherwise, to record medications which are stated to have taken, the moodCode shall be set to 'EVN'.</substanceAdministration>
| | | CONF | | The value of @moodCode shall be drawn from value set 2.16.840.1.113883.11.20.9.18 MoodCodeEvnInt (DYNAMIC) |
|  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...ent) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.4 |  | hl7:id
|
| II | 0 … * | R | | (IPS...ent) |  | hl7:code
|
| CD.IPS | 1 … 1 | R | The <code> element is valorized with the Substance Administration ACT code "DRUG" unless it is used for asserting the known absence of medication treatments or no information about them.
| (IPS...ent) | | |  | | hl7ips-dataelement-220 |  Medication Summary content status Medication Summary content status |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.14 DRUGActCode (DYNAMIC) | | or | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.79 IPS No Medications (DYNAMIC) |
|  | hl7:text
|
| ED | 0 … 1 | R | The URI given in the value attribute of the <reference> element points to an element in the narrative content that contains the complete text describing the medication.
In a CDA document, the URI given in the value attribute of the <reference> element points to an element in the narrative content that contains the complete text describing the medication.
| (IPS...ent) |  |  | hl7:reference
|
| TEL | 1 … 1 | M | | (IPS...ent) | | url | 1 … 1 | R | Reference pointing to the narrative, typically #{label}-{generated-id}, e.g. #xxx-1 |  | hl7:statusCode
|
| CS | 1 … 1 | M | | (IPS...ent) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.12 ActStatusActiveCompletedAbortedSuspended (DYNAMIC) |
| | | Example | <statusCode code="active"/> | | Included | 1 … 1 | R | from 2.16.840.1.113883.10.21.9.1 UV Use Period (DYNAMIC) | | Choice | 1 … 1 | | The effectiveTime element encodes the use period of the medication, it is always expressed as an interval of time.
It may be expressed using the low and high OR with the width element.
The first is used to indicate a specified interval (e.g. from march 15th, 2017); the latter for indicating a 'floating' period (e.g. 2 weeks). Elements to choose from:- hl7:effectiveTime[hl7:low | hl7:high][not(hl7:width)]
- hl7:effectiveTime[hl7:width][not(hl7:low|hl7:high)]
- hl7:effectiveTime[hl7:low | hl7:width][not(hl7:high)]
|  |  | hl7:effectiveTime
|
| IVL_TS | 0 … 1 | C | Case 1: specified interval
The low and high values of the first effectiveTime element represent the start and stop times for the medication. The low value represents the start time, and the high value represents the stop time. If either the low or the high value is unknown, this shall be recorded by setting the nullFlavor attribute to UNK.
In case of unbounded period (continuous therapy) the high element will be valued with the nullFlavor attribute to NA.
The high value records the end of the medication regime according to the information provided in the prescription or order. For example, if the prescription is for enough medication to last 30 days, then the high value should contain a date that is 30 days later then the low value. The rationale is that a provider, seeing a prescription that has not been refilled would normally assume that the medication is no longer being taken, even if the intent of the treatment plan is to continue the medication indefinitely. | (IPS...ent) | where [hl7:low or
[not(hl7:width)] | | | cs | 0 … 1 | | | | | Example | Known Interval <effectiveTime type="IVL_TS">
<low value="20130321"/> <high value="20140321"/></effectiveTime> | | | Example | Information not available about the period <effectiveTime type="IVL_TS" nullFlavor="NI"/> | | | Example | Unknown end date <effectiveTime type="IVL_TS">
<low value="20130321"/> <high nullFlavor="UNK"/></effectiveTime> | | | Example | continous therapy <effectiveTime type="IVL_TS">
<low value="20130321"/> <high nullFlavor="NA"/></effectiveTime> | | IVXB_TS | 1 … 1 | R | | (IPS...ent) | | IVXB_TS | 0 … 1 | R | | (IPS...ent) |  |  | hl7:effectiveTime
|
| IVL_TS | 0 … 1 | C | Case 2: 'floating' period:
The width element is used to specify a period of (actual or intended) administration that is not anchored to any specific date (e.g. a two weeks therapy) | (IPS...ent) | where [hl7:width] [not(hl7:lowor
hl7:high)] | | | | Example | 2 week period <effectiveTime type="IVL_TS">
<width value="2" unit="w"/></effectiveTime> | | | | NP | | (IPS...ent) | | | | NP | | (IPS...ent) | | | | NP | | (IPS...ent) | | PQ | 1 … 1 | R | | (IPS...ent) | | cs | 1 … 1 | R | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.21.1 Medication Time Units (UCUM) (DYNAMIC) |
|  |  | hl7:effectiveTime
|
| IVL_TS | 0 … 1 | C | Case 3: anchored period:
The width element is used to specify a period of (actual or intended) administration anchored to a specific date (e.g. a two weeks therapy starting today) | (IPS...ent) | where [hl7:low or
[not(hl7:high)] | | | | Example | 2 week period starting on 2013-03-21 <effectiveTime type="IVL_TS">
<low value="20130321"/> <width value="2" unit="w"/></effectiveTime> | | IVXB_TS | 0 … 1 | C | | (IPS...ent) | | PQ | 1 … 1 | R | | (IPS...ent) | | cs | 1 … 1 | R | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.21.1 Medication Time Units (UCUM) (DYNAMIC) |
|  | hl7:routeCode
|
| CE.IPS | 0 … 1 | R | The <routeCode> element specifies the route of administration using the EDQM route of administration vocabulary.
A code must be specified if the route is known.
Since the EDQM Standards Terms, together with UCUM, is one of the IDMP terminologies actually available for usage, this code system has been selected as referecne terminology for representing Pharmaceutical Dose forms; Packages and Route of Administration.
It is known that also alternative jurisdictional and international terminologies are also used for this concept domain, as NCI or SNOMED CT.
Official NCI and EDQM maps for the route of administration are available from the EDQM site.
| (IPS...ent) | | |  | | hl7ips-dataelement-102 |  Route of administration Route of administration |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.33 IPS Medicine Route of Administration (DYNAMIC) |
|  | hl7:doseQuantity
|
| IVL_PQ | | NP | | (IPS...ent) |  | hl7:rateQuantity
|
| IVL_PQ | | NP | | (IPS...ent) |  | hl7:administrationUnitCode
|
| CE | | NP | | (IPS...ent) |  | hl7:consumable
|
| | 1 … 1 | M | | (IPS...ent) |  |  | @typeCode
|
| cs | 1 … 1 | F | CSM | | Included | | | from 2.16.840.1.113883.10.22.4.2 IPS Medication Information (detail) (DYNAMIC) |  |  | hl7:manufacturedProduct
|
| | 0 … * | R | | (IPS...ent) | | |  | | hl7ips-dataelement-2 |  Medicinal Product Medicinal Product |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | cs | 0 … 1 | F | MANU | | II | 1 … 1 | M | | (IPS...ent) | | uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.2 | | Included | 1 … 1 | R | from 2.16.840.1.113883.10.22.4.3 IPS Manufactured Material (DYNAMIC) |  |  |  | hl7:manufacturedMaterial
|
| | 1 … 1 | R | | (IPS...ent) | | cs | 0 … 1 | F | MMAT | | cs | 0 … 1 | F | KIND | | II | 1 … 1 | M | | (IPS...ent) | | oid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.3 | | Choice | 0 … 1 | | Elements to choose from:- hl7:code
- hl7:code[@codeSystem='2.16.840.1.113883.6.96']
| | CE.IPS | 0 … 1 | R | This element is generally used to identify a medicinal product.
When the IDMP identifiers will be concretely available for usage this element will be used for conveying the Medicinal Product Identifier (MPID).
For the time being, it could be optionally used for conveying jurisdictional or agreed cross jurisdictional medicinal product code. | (IPS...ent) | | |  | | hl7ips-dataelement-105 |  Product Code Product Code |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | CE.IPS | 0 … 1 | R | Non IDMP codes from SNOMED CT value set | (IPS...ent) | | where [@codeSystem='2.16.840.1.113883.6.96'] | | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.71 IPS Medications Products (DYNAMIC) |
| | EN | 0 … 1 | R | This element is supposed to be valorized with the complete Medicinal Product Name as approved by the Medicines Regulatory Agency in a jurisdiction.
The name may be applicable in one or more country/language combinations.
| (IPS...ent) | | |  | | hl7ips-dataelement-117 |  Brand Name Brand Name |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-171 |  Product Common Name (and Strength) Product Common Name (and Strength) |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | CE.IPS | 0 … 1 | R | Administrable Pharmaceutical Dose Form.
This code represents the form of the medication (e.g. tablet, capsule, liquid)
Since the EDQM Standards Terms, together with UCUM, is one of the IDMP terminologies actually available for usage, this code system has been selected as referecne terminology for representing Pharmaceutical Dose forms;Pakages and Route of Administration.
It is known that also alternative jurisdictional and international terminologies are known to be used for this concept domain, as NCI or SNOMED CT.
| (IPS...ent) | | |  | | hl7ips-dataelement-227 |  Pharmaceutical dose form Pharmaceutical dose form |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.25 IPS Medicine Doseform (DYNAMIC) |
| | | Example | <formCode code="10211000" codeSystem="0.4.0.127.0.16.1.1.2.1" codeSystemName="EDQM" displayName="Capsule, soft">...</formCode> | | | 0 … * | | This structure describes the packaging of the medication.
The <pharm:formCode> element provides the code for the particular package.
If the package has a brand name, it can be described in the <pharm:name> element.
The <pharm:capacityQuantity> element describes the capacity of the packaging, while the <pharm:quantity> the actual quantity of inner packaged items in the outer packaging container.
The product might have a single (30 pills bottle) or multiple (5 vials 10 ml; box with 2 blisters of 20 tablets) layers of packaging.
In the latter case, the most inner (nested) item represents the most outer package item.
For example the case
\--Box
\-----2 blisters
\--------20 tablets
is described as "20 tablets" contained by "a blister"; "2 blisters" contained by one box.
The most inner package represents the Packaged Medicinal Product.
When the IDMP Packaged Medicinal Product ID (PCID) will become actually available for usage, the most inner package <code> element will be used to convey the IDMP PCID.
| (IPS...ent) | | cs | 1 … 1 | F | CONT | | | Example | Packaged Medicinal Product with multiple layers packaging <asContent>
<containerPackagedProduct>
<!-- Inner Package -->
<code codeSystem="..." code="..." displayName="...">
<asContent>
<containerPackagedProduct>
<!-- Intermediate Package -->
<asContent>
<containerPackagedProduct>
<!-- Outer Package / Packaged Medicinal Product -->
</containerPackagedProduct> </asContent> </containerPackagedProduct> </asContent> </code> </containerPackagedProduct></asContent> | | | Example | Packaged Medicinal Product with formCode <asContent>
<containerPackagedProduct>
<!-- Packaged Medicinal Product -->
<code codeSystem="1.999.999" code="PC_ID" displayName="Packaged Product Name">
<name>100 MIRACLE PILLS(TM)</name> <formCode codeSystem="0.4.0.127.0.16.1.1.2.1" code="30009000" displayName="Box" CodeSystemName="EDQM"/> </code> </containerPackagedProduct></asContent> | | PQ | 0 … 1 | | The quantity which specified how many inner packaged content entities are in an outer packaging container entity.
| (IPS...ent) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | Example | <quantity value="20" unit="{tablet}"/> | | | Example | <quantity value="10" unit="mL"/> |  |  |  |  |  | pharm:containerPackagedProduct
|
| | 1 … 1 | R | It represents the most inner Package Item or the Packaged Medicinal Product. | (IPS...ent) | | cs | 1 … 1 | F | CONT | | cs | 1 … 1 | F | KIND | | | 0 … 1 | | If this is also the most outer <pharm:containerPackagedProduct> than the <code> element can be used to convey the (IDMP) Packaged Medicinal Product ID (e.g. the IDMP PCID when it will become actually available for usage).
The presence of the PCID indicates that that element represents the "Packaged Medicinal Product".
| (IPS...ent) | | ST | 0 … 1 | | It represents the Name of the Package Item or of the Packaged Medicinal Product.
If this is also the most outer <pharm:containerPackagedProduct> than this element can be used for the brand name.
| (IPS...ent) | | | Example | <name>AMOXIFEN(R) 20 compresse 20 mg</name> | | CE.IPS | 0 … 1 | R | This element encodes the type of the most inner package item or of the or the Packaged Medicinal Product.
Since the EDQM Standards Terms, together with UCUM, is one of the IDMP terminologies actually available for usage, this code system has been selected as referecne terminology for representing Pharmaceutical Dose forms; Packages and Route of Administration. | (IPS...ent) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.27 Medicine Package (DYNAMIC) |
| | | Example | <formCode code="30007000" codeSystem="0.4.0.127.0.16.1.1.2.1" codeSystemName="EDQM" codeSystemVersion="2010" displayName="Blister">...</formCode> | | PQ | 0 … 1 | | It represents the functional capacity of the container: e.g. bottle containing up to 20 tablets or ampule of 10 ml. | (IPS...ent) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | 0 … * | R | In case of multiple layers of packaging (5 vials 10 ml; box with 2 blisters of 20 tablets) this element can be used for describing the intermediate Packaged Medicinal Product Item or the Packaged Medicinal Product.
For example in the case
\--Box
\-----2 blisters
\--------20 tablets
it describes the "2 blisters"
In the case of
\--Box
\-----5 vials
it represents the Packaged Medicinal Product.
| (IPS...ent) | | cs | 1 … 1 | F | CONT | | PQ | 0 … 1 | R | The quantity which specified how many inner packaged content entities are in an outer packaging container entity.
| (IPS...ent) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | Example | <quantity value="20" unit="{tablet}"/> | | | Example | <quantity value="10" unit="mL"/> |  |  |  |  |  |  |  | pharm:containerPackagedProduct
|
| | 1 … 1 | R | It represents the intermediate Package Item or the Packaged Medicinal Product | (IPS...ent) | | cs | 1 … 1 | F | CONT | | cs | 1 … 1 | F | KIND | | CD.IPS | 0 … 1 | | If this is also the most inner <pharm:containerPackagedProduct> than the <code> element can be used to convey the (IDMP) Packaged Medicinal Product ID (e.g. the IDMP PCID when it will become actually available for usage).
The presence of the PCID indicates that that element represents the "Packaged Medicinal Product".
| (IPS...ent) | | ST | 0 … 1 | R | It represents the Name of the Package Item or of the Packaged Medicinal Product
If this is also the most inner <pharm:containerPackagedProduct> than this element can be used for the brand name.
| (IPS...ent) | | | Example | <name>...</name> | | CE.IPS | 1 … 1 | R | This element encodes the type of the most inner package item or of the or the Packaged Medicinal Product.
Since the EDQM Standards Terms, together with UCUM, is one of the IDMP terminologies actually available for usage, this code system has been selected as reference terminology for representing Pharmaceutical Dose forms; Packages and Route of Administration. | (IPS...ent) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.27 Medicine Package (DYNAMIC) |
| | PQ | 0 … 1 | | It represents the functional capacity of the container: e.g. bottle containing up to 20 tablets or ampule of 10 ml. | (IPS...ent) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | 0 … * | R | In case of multiple layers of packaging (box with 2 blisters of 20 tablets) this element is used for describing the most outer Packaged Medicinal Product Item or the Packaged Medicinal Product.
For example in the case
\--Box
\-----2 blisters
\--------20 tablets
it describes the Packaged Medicinal Product.
| (IPS...ent) | | cs | 1 … 1 | F | CONT | | PQ | 0 … 1 | R | The quantity which specified how many inner packaged content entities are in an outer packaging container entity.
| (IPS...ent) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | Example | <quantity value="20" unit="{tablet}"/> | | | Example | <quantity value="10" unit="mL"/> |  |  |  |  |  |  |  |  |  | pharm:containerPackagedProduct
|
| | 1 … 1 | R | When present, it represents the Packaged Medicinal Product | (IPS...ent) | | cs | 1 … 1 | F | CONT | | cs | 1 … 1 | F | KIND | | CD.IPS | 0 … 1 | | When present, it can be used to convey the (IDMP) Packaged Medicinal Product ID (e.g. the IDMP PCID when it will become actually available for usage). | (IPS...ent) | | ST | 0 … 1 | R | When present, it can be used for the representing the brand name. | (IPS...ent) | | CE.IPS | 1 … 1 | R | When present, it encodes the type of the outer package.
Since the EDQM Standards Terms, together with UCUM, is one of the IDMP terminologies actually available for usage, this code system has been selected as referecne terminology for representing Pharmaceutical Dose forms; Packages and Route of Administration. | (IPS...ent) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.27 Medicine Package (DYNAMIC) |
| | PQ | 0 … 1 | | It represents the functional capacity of the container: e.g. bottle containing up to 20 tablets or ampule of 10 ml. | (IPS...ent) | | cs | 0 … 1 | | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.28 Quantity Units (DYNAMIC) |
| | | 1 … 1 | R | | | | 0 … 1 | R | This module is used for representing the classification of the Substance according to the WHO Anatomical Therapeutic Chemical (ATC) Classification System.
The classCode of "GRIC" identifies this structure as the representation of a generic equivalent of the medication described in the current Medicine entry.
| (IPS...ent) | | where [generalizedMaterialKind/code/@codeSystem='2.16.840.1.113883.6.73'] | | | cs | 1 … 1 | F | GRIC | | | Example | <asSpecializedKind classCode="GRIC">
<generalizedMaterialKind classCode="MMAT">
<!-- Pharmaceutical Substance (ATC Code)-->
<code code=" " codeSystem="2.16.840.1.113883.6.73" displayName=" " codeSystemName="WHO ATC"/> </generalizedMaterialKind></asSpecializedKind> |  |  |  |  |  | pharm:generalizedMaterialKind
|
| | 1 … 1 | M | | (IPS...ent) | | cs | 1 … 1 | F | MMAT | | CD.IPS | 1 … 1 | R | The <code> element contains the ATC code of this medicine. | (IPS...ent) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.29 IPS WHO ATC (DYNAMIC) |
| | | Example | <code codeSystem="2.16.840.1.113883.6.73" code=" " displayName=" " codeSystemName="WHO ATC"/> | | | 0 … * | | | (IPS...ent) | | | 0 … * | R | The Medicinal Product can be classified according to various classification systems, which may be jurisdictional or international.
The classification system itself is specified using an appropriate identification system; the controlled term and the controlled term identifier shall be specified.
When the IDMP Pharmaceutical Product Identifier(s) (PhPID Set) will become actually available for use, the PhPID will be represented by the generalizedMaterialKind/code element. | (IPS...ent) | | cs | 1 … 1 | F | GRIC | | | Example | <asSpecializedKind classCode="GRIC">
<generalizedMaterialKind classCode="MMAT">
<code code="PhPID_Lvl1" codeSystem="1.999.999" displayName="Pharmaceutical Product Name" codeSystemName="PhPID Level 1">
<name/> </code> </generalizedMaterialKind></asSpecializedKind> |  |  |  |  |  | pharm:generalizedMaterialKind
|
| | | R | | (IPS...ent) | | cs | 1 … 1 | F | MMAT | | CD.IPS | 1 … 1 | R | When the IDMP Pharmaceutical Product Identifier(s) (PhPID Set) will become actually available for use, this element will be used for representing the IDMP PhP Id.
The level and the stratum of the PhPID will be distiguished by the OID of the code system.
| (IPS...ent) | | | 0 … * | R | | (IPS...ent) | | | 0 … * | R | This module provides the list of the ingredients (substances with a role) used for this product; one or more ingredients may be present.
The classCode of "ACTI" indicates that this is an active ingredient.
| (IPS...ent) | | cs | 1 … 1 | R | | | | CONF | | The value of @classCode shall be drawn from value set 2.16.840.1.113883.1.11.10430 RoleClassIngredientEntity (DYNAMIC) |
| | | 1 … 1 | M | The medication strength is represented as the ratio of the active ingredient(s) to a unit of medication. The <quantity> element contains the numerator and denominator of the strength ratio.</quantity>
| (IPS...ent) | | | Example | <quantity>...</quantity> | | PQ | 1 … 1 | R | | (IPS...ent) | | cs | 1 … 1 | R | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.30 Medicine Strength Numerator (DYNAMIC) |
| | | 1 … 1 | R | | | PQ | 1 … 1 | R | | (IPS...ent) | | cs | 1 … 1 | R | | | | CONF | | The value of @unit shall be drawn from value set 2.16.840.1.113883.11.22.31 Medicine Strength Denominator (DYNAMIC) |
| | | 1 … 1 | R | |  |  |  |  |  | pharm:ingredientSubstance
|
| | 1 … 1 | R | | (IPS...ent) | | cs | 1 … 1 | F | MMAT | | cs | 1 … 1 | F | KIND | | CD.IPS (extensible) | 0 … 1 | C | The IDMP ISO 11238 standard addresses the identification and exchange of regulated information on substances.
The Global Ingredient Archival System (GInAS) will provide a common global identifier for all of the substances used in medicinal products, providing a definition of substances globally consistent with this standard.
Those identifiers however are yet available for concrete usage, therefore in this version of the template, SNOMED CT has been chosen as reference terminology also for the active substances.
This choice will be revised based on the availability and the maturity of GInAS. | (IPS...ent) | | | CONF | | The value of @code should be drawn from value set 2.16.840.1.113883.11.22.32 IPS Medicine Active Substances (DYNAMIC) |
| | ED | 0 … * | | | (IPS...ent) | | TEL | 0 … * | | | (IPS...ent) | | CD | 0 … * | | This element can be used to provide alternative identifications for the described substance. | (IPS...ent) | | | 0 … 1 | C | Name of the substance | (IPS...ent) | | | Schematron assert | role | error | | | | test | pharm:code or pharm:name | | | | Message | Either the name or the code of the substance (or both) shall be provided | | | Included | 0 … * | | from 2.16.840.1.113883.10.22.4.14 IPS Body Author (DYNAMIC) |  | hl7:author
|
| | 0 … * | | | (IPS...ent) |  |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...ent) | | uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.14 |  |  | hl7:time
|
| TS.IPS.TZ | 1 … 1 | R | | (IPS...ent) |  |  | hl7:assignedAuthor
|
| | 1 … 1 | M | | (IPS...ent) | | II | 1 … * | R | | (IPS...ent) | | | 0 … 1 | R | | (IPS...ent) | | Choice | 0 … 1 | | Elements to choose from:- hl7:assignedPerson
- hl7:assignedAuthoringDevice
| | | 0 … 1 | C | | (IPS...ent) | | cs | 0 … 1 | F | PSN | | cs | 0 … 1 | F | INSTANCE | | PN | 1 … * | R | Name of the person (e.g. the Healthcare Professional) authoring this document | (IPS...ent) | | | Example | <name>
<given>John</given> <family>Español Smith</family></name> | | | 1 … * | R | | (IPS...ent) | | | 1 … * | R | | (IPS...ent) |  |  |  |  | hl7:assignedAuthoringDevice
|
| | 0 … 1 | C | | (IPS...ent) | | | Example | <assignedAuthoringDevice classCode="DEV" determinerCode="INSTANCE">
<softwareName displayName="Turriano"/></assignedAuthoringDevice> | | Included | | | from 2.16.840.1.113883.10.22.9.2 IPS CDA Device (DYNAMIC) | | cs | 0 … 1 | F | DEV | | cs | 0 … 1 | F | INSTANCE | | CE | 0 … 1 | | | (IPS...ent) |  |  |  |  |  | hl7:manufacturerModelName
|
| SC | 0 … 1 | | | (IPS...ent) | | SC | 0 … 1 | | | (IPS...ent) |  |  |  | hl7:representedOrganization
|
| | 0 … 1 | | | (IPS...ent) | | II | 0 … * | | | (IPS...ent) | | | 0 … * | | | (IPS...ent) | | TEL | 0 … * | | | (IPS...ent) | | AD | 0 … * | | | (IPS...ent) |  | hl7:entryRelationship
|
| | 0 … * | C | Subordinate Substance Administration Statement as a component of the overall medication statement.
Unless medications are unknown or known absent, at least one subordinated <substanceAdministration> has to be present to convey information about dosages (dose, frequency of intakes,..).
Subordinated <substanceAdministration> elements can be also used either to handle split dosing, or to support combination medications.
Contains 2.16.840.1.113883.10.21.4.6 UV Subordinate Substance Administration (DYNAMIC) | (IPS...ent) |  |  | @typeCode
|
| cs | 1 … 1 | F | COMP | | | Constraint | At least one subordinate <substanceAdministration> element SHALL be present unless medications are unknown or known absent. </substanceAdministration>
| | | Example | <hl7:entryRelationship typeCode="COMP">
<!-- component: Subordinate Substance Administration Statement. -->
<hl7:substanceAdministration classCode="SBADM" moodCode="EVN">
<hl7:templateId root="2.16.840.1.113883.10.22.4.33"/> <!-- .. -->
</hl7:substanceAdministration> <hl7:sequenceNumber value="1"/></hl7:entryRelationship> |  |  | hl7:sequenceNumber
|
| INT | 0 … 1 | | Sequence number of the Subordinate Substance Administration | (IPS...ent) |
|
IPS ObservationMedia
IPS Pathology Result Observation
| Id | 2.16.840.1.113883.10.22.4.11 | Effective Date | 2024‑08‑04 11:10:42Other versions this id:  IPSPathologyResultObservation as of 2017‑03‑21 IPSPathologyResultObservation as of 2017‑03‑21
|
|---|
| Status |  Draft Draft | Version Label | STU2 |
|---|
| Name | IPSPathologyResultObservation | Display Name | IPS Pathology Result Observation |
|---|
| Description | This template constrains the results of an anatomic pathology observation. The result observation includes a statusCode to allow recording the status of an observation. “Pending” results (e.g., a test has been run but results have not been reported yet) should be repnresented as “active” ActStatus. |
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.11 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Uses | | Uses 2 templates | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.4.14 | Include |  IPS Body Author (STU1) IPS Body Author (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.4.22 | Containment |  IPS Comment Activity (STU1) IPS Comment Activity (STU1) | DYNAMIC |
|
|
|---|
| Relationship | Specialization: template 2.16.840.1.113883.10.22.4.11 IPS Pathology Result Observation (2017‑03‑21)
Adaptation: template 2.16.840.1.113883.10.12.303 CDA Observation (2005‑09‑07) ref ad1bbr-
Adaptation: template 2.16.840.1.113883.10.20.22.4.2 Result Observation (V3) (2015‑08‑01) ref ccda-
Adaptation: template 2.16.840.1.113883.10.22.4.10 IPS Result Observation (2017‑03‑02) |
|---|
| Example | | Example | <observation classCode="OBS" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.11"/> <id root="1.2.3.999" extension="__example only__"/> <code code="44638-5" codeSystem="2.16.840.1.113883.6.1" displayName="Histologic type in Breast tumor by CAP cancer protocols">
<statusCode code="completed"/> <effectiveTime>
<low value="20171210085407"/> </effectiveTime> <value xsi:type="CD" code="399935008" displayName="Ductal carcinoma in situ - category (morphologic abnormality)" codeSystem="2.16.840.1.113883.6.96"/> <methodCode code="104157003" displayName="Light microscopy" codeSystem="2.16.840.1.113883.6.96"/> </code></observation> |
|
|---|
|
IPS Planned Act
IPS Planned Encounter
| Id | 2.16.840.1.113883.10.22.4.40 | Effective Date | 2020‑05‑08 18:18:38 |
|---|
| Status |  Under pre-publication review Under pre-publication review | Version Label | TI-2020 |
|---|
| Name | IPSPlannedEncounter | Display Name | IPS Planned Encounter |
|---|
| Description |
An Encounter is an interaction between a patient and care provider(s) for the purpose of providing healthcare-related service(s). Healthcare services include health assessment. Examples: outpatient visit to multiple departments, home health support (including physical therapy), inpatient hospital stay, emergency room visit, field visit (e.g., traffic accident), office visit,
occupational therapy, or telephone call.
|
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.40 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Relationship | Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.4.14 IHE Encounters (2017‑03‑22 15:35:30) ref IHE-PCC-
Adaptation: template 2.16.840.1.113883.10.20.1.25 (DYNAMIC)
Adaptation: template 2.16.840.1.113883.10.20.1.21 Encounter activity (DYNAMIC) ref ccd1- |
|---|
| Example | | Example | <encounter classCode="ENC" moodCode="cs">
<templateId root="1.3.6.1.4.1.19376.1.5.3.1.4.14"/> <templateId root="2.16.840.1.113883.10.20.1.21"/> <templateId root="2.16.840.1.113883.10.20.1.25"/> <id root="1.2.3.999" extension="--example only--"/> <code/> <text>
<reference value="tel:+1-12345678"/> </text> <effectiveTime>
<low value="20170413114059"/> </effectiveTime> <priorityCode/> <performer>
<time/> </performer> <participant typeCode="LOC">
<participantRole classCode="SDLOC"/> <id/> <code/> <addr/> <telecom/> <playingEntity classCode="PLC">
<name/> </playingEntity> </participant></encounter> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | | | | (IPS...ter) |  | @classCode
|
| cs | 0 … 1 | F | ENC |  | @moodCode
|
| cs | 1 … 1 | R | | | | CONF | | @moodCode shall be "APT" | | or | | @moodCode shall be "ARQ" | | or | | @moodCode shall be "PRP" |
|  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...ter) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.40 |  | hl7:id
|
| II | 0 … * | R | | (IPS...ter) |  | hl7:code
|
| CD | 1 … 1 | R | | (IPS...ter) |  | hl7:text
|
| ED | 0 … 1 | R | The <text> element if present points to the text describing the problem being recorded; including any dates, comments, et cetera. The <reference> contains a URI in value attribute. This URI points to the free text description of the problem in the document that is being described.</reference></text> | (IPS...ter) | | | Example | <text>
<reference value="#problem-1"/></text> |  |  | hl7:reference
|
| TEL | 1 … 1 | M | | (IPS...ter) | | url | 1 … 1 | R | When used it shall refer to the narrative, typically #{label}-{generated-id}, e.g. #xxx-1 |  | hl7:effectiveTime
|
| IVL_TS | 0 … 1 | | | (IPS...ter) |  | hl7:priorityCode
|
| CE | 0 … 1 | | | (IPS...ter) |  | hl7:performer
|
| | 0 … * | | | (IPS...ter) |  |  | hl7:time
|
| | 0 … 1 | | | (IPS...ter) |  | hl7:participant
|
| | 0 … * | | | (IPS...ter) |  |  | @typeCode
|
| cs | 1 … 1 | F | LOC |  |  | hl7:participantRole
|
| | 1 … 1 | M | | (IPS...ter) | | cs | 1 … 1 | F | SDLOC | | | 0 … * | | | (IPS...ter) | | | 0 … 1 | | | (IPS...ter) | | | 0 … * | | | (IPS...ter) | | | 0 … * | | | (IPS...ter) | | | 1 … 1 | M | | (IPS...ter) | | cs | 1 … 1 | F | PLC | | | 1 … * | R | | (IPS...ter) |
|
IPS Planned Immunization
IPS Planned Observation
IPS Planned Procedure
IPS Pregnancy Expected Delivery Date Observation
| Id | 2.16.840.1.113883.10.22.4.29 | Effective Date | 2024‑08‑04 11:02:42Other versions this id:  IPSPregnancyExpectedDeliveryDateObservation as of 2017‑04‑13 IPSPregnancyExpectedDeliveryDateObservation as of 2017‑04‑13
|
|---|
| Status |  Draft Draft | Version Label | STU2 |
|---|
| Name | IPSPregnancyExpectedDeliveryDateObservation | Display Name | IPS Pregnancy Expected Delivery Date Observation |
|---|
| Description | This observation records the Pregnancy Expected Delivery Date for pregnant patients, expressed as a time stamp.
The code reflects the method (operationalisation) of how the date was determined, e.g. clinically estimated, estimated from last menstruation date or last ovulation date.
|
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.29 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Associated with | | Associated with 1 concept | | Id | Name | Data Set |
|---|
| hl7ips-dataelement-213 |  Expected delivery date Expected delivery date |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|
|
|---|
| Relationship | Specialization: template 2.16.840.1.113883.10.22.4.29 IPS Pregnancy Expected Delivery Date Observation (2017‑04‑13)
Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.4.13.5 Pregnancy Observation (2013‑12‑20) ref epsos-
Adaptation: template 2.16.840.1.113883.10.20.1.33 Social history observation (DYNAMIC) ref ccd1- |
|---|
| Example | | Example | <observation typeCode="OBS" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.29"/> <code code="11778-8" codeSystem="2.16.840.1.113883.6.1" displayName="Delivery date estimated (clinical)" codeSystemName="LOINC">
<text>
<reference value="#xxx"/> </text> <statusCode code="completed"/> <effectiveTime value="20160819"/> <value xsi:type="TS" value="20170414"/> </code></observation> |
|
|---|
|
IPS Pregnancy Observation
| Id | 2.16.840.1.113883.10.22.4.36 | Effective Date | 2020‑05‑07 18:36:37 |
|---|
| Status |  Under pre-publication review Under pre-publication review | Version Label | TI-2020 |
|---|
| Name | IPSPregnancyObservation | Display Name | IPS Pregnancy Observation |
|---|
| Description |
A pregnancy observation is a Simple Observation that uses a specific vocabulary to record observations about a patient's current or historical pregnancies.
|
|---|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.36 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Relationship | Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.4.13.5 IHE Pregnancy Observation (2016‑10‑21 14:59:35) ref IHE-PCC-
Adaptation: template 2.16.840.1.113883.10.12.303 CDA Observation (2005‑09‑07) ref ad1bbr-
Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.4.13 IHE Simple Observation (2016‑09‑26 08:43:40) ref IHE-PCC- |
|---|
| Example | | Example | <cda:observation moodCode="EVN">
<cda:templateId root="2.16.840.1.113883.10.22.4.36"/> <cda:code codeSystem="2.16.840.1.113883.6.1"/> <cda:statusCode code="completed"/> <cda:value xsi:type="ANY"/></cda:observation> |
|
|---|
|
IPS Pregnancy Outcome Observation
IPS Pregnancy Status Observation
IPS Problem Concern Entry
| Id | 2.16.840.1.113883.10.22.4.7 | Effective Date | 2021‑08‑04 08:49:27Other versions this id:  IPSProblemConcernEntry as of 2017‑02‑15 IPSProblemConcernEntry as of 2017‑02‑15
|
|---|
| Status |  Draft Draft | Version Label | 2021 |
|---|
| Name | IPSProblemConcernEntry | Display Name | IPS Problem Concern Entry |
|---|
| Description |
This template reflects an ongoing concern on behalf of the provider that placed the concern on a patient’s problem list.
The purpose of the concern act is that of supporting the tracking of a problem or a condition.
There are different kinds of status that could be related to a condition:
- The status of the concern (active, inactive,..)
- The status of the condition (e.g. active, inactive, resolved,..)
- The confirmation status [clinical workflow status, certainty] (e.g. confirmed, likely, unlikely,…)
Not all of them can be represented in a CDA using the statusCode elements of the concern (ACT) and observation (condition).
So long as the underlying conditions are of concern to the provider (i.e., as long as the condition, whether active or resolved, is of ongoing concern and interest to the provider), the statusCode is “active”.
Only when the underlying conditions are no longer of concern is the statusCode set to “completed”.
The effectiveTime reflects the time that the underlying condition was felt to be a concern; it may or may not correspond to the effectiveTime of the condition (e.g., even five years later, the clinician may remain concerned about a prior heart attack).
The effectiveTime/low of the Problem Concern Act asserts when the concern became active. This equates to the time the concern was authored in the patient's chart.
The effectiveTime/high asserts when the concern become inactive, and it is present if the statusCode of the concern act is "completed".
A Problem Concern Act can contain many Problem Observations.
The many Problem Observations nested under a Problem Concern Act reflect the change in the clinical understanding of a condition over time. For instance, a Concern may initially contain a Problem Observation of “chest pain”:
- Problem Concern 1
--- Problem Observation: Chest Pain
Later, a new Problem Observation of “esophagitis” will be added, reflecting a better understanding of the nature of the chest pain. The later problem observation will have a more recent author time stamp.
- Problem Concern 1
--- Problem Observation (author/time Jan 3, 2012): Chest Pain
--- Problem Observation (author/time Jan 6, 2012): Esophagitis
Many systems display the nested Problem Observation with the most recent author time stamp, and provide a mechanism for viewing prior observations.
|
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.7 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Uses | | Uses 1 template | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.4.8 | Containment |  IPS Problem Entry (2021) IPS Problem Entry (2021) | DYNAMIC |
|
|
|---|
| Relationship | Version: template 2.16.840.1.113883.10.22.4.7 IPS Problem Concern Entry (2017‑02‑15)
Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.4.5.1 IHE Concern Entry (DYNAMIC) ref IHE-PCC-
Adaptation: template 2.16.840.1.113883.10.20.1.27 Problem act (DYNAMIC) ref ccd1-
Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.4.5.2 eHDSI Problem Concern (DYNAMIC) ref epsos- |
|---|
| Example | | Active Concern with several conditions | <act classCode="ACT" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.7"/> <id root="1.2.3.999" extension="__example only__"/> <code code="CONC" codeSystem="2.16.840.1.113883.5.6"/> <statusCode code="active"/> <effectiveTime>
<low value="20170309"/> </effectiveTime> <entryRelationship typeCode="SUBJ" inversionInd="false">
<!-- template 2.16.840.1.113883.10.22.4.8 'IPS Problem Entry' (dynamic) -->
<!-- A condition could be active, inactive,.... -->
</entryRelationship> <entryRelationship typeCode="SUBJ" inversionInd="false">
<!-- template 2.16.840.1.113883.10.22.4.8 'IPS Problem Entry' (dynamic) -->
</entryRelationship></act> |
|
|---|
| Example | | Concern no longer tracked | <act classCode="ACT" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.7"/> <id root="1.2.3.999" extension="__example only__"/> <code code="CONC" codeSystem="2.16.840.1.113883.5.6"/> <statusCode code="completed"/> <effectiveTime>
<low value="20161210"/> <low value="20170309"/> </effectiveTime> <entryRelationship typeCode="SUBJ" inversionInd="false">
<!-- template 2.16.840.1.113883.10.22.4.8 'IPS Problem Entry' (dynamic) -->
</entryRelationship></act> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | | R | | (IPS...try) |  | @classCode
|
| cs | 1 … 1 | F | ACT |  | @moodCode
|
| cs | 1 … 1 | F | EVN |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...try) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.7 |  | hl7:id
|
| II | 0 … * | R | | (IPS...try) |  | hl7:code
|
| CD | 1 … 1 | M | | (IPS...try) |  |  | @code
|
| | 1 … 1 | F | CONC |  |  | @codeSystem
|
| | 1 … 1 | F | 2.16.840.1.113883.5.6 |  | hl7:statusCode
|
| CS | 1 … 1 | R | So long as the underlying conditions are of concern to the provider (i.e., as long as the condition, whether active or resolved, is of ongoing concern and interest to the provider), the statusCode is “active”.
Only when the underlying conditions are no longer of concern is the statusCode set to “completed”. | (IPS...try) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.1.11.19890 x_ActStatusActiveComplete (DYNAMIC) |
|  | hl7:effectiveTime
|
| IVL_TS | 1 … 1 | M | | (IPS...try) |  |  | hl7:low
|
| IVXB_TS | 1 … 1 | R | This element asserts when the concern became active. This equates to the time the concern was authored in the patient's chart and the author started tracking this concern. | (IPS...try) |  |  | hl7:high
|
| IVXB_TS | 0 … 1 | C | This element asserts when the clinician deemed there is no longer any need to track the underlying conditions. | (IPS...try) | | | Constraint | If the statusCode is completed this element is required |  | hl7:entryRelationship
|
| | 1 … * | R | Contains 2.16.840.1.113883.10.22.4.8 IPS Problem Entry (DYNAMIC) | (IPS...try) |  |  | @typeCode
|
| cs | 1 … 1 | F | SUBJ |  |  | @inversionInd
|
| bl | 0 … 1 | F | false |
|
IPS Problem Entry
| Id | 2.16.840.1.113883.10.22.4.8 | Effective Date | 2024‑08‑04 11:06:03Other versions this id:  IPSProblemEntry as of 2021‑08‑04 08:52:52 IPSProblemEntry as of 2021‑08‑04 08:52:52 IPSProblemEntry as of 2017‑02‑15 IPSProblemEntry as of 2017‑02‑15
|
|---|
| Status |  Draft Draft | Version Label | 2021 |
|---|
| Name | IPSProblemEntry | Display Name | IPS Problem Entry |
|---|
| Description | This template reflects a discrete observation about a patient's problem. Because it is a discrete observation, it will have a statusCode of "completed". The effectiveTime, also referred to as the “biologically relevant time” is the time at which the observation holds for the patient. For a provider seeing a patient in the
clinic today, observing a history of heart attack that occurred five years ago, the effectiveTime is five years ago.
The effectiveTime of the Problem Observation is the definitive indication of whether or not the underlying condition is resolved. If the problem is known to be resolved, then an effectiveTime/high would be present. If the date of resolution is not known, then effectiveTime/high will be present with a nullFlavor of "UNK".
|
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.8 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Associated with | | Associated with 12 concepts | | Id | Name | Data Set |
|---|
| hl7ips-dataelement-101 |  Problem type Problem type |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-115 |  Problem content status Problem content status |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-127 |  Onset date Onset date |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-128 |  Severity Severity |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-131 |  Diagnosis Diagnosis |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-136 |  Severity Severity |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-140 |  Problem Problem |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-209 |  Health condition / Problem Health condition / Problem |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-210 |  Problem Type Problem Type |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-32 |  Onset Date Onset Date |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-34 |  Date resolved Date resolved |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-36 |  Diagnosis Diagnosis |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|
|
|---|
| Uses | | Uses 3 templates | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.4.25 | Containment |  IPS Severity Observation (STU1) IPS Severity Observation (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.4.19 | Containment |  IPS Certainty Observation (STU1) IPS Certainty Observation (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.4.20 | Containment |  IPS Problem Status Observation (STU1) IPS Problem Status Observation (STU1) | 2017‑03‑29 |
|
|
|---|
| Relationship | Specialization: template 2.16.840.1.113883.10.22.4.8 IPS Problem Entry (2021‑08‑04 08:52:52)
Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.4.5 IHE Problem Entry (DYNAMIC) ref ch-pcc- |
|---|
| Example | | Active Problem | <observation classCode="OBS" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.8"/> <id root="1.2.3.999" extension="__example only__"/> <code code="75326-9" codeSystem="2.16.840.1.113883.6.1" displayName="Problem">
<text>
<reference value="#problem-1"/> </text> <statusCode code="completed"/> <effectiveTime>
<low value="20100507"/> </effectiveTime> <value code="38341003" displayName="Hypertensive disorder, systemic arterial (disorder)" codeSystem="2.16.840.1.113883.6.96"/> <entryRelationship typeCode="SUBJ" inversionInd="true">
<!-- template 2.16.840.1.113883.10.22.4.25 'IPS Severity Observation' (dynamic) -->
</entryRelationship> <entryRelationship typeCode="SUBJ" inversionInd="true">
<!-- template 2.16.840.1.113883.10.22.4.19 'IPS Certainty Observation' (dynamic) -->
</entryRelationship> <entryRelationship typeCode="REFR" inversionInd="false">
<!-- template 2.16.840.1.113883.10.22.4.20 'IPS Problem Status Observation' (2017-03-29T00:00:00) -->
<!-- this referred observation should report that the condition is still active -->
</entryRelationship> </code></observation> |
|
|---|
| Example | | Closed Problem (resolution date known) | <observation classCode="OBS" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.8"/> <id root="1.2.3.999" extension="__example only__"/> <code code="75326-9" codeSystem="2.16.840.1.113883.6.1" displayName="Problem">
<statusCode code="completed"/> <effectiveTime>
<low value="2010"/> <high value="2015"/> </effectiveTime> <value code="38341003" displayName="Hypertensive disorder, systemic arterial (disorder)" codeSystem="2.16.840.1.113883.6.96"/> <entryRelationship typeCode="SUBJ" inversionInd="true">
<!-- template 2.16.840.1.113883.10.22.4.25 'IPS Severity Observation' (dynamic) -->
</entryRelationship> <entryRelationship typeCode="SUBJ" inversionInd="true">
<!-- template 2.16.840.1.113883.10.22.4.19 'IPS Certainty Observation' (dynamic) -->
</entryRelationship> <entryRelationship typeCode="REFR" inversionInd="false">
<!-- template 2.16.840.1.113883.10.22.4.20 'IPS Problem Status Observation' (2017-03-29T00:00:00) -->
<!-- this referred observation should report that the condition is resolved -->
</entryRelationship> </code></observation> |
|
|---|
| Example | | Known absent problems | <observation classCode="OBS" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.8"/> <id root="1.2.3.999" extension="__example only__"/> <code code="75326-9" codeSystem="2.16.840.1.113883.6.1" displayName="Problem">
<statusCode code="completed"/> <effectiveTime>
<low nullFlavor="NI"/> </effectiveTime> <value code="no-known-problems" displayName="No known problems" codeSystem="2.16.840.1.113883.5.1150.1"/> </code></observation> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | | R | | (IPS...try) | | |  | | hl7ips-dataelement-140 |  Problem Problem |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-209 |  Health condition / Problem Health condition / Problem |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  | @classCode
|
| cs | 0 … 1 | F | OBS |  | @moodCode
|
| cs | 1 … 1 | F | EVN |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...try) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.8 |  | hl7:id
|
| II | 0 … * | R | | (IPS...try) |  | hl7:code
|
| CD.IPS | 1 … 1 | R | This element describes the type of condition this observation is referring to. | (IPS...try) | | |  | | hl7ips-dataelement-101 |  Problem type Problem type |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-210 |  Problem Type Problem Type |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.16 IPS Problem Type (DYNAMIC) |
| | | Example | <code code="75326-9" codeSystem="2.16.840.1.113883.6.1" displayName="Problem"/> |  | hl7:text
|
| ED | 0 … 1 | R | The <text> element if present points to the text describing the problem being recorded; including any dates, comments, et cetera. The <reference> contains a URI in value attribute. This URI points to the free text description of the problem in the document that is being described.</reference>
</text>
| (IPS...try) | | | Example | <text>
<reference value="#problem-1"/></text> |  |  | hl7:reference
|
| TEL | 1 … 1 | M | | (IPS...try) | | url | 1 … 1 | R | When used it shall refer to the narrative, typically #{label}-{generated-id}, e.g. #xxx-1 |  | hl7:statusCode
|
| CS | 1 … 1 | M | A clinical document normally records only those condition observation events that have been completed, not observations that are in any other state. Therefore, the <statusCode> shall always have code='completed'.</statusCode>
| (IPS...try) |  |  | @code
|
| CONF | 1 … 1 | F | completed |  | hl7:effectiveTime
|
| IVL_TS | 1 … 1 | M | The effectiveTime, also referred to as the “biologically relevant time” is the time at which the observation holds for the patient. For a provider seeing a patient in the clinic today, observing a history of heart attack that occurred five years ago, the effectiveTime is five years ago.
The <low> and <high> values should be no more precise than known, but as precise as possible.
| (IPS...try) | | | Example | Known onset date (active condition) <effectiveTime>
<low value="20100507"/></effectiveTime> | | | Example | Unknown onset date (active condition) <effectiveTime>
<low nullFlavor="UNK"/></effectiveTime> | | | Example | Unknown resolution date <effectiveTime>
<low value="2010"/> <high nullFlavor="UNK"/></effectiveTime> | | | Example | Known resolution date <effectiveTime>
<low value="201007"/> <high value="201703"/></effectiveTime> |  |  | hl7:low
|
| IVXB_TS | 1 … 1 | R | The effectiveTime/low (a.k.a. "onset date") asserts when the condition became biologically active. | (IPS...try) | | |  | | hl7ips-dataelement-127 |  Onset date Onset date |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-32 |  Onset Date Onset Date |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | hl7:high
|
| IVXB_TS | 0 … 1 | C | The effectiveTime/high (a.k.a. "resolution date") asserts when the condition e became biologically resolved.
If the date of resolution is not known, then effectiveTime/high will be present with a nullFlavor of "UNK". | (IPS...try) | | |  | | hl7ips-dataelement-34 |  Date resolved Date resolved |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | Constraint | If this condition is known to be resolved, then the effectiveTime/high would be present.
|  | hl7:value
|
| CD.IPS (preferred) | 1 … 1 | M | The <value> is the condition that was found.
It may a coded or an un-coded string, but its type is always coded.
The coded form shall be used also to indicate known absent conditions or the nonavailability of information about them. | (IPS...try) | | |  | | hl7ips-dataelement-115 |  Problem content status Problem content status |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-131 |  Diagnosis Diagnosis |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-36 |  Diagnosis Diagnosis |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | @xsi:type
|
| | 0 … 1 | F | CD | | | CONF | | The value of @code comes preferably from value set 2.16.840.1.113883.11.22.73 IPS Problems (DYNAMIC) |
| | | Example | Multiple Coding <value code="302231008" displayName="Salmonella infection" codeSystem="2.16.840.1.113883.6.96">
<translate code="A02.9" displayName="Infezioni da Salmonella non specificate" codeSystem="2.16.840.1.113883.6.3"/></value> | | | Example | Local code not mappable in the reference terminology <value nullFlavor="OTH">
<translate code="12345" displayName="Not in the reference value set" codeSystem="1.2.3.999" codeSystemName="--example only--"/></value> | | | Example | Textual Information <value nullFlavor="NI">
<originalText>
<reference value="#value_as_text"/> </originalText></value> | | | Example | Known absent problems <value code="160245001" displayName="No current problems or disability" codeSystem="2.16.840.1.113883.6.96"/> |  |  | hl7:originalText
|
| ED | | R | The <originalText> element within the <code> element described above is used as follows: the <value> contains a <reference> to the <originalText> in order to link the coded value to the problem narrative text (minus any dates, comments, et cetera). The <reference> contains a
URI in value attribute. This URI points to the free text description of the problem in the document that is being described.
| (IPS...try) | | TEL | 0 … 1 | R | The URI given in the value attribute of the <reference> element points to an element in the narrative content that contains the complete text describing the medication. In a CDA document, the URI given in the value attribute of the <reference> element points to an element in the narrative content that contains
the complete text describing the medication. </reference>
</reference>
| (IPS...try) | | | Example | <reference value="#value_as_text"/> |  |  | hl7:qualifier
|
| CR | 0 … * | | | (IPS...try) |  |  | hl7:translation
|
| CD.IPS | 0 … * | | The translation element may be used to transmit a set of other concept descriptors, using for example ICD-10 or other international or jurisdictional code systems. | (IPS...try) |  | hl7:entryRelationship
|
| | 0 … 1 | R | Severity
The contained entry describes a subjective assessment of the severity of the condition as evaluated by the clinician.
Contains 2.16.840.1.113883.10.22.4.25 IPS Severity Observation (DYNAMIC) | (IPS...try) | | |  | | hl7ips-dataelement-128 |  Severity Severity |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-136 |  Severity Severity |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | @typeCode
|
| cs | 1 … 1 | F | SUBJ |  |  | @inversionInd
|
| bl | 1 … 1 | F | true |  | hl7:entryRelationship
|
| | 0 … 1 | R | Certainty or Verification Status
The contained entry describes the certainty associated with a condition.
Contains 2.16.840.1.113883.10.22.4.19 IPS Certainty Observation (DYNAMIC) | (IPS...try) |  |  | @typeCode
|
| cs | 1 … 1 | F | SUBJ |  |  | @inversionInd
|
| bl | 1 … 1 | F | true |  | hl7:entryRelationship
|
| | 0 … 1 | R | Status of the Problem
The contained entry describes the current status of the condition, for example, whether it is active, in remission, resolved, and so on ...
Contains 2.16.840.1.113883.10.22.4.20 IPS Problem Status Observation (2017‑03‑29) | (IPS...try) |  |  | @typeCode
|
| cs | 1 … 1 | F | REFR |  |  | @inversionInd
|
| bl | 0 … 1 | F | false |
|
IPS Problem Status Observation
| Id | 2.16.840.1.113883.10.22.4.20 | Effective Date | 2021‑09‑02 11:41:28Other versions this id:  IPSProblemStatusObservation as of 2017‑03‑29 IPSProblemStatusObservation as of 2017‑03‑29
|
|---|
| Status |  Draft Draft | Version Label | 2021 |
|---|
| Name | IPSProblemStatusObservation | Display Name | IPS Problem Status Observation |
|---|
| Description | This subordinated observation used by the problem observation records information about the current status of a condition, for example, whether it is active, in remission, resolved, et cetera. |
|---|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.20 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Relationship | Version: template 2.16.840.1.113883.10.22.4.20 IPS Problem Status Observation (2017‑03‑29)
Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.4.1.1 IHE Problem Status Observation (2013‑12‑20) ref IHE-PCC- |
|---|
| Example | | Example | <observation classCode="OBS" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.20"/> <code code="33999-4" codeSystem="2.16.840.1.113883.6.1" displayName="Status"/> <text>
<reference value="#example"/> </text> <statusCode code="completed"/> <value code="active" displayName="Active" codeSystem="2.16.840.1.113883.4.642.3.155"/></observation> |
|
|---|
|
IPS Procedure Entry
| Id | 2.16.840.1.113883.10.22.4.17 | Effective Date | 2024‑08‑04 11:08:35Other versions this id:  IPSProcedureEntry as of 2020‑07‑14 16:35:58 IPSProcedureEntry as of 2020‑07‑14 16:35:58 IPSProcedureEntry as of 2017‑03‑27 IPSProcedureEntry as of 2017‑03‑27
|
|---|
| Status |  Draft Draft | Version Label | STU2 |
|---|
| Name | IPSProcedureEntry | Display Name | IPS Procedure Entry |
|---|
| Description | The procedure entry is used to record procedures that have occurred, or which are planned for in the future. |
|---|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.17 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Associated with | | Associated with 5 concepts | | Id | Name | Data Set |
|---|
| hl7ips-dataelement-216 |  Body site Body site |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-44 |  Procedures Content Status Procedures Content Status |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-47 |  Procedure Procedure |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-48 |  Procedure date Procedure date |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-49 |  Procedure code Procedure code |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|
|
|---|
| Uses | | Uses 1 template | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.4.31 | Containment |  IPS Internal Reference (STU1) IPS Internal Reference (STU1) | DYNAMIC |
|
|
|---|
| Relationship | Specialization: template 2.16.840.1.113883.10.22.4.17 IPS Procedure Entry (2020‑07‑14 16:35:58)
Version: template 2.16.840.1.113883.10.22.4.17 IPS Procedure Entry (2017‑03‑27)
Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.4.19 IHE Procedure Entry (2016‑09‑28 10:37:28) ref IHE-PCC-
Adaptation: template 2.16.840.1.113883.10.12.306 CDA Procedure (2005‑09‑07) ref ad1bbr- |
|---|
| Example | | Example | <hl7:procedure classCode="PROC" moodCode="EVN">
<hl7:templateId root="2.16.840.1.113883.10.22.4.17"/> <hl7:id root="1.2.3.999" extension="--example only--"/> <hl7:code/> <hl7:text>
<hl7:reference value="value"/> </hl7:text> <hl7:statusCode code="completed"/> <hl7:effectiveTime>
<hl7:low value="20200714163551"/> </hl7:effectiveTime> <hl7:targetSiteCode/> <hl7:entryRelationship typeCode="COMP" inversionInd="true">
<!-- template 2.16.840.1.113883.10.22.4.31 'IPS Internal Reference' (2017-05-02T00:00:00) -->
</hl7:entryRelationship></hl7:procedure> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | | | | (IPS...try) | | |  | | hl7ips-dataelement-47 |  Procedure Procedure |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  | @classCode
|
| cs | 1 … 1 | F | PROC |  | @moodCode
|
| cs | 1 … 1 | R | | | | CONF | | The value of @moodCode shall be drawn from value set 2.16.840.1.113883.11.20.9.18 MoodCodeEvnInt (DYNAMIC) |
|  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...try) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.17 |  | hl7:id
|
| II | 0 … * | R | | (IPS...try) |  | hl7:code
|
| CD.IPS (preferred) | 1 … 1 | R | | (IPS...try) | | |  | | hl7ips-dataelement-44 |  Procedures Content Status Procedures Content Status |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-49 |  Procedure code Procedure code |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | CONF | | The value of @code comes preferably from value set 2.16.840.1.113883.11.22.35 IPS Procedures (DYNAMIC) |
|  | hl7:text
|
| ED | 0 … 1 | R | The <text> element if present points to the text describing the data being recorded; including any dates, comments, et cetera. The <reference> contains a URI in value attribute. This URI points to the free text description of the element (problem, procedure,...) in the document that is being described.</reference>
</text>
| (IPS...try) |  |  | hl7:reference
|
| TEL | 1 … 1 | M | | (IPS...try) | | url | 1 … 1 | R | When used it shall refer to the narrative, typically #{label}-{generated-id}, e.g. #xxx-1 |  | hl7:statusCode
|
| CS | 1 … 1 | M | | (IPS...try) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.22 ActStatusActiveCompletedAbortedCancelled (DYNAMIC) |
|  | hl7:effectiveTime
|
| IVL_TS | 1 … 1 | R | | (IPS...try) | | |  | | hl7ips-dataelement-48 |  Procedure date Procedure date |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  | hl7:targetSiteCode
|
| CD.IPS (preferred) | 0 … * | | | (IPS...try) | | |  | | hl7ips-dataelement-216 |  Body site Body site |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | CONF | | The value of @code comes preferably from value set 2.16.840.1.113883.11.22.55 IPS Body Site (DYNAMIC) |
|  | hl7:participant
|
| | 1 … * | R | The device is represented as a participant in the procedure structure. The following descriptions apply to the device structure. | (IPS...try) |  |  | @typeCode
|
| cs | 1 … 1 | F | DEV | | | Example | <participant typeCode="DEV">
<participantRole classCode="MANU">
<id root="1.2.3.999" extension="__example_only__"/> <playingDevice classCode="DEV" determinerCode="INSTANCE">
<code code="" codeSystem="">
<!-- ... -->
</code> </playingDevice> </participantRole></participant> |  |  | hl7:participantRole
|
| | 1 … 1 | R | | (IPS...try) | | cs | 1 … 1 | F | MANU | | II | 0 … * | R | The device ID, e.g. using UDI, is represented by the id element of the participant role. This element is optional, as not all production identifiers (e.g., serial number, lot/batch number, distinct identification number) may be known to the provider or patient.
| (IPS...try) | | | Example | UDI GS1: DeviceIdentifier 00844588003288, Serial# 10987654d321, Lot# 7654321D <id root="2.16.840.1.113883.3.3719" extension="{01}00844588003288{17}141120{10}7654321D{21}10987654d321"/> | | | Example | UDI ICCBBA: DeviceIdentifier 00844588003288 <id root="2.16.840.1.113883.3.3719" extension="A9999XYZ100T0474"/> | | | Example | UDI HIBCC: Serial# XYZ456789012345678, Lot# LOT123456789012345 <id root="2.16.840.1.113883.3.3719" extension="+H123PARTNO1234567890120/$$420020216LOT123456789012345/SXYZ456789012345678/16D20130202C"/> | | | 1 … 1 | R | The playingDevice element describes the device instance. | (IPS...try) | | cs | 1 … 1 | F | DEV | | cs | 1 … 1 | F | INSTANCE | | CE.IPS (preferred) | 1 … 1 | R | The device code describes the type of device (e.g. arm prosthesis, arterial stent). | (IPS...try) | | | CONF | | The value of @code comes preferably from value set 2.16.840.1.113883.11.22.23 IPS Medical Devices (DYNAMIC) | | or | | The value of @code comes preferably from value set 2.16.840.1.113883.11.22.61 Absent or Unknown Devices (DYNAMIC) |
|  | hl7:entryRelationship
|
| | 0 … * | | Contains 2.16.840.1.113883.10.22.4.31 IPS Internal Reference (DYNAMIC) | (IPS...try) |  |  | @typeCode
|
| cs | 1 … 1 | F | COMP |  |  | @inversionInd
|
| bl | 1 … 1 | F | true |
|
IPS Radiology Result Observation
IPS Radiology Result Observation Component
| Id | 2.16.840.1.113883.10.22.17 | Effective Date | 2024‑08‑04 20:18:47 |
|---|
| Status |  Draft Draft | Version Label | STU2 |
|---|
| Name | IPSRadiologyResultObservationComponent | Display Name | IPS Radiology Result Observation Component |
|---|
| Description | IPS Radiology Result Observation Component The component carries additional detailed radiological results. Depending on it's type the code and value have different constraints to reflect proper results. Type of result | Constraints for code (Value Set) | Value Domain value type | Observation Text | IPS Results Radiology Textual Observations | String | Observation Code | IPS Results Radiology Observation | Code | Observation Measurement, including linear, area and
volume quantity measurements | IPS Results Radiology Measurement Observation | Quantity | Observation Measurement, including linear, area and volume range measurements | IPS Results Radiology Measurement Observation | Interval of quantity | Observation Measurement,
including linear, area and volume ratio measurements | IPS Results Radiology Measurement Observation | Ratio of quantity | Observation Measurement, including linear, area and volume sampled data measurements | IPS Results Radiology Measurement Observation | Sample Data
(Binary) |
|
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.17 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Relationship | Specialization: template 2.16.840.1.113883.10.12.303 CDA Observation (2005‑09‑07) ref ad1bbr- |
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | | | | (IPS...ent) |  | @classCode
|
| cs | 1 … 1 | F | OBS |  | @moodCode
|
| cs | 1 … 1 | R | | | | CONF | | The value of @moodCode shall be drawn from value set 2.16.840.1.113883.1.11.18943 x_ActMoodDocumentObservation (DYNAMIC) |
|  | hl7:templateId
|
| II | 1 … 1 | R | | (IPS...ent) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.12.303 |  | hl7:id
|
| II | 0 … * | | | (IPS...ent) | | Choice | 1 … 1 | | Elements to choose from:- hl7:code
- hl7:code[concat(@code, @codeSystem) = doc('include/voc-2.16.840.1.113883.11.22.66-DYNAMIC.xml')//valueSet[1]/conceptList/concept/concat(@code, @codeSystem) or @nullFlavor]
- hl7:code[concat(@code, @codeSystem) = doc('include/voc-2.16.840.1.113883.11.22.80-DYNAMIC.xml')//valueSet[1]/conceptList/concept/concat(@code, @codeSystem) or @nullFlavor]
|  |  | hl7:code
|
| CE | 0 … 1 | R | | (IPS...ent) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.40 IPS Results Radiology Observation (DYNAMIC) |
|  |  | hl7:code
|
| CE | 0 … 1 | R | | (IPS...ent) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.66 IPS Results Radiology Textual Observations (DYNAMIC) |
|  |  | hl7:code
|
| CE | 0 … 1 | R | Codes identifying numeric measurements | (IPS...ent) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.80 IPS Results Radiology Measurement Observation (DYNAMIC) |
|  | hl7:text
|
| ED | 0 … 1 | | | (IPS...ent) |  | hl7:statusCode
|
| CS | 0 … 1 | | | (IPS...ent) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.1.11.15933 ActStatus (DYNAMIC) |
|  | hl7:effectiveTime
|
| IVL_TS | 0 … 1 | | | (IPS...ent) | | Choice | 1 … 1 | | Elements to choose from:- hl7:value[@xsi:type='ST']
- hl7:value[@xsi:type='CE.IPS']
- hl7:value[@xsi:type='PQ']
- hl7:value[@xsi:type='IVL_PQ']
- hl7:value[@xsi:type='RTO_QTY_QTY']
- hl7:value[@xsi:type='BIN']
|  |  | hl7:value
|
| ST | 0 … 1 | R | Observation Text | (IPS...ent) | | where [@xsi:type='ST'] | |  |  | hl7:value
|
| CE.IPS | 0 … 1 | R | Observation Code | (IPS...ent) | | where [@xsi:type='CE.IPS'] | |  |  | hl7:value
|
| PQ | 0 … 1 | R | Observation Measurement, including linear, area and volume quantity measurements | (IPS...ent) | | where [@xsi:type='PQ'] | | | | Constraint | If Observation/value is a physical quantity (xsi:type="PQ"), the unit of measure SHALL be selected from ValueSet UnitsOfMeasureCaseSensitive 2.16.840.1.113883.1.11.12839 DYNAMIC |  |  | hl7:value
|
| IVL_PQ | 0 … 1 | R | Observation Measurement, including linear, area and volume range measurements | (IPS...ent) | | where [@xsi:type='IVL_PQ'] | | | | Constraint | If Observation/value is a physical quantity, the unit of measure SHALL be selected from ValueSet UnitsOfMeasureCaseSensitive 2.16.840.1.113883.1.11.12839 DYNAMIC |  |  | hl7:value
|
| RTO_QTY_QTY | 0 … 1 | R | Observation Measurement, including linear, area and volume ratio measurements | (IPS...ent) | | where [@xsi:type='RTO_QTY_QTY'] | | | | Constraint | If Observation/value is a physical quantity, the unit of measure SHALL be selected from ValueSet UnitsOfMeasureCaseSensitive 2.16.840.1.113883.1.11.12839 DYNAMIC |  |  | hl7:value
|
| BIN | 0 … 1 | R | Observation Measurement, including linear, area and volume sampled data measurements | (IPS...ent) | | where [@xsi:type='BIN'] | |
|
IPS Reaction Manifestation
| Id | 2.16.840.1.113883.10.22.4.6 | Effective Date | 2024‑08‑04 10:18:47Other versions this id:  IPSAdverseReactionMFST as of 2016‑11‑15 IPSAdverseReactionMFST as of 2016‑11‑15
|
|---|
| Status |  Draft Draft | Version Label | STU2 |
|---|
| Name | IPSAdverseReactionMFST | Display Name | IPS Reaction Manifestation |
|---|
| Description | This clinical statement represents the response to an undesired symptom, finding, etc. due to administered or exposed substance. This reaction may be an undesired symptom, finding, etc. or it could be a desired response to a treatment. A reaction can be defined with respect to its severity, and can have been treated by one or
more interventions. |
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.6 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Associated with | | Associated with 2 concepts | | Id | Name | Data Set |
|---|
| hl7ips-dataelement-193 |  Manifestation of the reaction Manifestation of the reaction |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-194 |  Severity Severity |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|
|
|---|
| Uses | | Uses 1 template | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.22.4.25 | Containment |  IPS Severity Observation (STU1) IPS Severity Observation (STU1) | DYNAMIC |
|
|
|---|
| Relationship | Specialization: template 2.16.840.1.113883.10.22.4.6 IPS Reaction Manifestation (2016‑11‑15)
Adaptation: template 1.3.6.1.4.1.19376.1.5.3.1.4.5 IHE Problem Entry (DYNAMIC) ref ch-pcc- |
|---|
| Example | | Example | <observation classCode="OBS" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.6"/> <id root="1.2.3.999" extension="__example only__"/> <code code="404684003" displayName="Clinical finding" codeSystem="2.16.840.1.113883.6.96">
<text>
<reference value="#ref1"/> </text> <statusCode code="completed"/> <effectiveTime>
<low value="201611"/> </effectiveTime> <value xsi:type="CD" code="1985008" displayName="Vomitus" codeSystem="2.16.840.1.113883.6.96">
<originalText>
<reference value="#ref2"/> </originalText> </value> <entryRelationship typeCode="SUBJ" inversionInd="true">
<!-- template 2.16.840.1.113883.10.22.4.25 'IPS Severity Observation' (dynamic) -->
</entryRelationship> </code></observation> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | | R | | (IPS...FST) |  | @classCode
|
| cs | 0 … 1 | F | OBS |  | @moodCode
|
| cs | 1 … 1 | F | EVN |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...FST) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.6 |  | hl7:id
|
| II | 0 … * | R | | (IPS...FST) |  | hl7:code
|
| CD.IPS | 1 … 1 | R | This element describes the type of condition this observation is referring to.
| (IPS...FST) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.16 IPS Problem Type (2017‑04‑07) |
|  | hl7:text
|
| ED | 0 … 1 | R | The <text> element if present points to the text describing the problem being recorded; including any dates, comments, et cetera. The <reference> contains a URI in value attribute. This URI points to the free text description of the problem in the document that is being described.
| (IPS...FST) |  |  | hl7:reference
|
| TEL | 1 … 1 | M | | (IPS...FST) | | url | 1 … 1 | R | When used it shall refer to the narrative, typically #{label}-{generated-id}, e.g. #xxx-1 |  | hl7:statusCode
|
| CS (required) | 0 … 1 | R | A clinical document normally records only those condition observation events that have been completed, not observations that are in any other state. Therefore, the <statusCode> shall always have code='completed'.
| (IPS...FST) |  |  | @code
|
| CONF | 0 … 1 | F | completed |  | hl7:effectiveTime
|
| IVL_TS | 1 … 1 | M | The effectiveTime, also referred to as the “biologically relevant time” is the time at which the observation holds for the patient. For a provider seeing a patient in the clinic today, observing a history of heart attack that occurred five years ago, the effectiveTime is five years ago.
The <low> and <high> values should be no more precise than known, but as precise as possible.
| (IPS...FST) |  |  | hl7:low
|
| IVXB_TS | 1 … 1 | R | The effectiveTime/low (a.k.a. "onset date") asserts when the condition became biologically active. | (IPS...FST) |  |  | hl7:high
|
| IVXB_TS | 0 … 1 | C | The effectiveTime/high (a.k.a. "resolution date") asserts when the condition e became biologically resolved.
If the date of resolution is not known, then effectiveTime/high will be present with a nullFlavor of "UNK". | (IPS...FST) | | | Constraint | If this condition is known to be resolved, then the effectiveTime/high would be present.
|  | hl7:value
|
| CD.IPS (preferred) | 1 … 1 | R | The <value> is the condition that was found. While the value may be a coded or an un-coded string, the type is always a coded value.
If uncoded, it shall contain a <reference> to the <originalText> in the narrative where the reaction is described.
| (IPS...FST) | | |  | | hl7ips-dataelement-193 |  Manifestation of the reaction Manifestation of the reaction |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | @xsi:type
|
| | 1 … 1 | F | CD | | | CONF | | The value of @code comes preferably from value set 2.16.840.1.113883.11.22.3 IPS Allergy Reaction (DYNAMIC) |
|  |  | hl7:originalText
|
| | 0 … 1 | R | The <originalText> element within the <code> element described above is used as follows: the <value> contains a <reference> to the <originalText> in order to link the coded value to the problem narrative text (minus any dates, comments, et cetera). The <reference> contains a
URI in value attribute. This URI points to the free text description of the problem in the document that is being described.
| (IPS...FST) | | | 0 … 1 | R | The URI given in the value attribute of the <reference> element points to an element in the narrative content that contains the complete text describing the medication. In a CDA document, the URI given in the value attribute of the <reference> element points to an element in the narrative content that contains
the complete text describing the medication. </reference></reference> | (IPS...FST) | | | Example | <reference value="#AdvReaction_1"/> |  |  | hl7:translation
|
| CD | 0 … * | | | (IPS...FST) |  | hl7:entryRelationship
|
| | 0 … 1 | R | Severity
The contained entry describes a subjective assessment of the severity of the condition as evaluated by the clinician.
Contains 2.16.840.1.113883.10.22.4.25 IPS Severity Observation (DYNAMIC) | (IPS...FST) | | |  | | hl7ips-dataelement-194 |  Severity Severity |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | @typeCode
|
| cs | 1 … 1 | F | SUBJ |  |  | @inversionInd
|
| bl | 1 … 1 | F | true |
|
IPS Result Observation
IPS Result Organizer
| Id | 2.16.840.1.113883.10.22.4.9 | Effective Date | 2017‑03‑02 |
|---|
| Status |  Under pre-publication review Under pre-publication review | Version Label | STU1 |
|---|
| Name | IPSResultOrganizer | Display Name | IPS Result Organizer |
|---|
| Description | This template provides a mechanism for grouping result observations. It contains information applicable to all of the contained result observations. The Result Organizer code categorizes the contained results into one of several commonly accepted values (e.g., “Hematology”, “Chemistry”, “Nuclear Medicine”). If any Result Observation within the organizer has a statusCode of "active", the Result Organizer must also have a statusCode of "active". However, the results selected for a patient summary are most often final results, with status "completed". So in most cases, the statusCode of the Organizer is "completed". The result observations contained within the organizer may use
either of these templates: - Laboratory Result Observation
- Radiology Result Observation
- Pathology Result Observation
- Result Observation (most generic template used whenever none of the above is applicable)
One Result Organizer entry groups results, which have a common context of production: - common specialty (imaging, bacteriology, serology, chemistry, surgical pathology, clinical, radiology ...),
- common overall interpretation, (which interprets the set of results of the Organizer),
- common biologic specimen for in vitro diagnostic observations,
- common associated illustrative
image (ObservationMedia).
The ultimate choice for sorting out results between Organizer entries belongs to the authoring person or system of the section. |
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.9 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Associated with | | Associated with 6 concepts | | Id | Name | Data Set |
|---|
| hl7ips-dataelement-106 |  Observation Result Observation Result |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-143 |  Date of observation Date of observation |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-144 |  Observation Type Observation Type |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-146 |  Performer Performer |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-177 |  Observer Observer |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 | | hl7ips-dataelement-23 |  Observation Result Observation Result |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|
|
|---|
| Uses | | Uses 9 templates | | Uses | as | Name | Version |
|---|
| 2.16.840.1.113883.10.12.323 | Containment |  CDA Performer (Body) CDA Performer (Body) | DYNAMIC | | 2.16.840.1.113883.10.22.4.14 | Include |  IPS Body Author (STU1) IPS Body Author (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.4.13 | Containment |  IPS Laboratory Result Observation (STU2) IPS Laboratory Result Observation (STU2) | DYNAMIC | | 2.16.840.1.113883.10.22.4.12 | Containment |  IPS Radiology Result Observation (STU2) IPS Radiology Result Observation (STU2) | DYNAMIC | | 2.16.840.1.113883.10.22.4.11 | Containment |  IPS Pathology Result Observation (STU2) IPS Pathology Result Observation (STU2) | DYNAMIC | | 2.16.840.1.113883.10.22.4.10 | Containment |  IPS Result Observation (STU1) IPS Result Observation (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.4.30 | Containment |  IPS Specimen Collection (STU2) IPS Specimen Collection (STU2) | DYNAMIC | | 2.16.840.1.113883.10.22.4.22 | Containment |  IPS Comment Activity (STU1) IPS Comment Activity (STU1) | DYNAMIC | | 2.16.840.1.113883.10.22.4.23 | Containment |  IPS ObservationMedia (2021) IPS ObservationMedia (2021) | DYNAMIC |
|
|
|---|
| Relationship | Adaptation: template 2.16.840.1.113883.10.12.305 CDA Organizer (2005‑09‑07) ref ad1bbr-
Adaptation: template 2.16.840.1.113883.10.20.22.4.1 Result Organizer (V3) (2015‑08‑01) ref ccda- |
|---|
| Example | | Example | <organizer classCode="BATTERY" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.9"/> <code code="11529-5" displayName="Surgical pathology studies (set)" codeSystemName="LOINC" codeSystem="2.16.840.1.113883.6.1"/> <satusCode code="completed"/> <author>
<!-- template 2.16.840.1.113883.10.12.318 'CDA Author (Body)' - a pathologist -->
</author> <component>
<!-- template 2.16.840.1.113883.10.22.4.11 'IPS Pathology Result Observation' -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.30 'IPS Specimen Collection' - excised tissue specimen -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.22 'IPS Comment Activity' - pathologist's interpretation -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.23 'IPS ObservationMedia' - an illustrative slide image -->
</component></organizer> |
|
|---|
| Example | | Example | <organizer classCode="BATTERY" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.9"/> <code code="18719-5" displayName="Chemistry studies (set)" codeSystemName="LOINC" codeSystem="2.16.840.1.113883.6.1"/> <satusCode code="completed"/> <author>
<!-- template 2.16.840.1.113883.10.12.318 'CDA Author (Body)' - a clinical laboratory director -->
</author> <component>
<!-- template 2.16.840.1.113883.10.22.4.13 'IPS Laboratory Result Observation' -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.13 'IPS Laboratory Result Observation' -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.30 'IPS Specimen Collection' - common blood serum specimen -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.22 'IPS Comment Activity' - interpretation of chemistry results -->
</component></organizer> |
|
|---|
| Example | | Example | <organizer classCode="BATTERY" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.22.4.9"/> <code code="18748-4" displayName="Diagnostic imaging study" codeSystemName="LOINC" codeSystem="2.16.840.1.113883.6.1"/> <satusCode code="completed"/> <author>
<!-- template 2.16.840.1.113883.10.12.318 'CDA Author (Body)' - a radiologist -->
</author> <component>
<!-- template 2.16.840.1.113883.10.22.4.12 'IPS Radiology Result Observation' -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.23 'IPS ObservationMedia' - an illustrative image -->
</component> <component>
<!-- template 2.16.840.1.113883.10.22.4.22 'IPS Comment Activity' - overall interpretation -->
</component></organizer> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | | | | (IPS...zer) | | |  | | hl7ips-dataelement-106 |  Observation Result Observation Result |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  | @classCode
|
| cs | 1 … 1 | R | |  | @moodCode
|
| cs | 1 … 1 | F | EVN |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...zer) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.9 |  | hl7:id
|
| II | 0 … * | | | (IPS...zer) |  | hl7:code
|
| CD.IPS | 1 … 1 | R | | (IPS...zer) | | |  | | hl7ips-dataelement-144 |  Observation Type Observation Type |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.22.37 IPS Results Organizer (DYNAMIC) |
|  | hl7:statusCode
|
| CS | 1 … 1 | M | | (IPS...zer) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.1.11.19890 x_ActStatusActiveComplete (DYNAMIC) |
|  | hl7:effectiveTime
|
| IVL_TS | 0 … 1 | | | (IPS...zer) | | |  | | hl7ips-dataelement-143 |  Date of observation Date of observation |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | hl7:low
|
| IVXB_TS | 0 … 1 | | | (IPS...zer) |  |  | hl7:high
|
| IVXB_TS | 0 … 1 | | | (IPS...zer) |  | hl7:performer
|
| | 0 … * | R | When present, this element represents the organization who performed the set of observations grouped under this organizer.
Contains 2.16.840.1.113883.10.12.323 CDA Performer (Body) (DYNAMIC) | (IPS...zer) | | |  | | hl7ips-dataelement-146 |  Performer Performer |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
| | Included | 0 … * | R | from 2.16.840.1.113883.10.22.4.14 IPS Body Author (DYNAMIC) | | |  | | hl7ips-dataelement-177 |  Observer Observer |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  | hl7:author
|
| | 0 … * | R | | (IPS...zer) |  |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...zer) | | uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.14 |  |  | hl7:time
|
| TS.IPS.TZ | 1 … 1 | R | | (IPS...zer) |  |  | hl7:assignedAuthor
|
| | 1 … 1 | M | | (IPS...zer) | | II | 1 … * | R | | (IPS...zer) | | | 0 … 1 | R | | (IPS...zer) | | Choice | 0 … 1 | | Elements to choose from:- hl7:assignedPerson
- hl7:assignedAuthoringDevice
| | | 0 … 1 | C | | (IPS...zer) | | cs | 0 … 1 | F | PSN | | cs | 0 … 1 | F | INSTANCE | | PN | 1 … * | R | Name of the person (e.g. the Healthcare Professional) authoring this document | (IPS...zer) | | | Example | <name>
<given>John</given> <family>Español Smith</family></name> | | | 1 … * | R | | (IPS...zer) | | | 1 … * | R | | (IPS...zer) |  |  |  |  | hl7:assignedAuthoringDevice
|
| | 0 … 1 | C | | (IPS...zer) | | | Example | <assignedAuthoringDevice classCode="DEV" determinerCode="INSTANCE">
<softwareName displayName="Turriano"/></assignedAuthoringDevice> | | Included | | | from 2.16.840.1.113883.10.22.9.2 IPS CDA Device (DYNAMIC) | | cs | 0 … 1 | F | DEV | | cs | 0 … 1 | F | INSTANCE | | CE | 0 … 1 | | | (IPS...zer) |  |  |  |  |  | hl7:manufacturerModelName
|
| SC | 0 … 1 | | | (IPS...zer) | | SC | 0 … 1 | | | (IPS...zer) |  |  |  | hl7:representedOrganization
|
| | 0 … 1 | | | (IPS...zer) | | II | 0 … * | | | (IPS...zer) | | | 0 … * | | | (IPS...zer) | | TEL | 0 … * | | | (IPS...zer) | | AD | 0 … * | | | (IPS...zer) | | Choice | 1 … * | | Elements to choose from:- hl7:component containing template 2.16.840.1.113883.10.22.4.13 IPS Laboratory Result Observation (DYNAMIC)
- hl7:component containing template 2.16.840.1.113883.10.22.4.12 IPS Radiology Result Observation (DYNAMIC)
- hl7:component containing template 2.16.840.1.113883.10.22.4.11 IPS Pathology Result Observation (DYNAMIC)
- hl7:component containing template 2.16.840.1.113883.10.22.4.10 IPS Result Observation (DYNAMIC)
- hl7:component containing template 2.16.840.1.113883.10.22.4.30 IPS Specimen Collection (DYNAMIC)
- hl7:component containing template 2.16.840.1.113883.10.22.4.22 IPS Comment Activity (DYNAMIC)
- hl7:component containing template 2.16.840.1.113883.10.22.4.23 IPS ObservationMedia (DYNAMIC)
|  |  | hl7:component
|
| | 0 … * | | Contains 2.16.840.1.113883.10.22.4.13 IPS Laboratory Result Observation (DYNAMIC) | (IPS...zer) |  |  | hl7:component
|
| | 0 … * | | Contains 2.16.840.1.113883.10.22.4.12 IPS Radiology Result Observation (DYNAMIC) | (IPS...zer) |  |  | hl7:component
|
| | 0 … * | | Contains 2.16.840.1.113883.10.22.4.11 IPS Pathology Result Observation (DYNAMIC) | (IPS...zer) |  |  | hl7:component
|
| | 0 … * | | Contains 2.16.840.1.113883.10.22.4.10 IPS Result Observation (DYNAMIC) | (IPS...zer) | | |  | | hl7ips-dataelement-23 |  Observation Result Observation Result |  CEN/TC 251 prEN 17269 CEN/TC 251 prEN 17269 |
|  |  | hl7:component
|
| | 0 … * | | Contains 2.16.840.1.113883.10.22.4.30 IPS Specimen Collection (DYNAMIC) | (IPS...zer) | | | Constraint | An IPS Specimen Collection SHALL be present only if the organizer carries anatomic pathology or microbiology laboratory observations, which need to be associated with the specific anatomic site the specimen was collected from. |  |  | hl7:component
|
| | 0 … * | | Contains 2.16.840.1.113883.10.22.4.22 IPS Comment Activity (DYNAMIC) | (IPS...zer) | | | Constraint | An IPS Comment Activity SHALL contain a general comment applying to the whole set of observations present in the organizer |  |  | hl7:component
|
| | 0 … * | | Contains 2.16.840.1.113883.10.22.4.23 IPS ObservationMedia (DYNAMIC) | (IPS...zer) |
|
IPS Severity Observation
IPS Social History Alcohol Use
IPS Social History Observation
| Id | 2.16.840.1.113883.10.22.4.48 | Effective Date | 2020‑05‑10 15:16:16 |
|---|
| Status |  Under pre-publication review Under pre-publication review | Version Label | TI-2020 |
|---|
| Name | IPSSocialHistoryObservation | Display Name | IPS Social History Observation |
|---|
| Description | This template represents a patient's occupations, lifestyle, and environmental health risk factors. Demographic data (e.g., marital status, race, ethnicity, religious affiliation) are captured in the header. Though tobacco use and exposure may be represented with a Social History Observation, it is recommended to use the Current Smoking Status template or the
Tobacco Use template instead, to represent smoking or tobacco habits. |
|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.22.4.48 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Relationship | Adaptation: template 2.16.840.1.113883.10.20.22.4.38 Social History Observation (V3) (2015‑08‑01) ref ccda-
Adaptation: template 2.16.840.1.113883.10.12.303 CDA Observation (2005‑09‑07) ref ad1bbr-
Version: template 2.16.840.1.113883.10.20.22.4.38 Social History Observation (2013‑01‑31) ref ccda- |
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
| | | | | (IPS...ion) |  | @classCode
|
| cs | 1 … 1 | F | OBS |  | @moodCode
|
| cs | 1 … 1 | F | EVN |  | hl7:templateId
|
| II | 1 … 1 | M | | (IPS...ion) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.22.4.48 |  | hl7:id
|
| II | 0 … * | | | (IPS...ion) |  | hl7:code
|
| CD.IPS (example) | 1 … 1 | R | | (IPS...ion) | | | CONF | | Examples of the value of @code are in the value set 2.16.840.1.113883.3.88.12.80.60 Social History Type (2008‑12‑18) |
|  | hl7:text
|
| ED | 0 … 1 | R | The <text> element if present points to the text describing the problem being recorded; including any dates, comments, et cetera. The <reference> contains a URI in value attribute. This URI points to the free text description of the problem in the document that is being described.</reference></text> | (IPS...ion) | | | Example | <text>
<reference value="#problem-1"/></text> |  |  | hl7:reference
|
| TEL | 1 … 1 | M | | (IPS...ion) | | url | 1 … 1 | R | When used it shall refer to the narrative, typically #{label}-{generated-id}, e.g. #xxx-1 |  | hl7:statusCode
|
| CS | 1 … 1 | M | | (IPS...ion) |  |  | @code
|
| CONF | 1 … 1 | F | completed |  | hl7:effectiveTime
|
| IVL_TS | 0 … 1 | | | (IPS...ion) |  | hl7:value
|
| | 0 … 1 | R | | (IPS...ion) | | | Constraint | If Observation/value is a physical quantity (xsi:type="PQ"), the unit of measure *SHALL* be selected from ValueSet UnitsOfMeasureCaseSensitive (2.16.840.1.113883.1.11.12839) *DYNAMIC* |
|
IPS Social History Tobacco Use
IPS Specimen Collection
IPS Survey Observation
IPS Survey Panel
UV Subordinate Substance Administration
| Id | 2.16.840.1.113883.10.21.4.6 | Effective Date | 2023‑01‑30 09:36:00Other versions this id:  UVSubordinateadministration as of 2017‑04‑30 UVSubordinateadministration as of 2017‑04‑30
|
|---|
| Status |  Draft Draft | Version Label | 2023 |
|---|
| Name | UVSubordinateadministration | Display Name | UV Subordinate Substance Administration |
|---|
| Description | Universal Subordinate Substance Administration to convey information about dosages |
|---|
| Context | Parent nodes of template element with id 2.16.840.1.113883.10.21.4.6 |
|---|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
| Relationship | Version: template 2.16.840.1.113883.10.21.4.6 UV Subordinate Substance Administration (2017‑04‑30)
Specialization: template 2.16.840.1.113883.10.12.308 CDA SubstanceAdministration (2005‑09‑07) ref ad1bbr- |
|---|
| Example | | Example | <substanceAdministration classCode="SBADM" moodCode="EVN">
<templateId root="2.16.840.1.113883.10.21.4.6"/> <statusCode code="active"/> <effectiveTime xsi:type="PIVL_TS" institutionSpecified="true">
<period value="12" unit="h"/> </effectiveTime> <doseQuantity xsi:type="IVL_PQ" value="2" unit="{puff}"/> <consumable>
<manufacturedProduct>
<manufacturedMaterial nullFlavor="NA"/> </manufacturedProduct> </consumable></substanceAdministration> |
|
|---|
| Item | DT | Card | Conf | Description | Label |
|---|
hl7:substanceAdministration
|
| | 1 … 1 | R | | (UVS...ion) |  | @classCode
|
| cs | 1 … 1 | F | SBADM |  | @moodCode
|
| cs | 1 … 1 | R | If the subordinate substance administration refers to Medication Order then a substance administration request (moodCode is 'RQO') is used.
If it refers to a Medication Statement, the moodCode shall be set to event/intent (moodCode is 'EVN' or 'INT'). | | | CONF | | The value of @moodCode shall be drawn from value set 2.16.840.1.113883.11.21.4 Mood Code Evn Int Rqo (DYNAMIC) |
| | | Constraint | The moodCode of this subordinate substance administration SHALL be the same of the parent substance administration |  | hl7:templateId
|
| II | 1 … 1 | M | | (UVS...ion) |  |  | @root
|
| uid | 1 … 1 | F | 2.16.840.1.113883.10.21.4.6 |  | hl7:statusCode
|
| CS | 1 … 1 | M | | (UVS...ion) | | | Constraint | The statusCode of this subordinate substance administration SHALL be the same of that of the parent substance administration. | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.11.21.2 ActStatusActiveCompletedAbortedSuspended (DYNAMIC) |
| | Choice | 1 … 1 | | Elements to choose from:- hl7:effectiveTime[@value or @nullFlavor]
- hl7:effectiveTime[@xsi:type='PIVL_TS']
- hl7:effectiveTime[@xsi:type='EIVL_TS']
- hl7:effectiveTime[@xsi:type='SXPR_TS']
|  |  | hl7:effectiveTime
|
| TS | 0 … 1 | C | This required element describes the frequency of intakes. If not known it shall be valued with the nullflavor "UNK" | (UVS...ion) | | where [@value or @nullFlavor] | | | | Example | Once (known date) <effectiveTime value="20170404"/> | | | Example | Unknown <effectiveTime nullFlavor="UNK"/> |  |  | hl7:effectiveTime
|
| PIVL_TS | 0 … 1 | C | Periodic Time Interval | (UVS...ion) | | where [@xsi:type='PIVL_TS'] | | | | Example | Every 4 hours <effectiveTime xsi:type="PIVL_TS" institutionSpecified="false">
<period value="4" unit="h"/></effectiveTime> | | | Example | Twice a day <effectiveTime xsi:type="PIVL_TS" institutionSpecified="true">
<period value="12" unit="h"/></effectiveTime> |  |  | hl7:effectiveTime
|
| EIVL_TS | 0 … 1 | C | Event Related Time Interval | (UVS...ion) | | where [@xsi:type='EIVL_TS'] | | | | Example | After meal <effectiveTime xsi:type="EIVL_TS">
<event code="PC" codeSystem="2.16.840.1.113883.5.139"/></effectiveTime> | | | Example | One hour before breakfast <effectiveTime xsi:type="EIVL_TS">
<event code="ACM" codeSystem="2.16.840.1.113883.5.139"/> <offset>
<low value="1" unit="h"/> </offset></effectiveTime> | | EIVL.event | 0 … 1 | C | | (UVS...ion) | | cs | 0 … 1 | | | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.1.11.10706 TimingEvent (DYNAMIC) |
|  |  | hl7:effectiveTime
|
| SXPR_TS | 0 … 1 | R | Combined Time Interval | (UVS...ion) | | where [@xsi:type='SXPR_TS'] | |  | hl7:doseQuantity
|
| IVL_PQ | 0 … 1 | R | The doseQuantity describes the amount of the medication given (the dosage).
If a dose range is given (e.g., 1-2 tablets, or 325-750mg), then the <low> and <high> bounds are specified in their respective elements; otherwise only one physical quantity is specified (e.g. 2 drops)
The dose can be in some known and measurable unit, such as grams, milligrams,or described in "administration" units (unit of presentation, such as capsules).
If the dose is in countable items (tablets, caplets, "eaches"), then the unit could be omitted or valorized using the UCUM annotations for describing the type of countable items (e.g. .{tablet}, {puff},..).
The unit attribute – when expresses unit of measures- shall be derived from the UCUM code system.
The used elements should contain a <translation> element that provides a reference to the originalText found in the narrative body of the document. | (UVS...ion) |  |  | @unit
|
| cs | 0 … 1 | | | | | Example | Not pre-coordinated consumable <doseQuantity value="25" unit="mg"/> | | | Example | Pre-coordinated consumable - Dose Range <doseQuantity>
<low value="1" unit="{tablet}"/> <high value="2" unit="{tablet}"/></doseQuantity> | | | Example | Pre-coordinated consumable <doseQuantity value="2" unit="{puff}"/> | | | Example | Pre-coordinated consumable with text reference <doseQuantity value="2" unit="{puff}">
<translation nullFlavor="NI">
<originalText>
<reference value="#text-ref-1"/> </originalText> </translation></doseQuantity> | | | Example | Textual dosage <doseQuantity nullFlavor="NI">
<translation nullFlavor="NI">
<originalText>
<reference value="#text-ref-1"/> </originalText> </translation></doseQuantity> |  | hl7:rateQuantity
|
| IVL_PQ | 0 … 1 | | | (UVS...ion) |  | hl7:maxDoseQuantity
|
| RTO_PQ_PQ | 0 … 1 | | | (UVS...ion) |  | hl7:administrationUnitCode
|
| CE | 0 … 1 | | | (UVS...ion) | | | CONF | | The value of @code shall be drawn from value set 2.16.840.1.113883.1.11.14570 AdministrableDrugForm (DYNAMIC) |
|  | hl7:consumable
|
| | 1 … 1 | R | | (UVS...ion) |  |  | hl7:manufacturedProduct
|
| | 1 … 1 | R | | (UVS...ion) |  |  |  | hl7:manufacturedMaterial
|
| | 1 … 1 | R | | (UVS...ion) | | cs | 1 … 1 | F | NA |
|
UV Use Period
| Id | 2.16.840.1.113883.10.21.9.1 | Effective Date | 2023‑01‑30 09:55:27Other versions this id:  Useperiod as of 2017‑05‑02 Useperiod as of 2017‑05‑02 Useperiod as of 2017‑01‑02 Useperiod as of 2017‑01‑02
|
|---|
| Status |  Draft Draft | Version Label | 2023 |
|---|
| Name | Useperiod | Display Name | UV Use Period |
|---|
| Description | This element encodes the start and stop time of the medication regimen. This is an interval of time (xsi:type='IVL_TS'), and must be specified as shown. This is an additional constraint placed upon CDA Release 2.0 by this profile, and simplifies the exchange of start/stop and frequency information between EMR systems. |
|
| Classification | CDA Entry Level Template |
|---|
| Open/Closed | Open (other than defined elements are allowed) |
|---|
|
Vital Signs Organizer
1.3.6.1.4.1.19376.1.5.3.1.4.13.1 (not listed here)
Terminology
Quantity Units
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.28 | Effective Date | 2017‑05‑03 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/quantityUnits |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSQuantityUnits | Display Name | Quantity Units |
|---|
| Usage: 12 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-3 | IPS Manufactured Material (STU1) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU1) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU1) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU1) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU1) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU1) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU2) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU2) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU2) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU2) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU2) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU2) | DYNAMIC |
|
|
A valid code from the code system:| Code System Name | Code System Id | Unified Code for Units of Measure | 2.16.840.1.113883.6.8 |
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Medicine Doseform
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.25 | Effective Date | 2024‑08‑02 10:06:10 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/medicine-doseform |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSDoseForm | Display Name | IPS Medicine Doseform |
|---|
| Description | EDQM (European Directorate for the Quality of Medicines and Healthcare) Dose Form codes. This Value Set includes all the EDQM Standard Terms having:
[Concept Status] = ‘Current’ AND
[Concept Class] IN (‘PDF’, ‘CMT’, ‘CDF’, ‘PFT’) AND
[Domain] = ‘Human and Veterinary’ PDF = ‘Pharmaceutical dose form’; CMT =
‘Combined terms’; CDF = ‘Combined pharmaceutical dose form’; PFT = ‘Patient Friendly’ |
|
| Usage: 3 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-3 | IPS Manufactured Material (STU1) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (2021) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU2) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name | Code System |
|---|
| 0‑L | 10101000 | Oral drops, solution | EDQM | | 0‑L | 10102000 | Oral drops, suspension | EDQM | | 0‑L | 10103000 | Oral drops, emulsion | EDQM | | 0‑L | 10104000 | Oral liquid | EDQM | | 0‑L | 10105000 | Oral solution | EDQM | | 0‑L | 10106000 | Oral suspension | EDQM | | 0‑L | 10107000 | Oral emulsion | EDQM | | 0‑L | 10108000 | Oral gel | EDQM | | 0‑L | 10109000 | Oral paste | EDQM | | 0‑L | 10110000 | Powder for oral solution | EDQM | | 0‑L | 10111000 | Powder for oral suspension | EDQM | | 0‑L | 10112000 | Granules for oral solution | EDQM | | 0‑L | 10113000 | Granules for oral suspension | EDQM | | 0‑L | 10114000 | Powder and solvent for oral solution | EDQM | | 0‑L | 10115000 | Powder and solvent for oral suspension | EDQM | | 0‑L | 10116000 | Lyophilisate for suspension | EDQM | | 0‑L | 10117000 | Syrup | EDQM | | 0‑L | 10118000 | Powder for syrup | EDQM | | 0‑L | 10119000 | Granules for syrup | EDQM | | 0‑L | 10120000 | Soluble tablet | EDQM | | 0‑L | 10121000 | Dispersible tablet | EDQM | | 0‑L | 10122000 | Herbal tea | EDQM | | 0‑L | 10201000 | Oral powder | EDQM | | 0‑L | 10202000 | Instant herbal tea | EDQM | | 0‑L | 10203000 | Effervescent powder | EDQM | | 0‑L | 10204000 | Granules | EDQM | | 0‑L | 10205000 | Effervescent granules | EDQM | | 0‑L | 10206000 | Gastro-resistant granules | EDQM | | 0‑L | 10207000 | Prolonged-release granules | EDQM | | 0‑L | 10208000 | Modified-release granules | EDQM | | 0‑L | 10209000 | Cachet | EDQM | | 0‑L | 10210000 | Capsule, hard | EDQM | | 0‑L | 10211000 | Capsule, soft | EDQM | | 0‑L | 10212000 | Gastro-resistant capsule, hard | EDQM | | 0‑L | 10213000 | Gastro-resistant capsule, soft | EDQM | | 0‑L | 10214000 | Chewable capsule, soft | EDQM | | 0‑L | 10215000 | Prolonged-release capsule, hard | EDQM | | 0‑L | 10216000 | Prolonged-release capsule, soft | EDQM | | 0‑L | 10217000 | Modified-release capsule, hard | EDQM | | 0‑L | 10218000 | Modified-release capsule, soft | EDQM | | 0‑L | 10219000 | Tablet | EDQM | | 0‑L | 10220000 | Coated tablet | EDQM | | 0‑L | 10221000 | Film-coated tablet | EDQM | | 0‑L | 10222000 | Effervescent tablet | EDQM | | 0‑L | 10223000 | Orodispersible tablet | EDQM | | 0‑L | 10224000 | Oral lyophilisate | EDQM | | 0‑L | 10225000 | Gastro-resistant tablet | EDQM | | 0‑L | 10226000 | Prolonged-release tablet | EDQM | | 0‑L | 10227000 | Modified-release tablet | EDQM | | 0‑L | 10228000 | Chewable tablet | EDQM | | 0‑L | 10229000 | Medicated chewing-gum | EDQM | | 0‑L | 10230000 | Oral gum | EDQM | | 0‑L | 10231000 | Pillules | EDQM | | 0‑L | 10236100 | Orodispersible film | EDQM | | 0‑L | 10301000 | Gargle | EDQM | | 0‑L | 10302000 | Concentrate for gargle | EDQM | | 0‑L | 10303000 | Gargle, powder for solution | EDQM | | 0‑L | 10304000 | Gargle, tablet for solution | EDQM | | 0‑L | 10305000 | Oromucosal solution | EDQM | | 0‑L | 10306000 | Oromucosal suspension | EDQM | | 0‑L | 10307000 | Oromucosal drops | EDQM | | 0‑L | 10308000 | Oromucosal spray | EDQM | | 0‑L | 10309000 | Sublingual spray | EDQM | | 0‑L | 10310000 | Mouthwash | EDQM | | 0‑L | 10311000 | Mouthwash, tablet for solution | EDQM | | 0‑L | 10312000 | Gingival solution | EDQM | | 0‑L | 10313000 | Oromucosal gel | EDQM | | 0‑L | 10314000 | Oromucosal paste | EDQM | | 0‑L | 10314005 | Oromucosal ointment | EDQM | | 0‑L | 10314010 | Oromucosal cream | EDQM | | 0‑L | 10314011 | Buccal film | EDQM | | 0‑L | 10315000 | Gingival gel | EDQM | | 0‑L | 10316000 | Gingival paste | EDQM | | 0‑L | 10317000 | Oromucosal capsule | EDQM | | 0‑L | 10318000 | Sublingual tablet | EDQM | | 0‑L | 10319000 | Muco-adhesive buccal tablet | EDQM | | 0‑L | 10320000 | Buccal tablet | EDQM | | 0‑L | 10321000 | Lozenge | EDQM | | 0‑L | 10322000 | Compressed lozenge | EDQM | | 0‑L | 10323000 | Pastille | EDQM | | 0‑L | 10401000 | Periodontal powder | EDQM | | 0‑L | 10402000 | Dental gel | EDQM | | 0‑L | 10403000 | Dental stick | EDQM | | 0‑L | 10404000 | Dental insert | EDQM | | 0‑L | 10405000 | Dental powder | EDQM | | 0‑L | 10406000 | Dental solution | EDQM | | 0‑L | 10407000 | Dental suspension | EDQM | | 0‑L | 10408000 | Dental emulsion | EDQM | | 0‑L | 10409000 | Toothpaste | EDQM | | 0‑L | 10410000 | Periodontal gel | EDQM | | 0‑L | 10411000 | Periodontal insert | EDQM | | 0‑L | 10501000 | Bath additive | EDQM | | 0‑L | 10502000 | Cream | EDQM | | 0‑L | 10503000 | Gel | EDQM | | 0‑L | 10504000 | Ointment | EDQM | | 0‑L | 10505000 | Cutaneous paste | EDQM | | 0‑L | 10506000 | Medicated plaster | EDQM | | 0‑L | 10507000 | Cutaneous foam | EDQM | | 0‑L | 10508000 | Shampoo | EDQM | | 0‑L | 10509000 | Cutaneous spray, solution | EDQM | | 0‑L | 10510000 | Cutaneous spray, suspension | EDQM | | 0‑L | 10511000 | Cutaneous spray, powder | EDQM | | 0‑L | 10512000 | Cutaneous liquid | EDQM | | 0‑L | 10513000 | Cutaneous solution | EDQM | | 0‑L | 10514000 | Concentrate for cutaneous solution | EDQM | | 0‑L | 10515000 | Cutaneous suspension | EDQM | | 0‑L | 10516000 | Cutaneous emulsion | EDQM | | 0‑L | 10517000 | Cutaneous powder | EDQM | | 0‑L | 10517500 | Cutaneous patch | EDQM | | 0‑L | 10518000 | Solution for iontophoresis | EDQM | | 0‑L | 10519000 | Transdermal patch | EDQM | | 0‑L | 10520000 | Collodion | EDQM | | 0‑L | 10521000 | Medicated nail lacquer | EDQM | | 0‑L | 10521500 | Nail solution | EDQM | | 0‑L | 10522000 | Poultice | EDQM | | 0‑L | 10523000 | Cutaneous stick | EDQM | | 0‑L | 10524000 | Cutaneous sponge | EDQM | | 0‑L | 10525000 | Impregnated dressing | EDQM | | 0‑L | 10539500 | Scrub | EDQM | | 0‑L | 10546500 | Transdermal spray, solution | EDQM | | 0‑L | 10547000 | Transdermal system | EDQM | | 0‑L | 10548000 | Solution for skin-prick test | EDQM | | 0‑L | 10549000 | Solution for skin-scratch test | EDQM | | 0‑L | 10550000 | Plaster for provocation test | EDQM | | 0‑L | 10601000 | Eye cream | EDQM | | 0‑L | 10602000 | Eye gel | EDQM | | 0‑L | 10603000 | Eye ointment | EDQM | | 0‑L | 10604000 | Eye drops, solution | EDQM | | 0‑L | 10604500 | Eye drops, emulsion | EDQM | | 0‑L | 10605000 | Eye drops, suspension | EDQM | | 0‑L | 10606000 | Eye drops, powder and solvent for solution | EDQM | | 0‑L | 10607000 | Eye drops, powder and solvent for suspension | EDQM | | 0‑L | 10608000 | Eye drops, solvent for reconstitution | EDQM | | 0‑L | 10609000 | Eye drops, prolonged-release | EDQM | | 0‑L | 10610000 | Eye lotion | EDQM | | 0‑L | 10611000 | Eye lotion, solvent for reconstitution | EDQM | | 0‑L | 10612000 | Ophthalmic insert | EDQM | | 0‑L | 10613000 | Ophthalmic strip | EDQM | | 0‑L | 10701000 | Ear cream | EDQM | | 0‑L | 10702000 | Ear gel | EDQM | | 0‑L | 10703000 | Ear ointment | EDQM | | 0‑L | 10704000 | Ear drops, solution | EDQM | | 0‑L | 10705000 | Ear drops, suspension | EDQM | | 0‑L | 10706000 | Ear drops, emulsion | EDQM | | 0‑L | 10707000 | Ear drops, powder and solvent for suspension | EDQM | | 0‑L | 10708000 | Ear powder | EDQM | | 0‑L | 10709000 | Ear spray, solution | EDQM | | 0‑L | 10710000 | Ear spray, suspension | EDQM | | 0‑L | 10711000 | Ear spray, emulsion | EDQM | | 0‑L | 10712000 | Ear wash, solution | EDQM | | 0‑L | 10713000 | Ear wash, emulsion | EDQM | | 0‑L | 10714000 | Ear tampon | EDQM | | 0‑L | 10715000 | Ear stick | EDQM | | 0‑L | 10801000 | Nasal cream | EDQM | | 0‑L | 10802000 | Nasal gel | EDQM | | 0‑L | 10803000 | Nasal ointment | EDQM | | 0‑L | 10804000 | Nasal drops, solution | EDQM | | 0‑L | 10805000 | Nasal drops, suspension | EDQM | | 0‑L | 10806000 | Nasal drops, emulsion | EDQM | | 0‑L | 10807000 | Nasal powder | EDQM | | 0‑L | 10808000 | Nasal spray, solution | EDQM | | 0‑L | 10809000 | Nasal spray, suspension | EDQM | | 0‑L | 10810000 | Nasal spray, emulsion | EDQM | | 0‑L | 10811000 | Nasal wash | EDQM | | 0‑L | 10812000 | Nasal stick | EDQM | | 0‑L | 10900500 | Intravaginal ring | EDQM | | 0‑L | 10901000 | Vaginal cream | EDQM | | 0‑L | 10902000 | Vaginal gel | EDQM | | 0‑L | 10903000 | Vaginal ointment | EDQM | | 0‑L | 10904000 | Vaginal foam | EDQM | | 0‑L | 10905000 | Vaginal solution | EDQM | | 0‑L | 10906000 | Vaginal suspension | EDQM | | 0‑L | 10907000 | Vaginal emulsion | EDQM | | 0‑L | 10908000 | Tablet for vaginal solution | EDQM | | 0‑L | 10909000 | Pessary | EDQM | | 0‑L | 10910000 | Vaginal capsule, hard | EDQM | | 0‑L | 10911000 | Vaginal capsule, soft | EDQM | | 0‑L | 10912000 | Vaginal tablet | EDQM | | 0‑L | 10913000 | Effervescent vaginal tablet | EDQM | | 0‑L | 10914000 | Medicated vaginal tampon | EDQM | | 0‑L | 10915000 | Vaginal delivery system | EDQM | | 0‑L | 10916000 | Vaginal sponge | EDQM | | 0‑L | 11001000 | Rectal cream | EDQM | | 0‑L | 11002000 | Rectal gel | EDQM | | 0‑L | 11003000 | Rectal ointment | EDQM | | 0‑L | 11004000 | Rectal foam | EDQM | | 0‑L | 11005000 | Rectal solution | EDQM | | 0‑L | 11006000 | Rectal suspension | EDQM | | 0‑L | 11007000 | Rectal emulsion | EDQM | | 0‑L | 11008000 | Concentrate for rectal solution | EDQM | | 0‑L | 11009000 | Powder for rectal solution | EDQM | | 0‑L | 11010 | Oral drops | EDQM | | 0‑L | 11010000 | Powder for rectal suspension | EDQM | | 0‑L | 11011000 | Tablet for rectal solution | EDQM | | 0‑L | 11012000 | Tablet for rectal suspension | EDQM | | 0‑L | 11013000 | Suppository | EDQM | | 0‑L | 11014000 | Rectal capsule | EDQM | | 0‑L | 11015000 | Rectal tampon | EDQM | | 0‑L | 11050 | Oral liquid | EDQM | | 0‑L | 11101000 | Nebuliser solution | EDQM | | 0‑L | 11102000 | Nebuliser suspension | EDQM | | 0‑L | 11103000 | Powder for nebuliser suspension | EDQM | | 0‑L | 11104000 | Powder for nebuliser solution | EDQM | | 0‑L | 11105000 | Nebuliser emulsion | EDQM | | 0‑L | 11106000 | Pressurised inhalation, solution | EDQM | | 0‑L | 11107000 | Pressurised inhalation, suspension | EDQM | | 0‑L | 11108000 | Pressurised inhalation, emulsion | EDQM | | 0‑L | 11109000 | Inhalation powder | EDQM | | 0‑L | 11110000 | Inhalation powder, hard capsule | EDQM | | 0‑L | 11111000 | Inhalation powder, pre-dispensed | EDQM | | 0‑L | 11112000 | Inhalation vapour, powder | EDQM | | 0‑L | 11113000 | Inhalation vapour, capsule | EDQM | | 0‑L | 11114000 | Inhalation vapour, solution | EDQM | | 0‑L | 11115000 | Inhalation vapour, tablet | EDQM | | 0‑L | 11116000 | Inhalation vapour, ointment | EDQM | | 0‑L | 11117000 | Inhalation vapour, liquid | EDQM | | 0‑L | 11118000 | Inhalation gas | EDQM | | 0‑L | 11201000 | Solution for injection | EDQM | | 0‑L | 11202000 | Suspension for injection | EDQM | | 0‑L | 11203000 | Emulsion for injection | EDQM | | 0‑L | 11204000 | Gel for injection | EDQM | | 0‑L | 11205000 | Powder for solution for injection | EDQM | | 0‑L | 11206000 | Powder for suspension for injection | EDQM | | 0‑L | 11207000 | Powder and solvent for solution for injection | EDQM | | 0‑L | 11208000 | Powder and solvent for suspension for injection | EDQM | | 0‑L | 11209000 | Concentrate for solution for injection | EDQM | | 0‑L | 11210000 | Solution for infusion | EDQM | | 0‑L | 11210500 | Solution for infusion in administration system | EDQM | | 0‑L | 11211000 | Emulsion for infusion | EDQM | | 0‑L | 11212000 | Powder for solution for infusion | EDQM | | 0‑L | 11213000 | Concentrate for solution for infusion | EDQM | | 0‑L | 11214000 | Powder and solvent for solution for infusion | EDQM | | 0‑L | 11214500 | Lyophilisate and solvent for solution for injection | EDQM | | 0‑L | 11215000 | Lyophilisate for solution for infusion | EDQM | | 0‑L | 11216000 | Solvent for parenteral use | EDQM | | 0‑L | 11217000 | Lyophilisate for solution for injection | EDQM | | 0‑L | 11218000 | Lyophilisate for suspension for injection | EDQM | | 0‑L | 11301000 | Implant | EDQM | | 0‑L | 11302000 | Implantation tablet | EDQM | | 0‑L | 11303000 | Implantation chain | EDQM | | 0‑L | 11303500 | Implantation suspension | EDQM | | 0‑L | 11304000 | Powder and solvent for implantation paste | EDQM | | 0‑L | 11401000 | Solution for peritoneal dialysis | EDQM | | 0‑L | 11402000 | Solution for haemofiltration | EDQM | | 0‑L | 11403000 | Solution for haemodiafiltration | EDQM | | 0‑L | 11404000 | Solution for haemodialysis | EDQM | | 0‑L | 11405000 | Concentrate for haemodialysis solution | EDQM | | 0‑L | 11501000 | Solution for intravesical use | EDQM | | 0‑L | 11502000 | Bladder irrigation | EDQM | | 0‑L | 11503000 | Powder for bladder irrigation | EDQM | | 0‑L | 11504000 | Urethral gel | EDQM | | 0‑L | 11505000 | Urethral stick | EDQM | | 0‑L | 11601000 | Endotracheopulmonary instillation, solution | EDQM | | 0‑L | 11602000 | Endotracheopulmonary instillation, powder for solution | EDQM | | 0‑L | 11603000 | Endotracheopulmonary instillation, suspension | EDQM | | 0‑L | 11604000 | Endotracheopulmonary instillation, powder and solvent for solution | EDQM | | 0‑L | 11701000 | Endocervical gel | EDQM | | 0‑L | 11702000 | Powder and solvent for endocervical gel | EDQM | | 0‑L | 11901000 | Intrauterine delivery system | EDQM | | 0‑L | 11902000 | Intrauterine solution | EDQM | | 0‑L | 11903000 | Intrauterine suspension | EDQM | | 0‑L | 11904000 | Intrauterine emulsion | EDQM | | 0‑L | 11905000 | Intrauterine tablet | EDQM | | 0‑L | 11906000 | Intrauterine capsule | EDQM | | 0‑L | 12004000 | Nebulisation solution | EDQM | | 0‑L | 12100 | Capsule | EDQM | | 0‑L | 12100500 | Absorbable coated sponge | EDQM | | 0‑L | 12101000 | Denture lacquer | EDQM | | 0‑L | 12102000 | Anticoagulant and preservative solution for blood | EDQM | | 0‑L | 12103000 | Solution for blood fraction modification | EDQM | | 0‑L | 12104000 | Wound stick | EDQM | | 0‑L | 12105000 | Radiopharmaceutical precursor | EDQM | | 0‑L | 12106000 | Radionuclide generator | EDQM | | 0‑L | 12107000 | Kit for radiopharmaceutical preparation | EDQM | | 0‑L | 12108000 | Gastroenteral solution | EDQM | | 0‑L | 12109000 | Dispersion | EDQM | | 0‑L | 12109500 | Fibrin sealant-powder and solvent for fibrin sealant | EDQM | | 0‑L | 12110000 | Gastroenteral suspension | EDQM | | 0‑L | 12111000 | Gastroenteral emulsion | EDQM | | 0‑L | 12112000 | Solution for organ preservation | EDQM | | 0‑L | 12113000 | Irrigation solution | EDQM | | 0‑L | 12114000 | Stomach irrigation | EDQM | | 0‑L | 12115000 | Sealant | EDQM | | 0‑L | 12115500 | Solution of perfusion of organs | EDQM | | 0‑L | 12116000 | Powder and solvent for sealant | EDQM | | 0‑L | 12117000 | Impregnated pad | EDQM | | 0‑L | 12118000 | Living tissue equivalent | EDQM | | 0‑L | 12119000 | Medicated sponge | EDQM | | 0‑L | 12120 | Gastro-resistant capsule | EDQM | | 0‑L | 12120000 | Intestinal gel | EDQM | | 0‑L | 12130000 | Medicated thread | EDQM | | 0‑L | 12131000 | Solution for provocation test | EDQM | | 0‑L | 12150 | Prolonged-release capsule | EDQM | | 0‑L | 12200 | Tablet | EDQM | | 0‑L | 12301000 | Medicinal gas, compressed | EDQM | | 0‑L | 12302000 | Medicinal gas, cryogenic | EDQM | | 0‑L | 12303000 | Medicinal gas, liquefied | EDQM | | 0‑L | 13050 | Oromucosal liquid | EDQM | | 0‑L | 13220 | Lozenge | EDQM | | 0‑L | 14050 | Dental liquid | EDQM | | 0‑L | 15090 | Cutaneous spray | EDQM | | 0‑L | 15130 | Cutaneous liquid | EDQM | | 0‑L | 16040 | Eye drops | EDQM | | 0‑L | 17040 | Ear drops | EDQM | | 0‑L | 17090 | Ear spray | EDQM | | 0‑L | 17120 | Ear wash | EDQM | | 0‑L | 18040 | Nasal drops | EDQM | | 0‑L | 18080 | Nasal spray | EDQM | | 0‑L | 19050 | Vaginal liquid | EDQM | | 0‑L | 19100 | Vaginal capsule | EDQM | | 0‑L | 20050 | Enema | EDQM | | 0‑L | 21010 | Nebuliser liquid | EDQM | | 0‑L | 21060 | Pressurised inhalation | EDQM | | 0‑L | 21100 | Inhalation powder | EDQM | | 0‑L | 21140 | Inhalation vapour | EDQM | | 0‑L | 22010 | Injection | EDQM | | 0‑L | 22050 | Powder for injection | EDQM | | 0‑L | 22090 | Sterile concentrate | EDQM | | 0‑L | 22100 | Infusion | EDQM | | 0‑L | 22120 | Powder for infusion | EDQM | | 0‑L | 26010 | Endotracheopulmonary instillation | EDQM | | 0‑L | 29020 | Intrauterine liquid | EDQM | | 0‑L | 30047500 | Pouch | EDQM | | 0‑L | 31030 | Blood fraction modifier | EDQM | | 0‑L | 31080 | Gastroenteral liquid | EDQM | | 0‑L | 50001000 | Chewable/dispersible tablet | EDQM | | 0‑L | 50001250 | Coated granules in sachet | EDQM | | 0‑L | 50001500 | Concentrate and diluent for solution for infusion | EDQM | | 0‑L | 50002000 | Concentrate and solvent for concentrate for solution for infusion | EDQM | | 0‑L | 50003000 | Concentrate and solvent for cutaneous solution | EDQM | | 0‑L | 50004000 | Concentrate and solvent for cutaneous use | EDQM | | 0‑L | 50005000 | Concentrate and solvent for injection | EDQM | | 0‑L | 50006000 | Concentrate and solvent for solution for infusion | EDQM | | 0‑L | 50007000 | Concentrate and solvent for solution for injection | EDQM | | 0‑L | 50008000 | Concentrate and solvent for suspension for injection | EDQM | | 0‑L | 50009000 | Concentrate for cutaneous spray, emulsion | EDQM | | 0‑L | 50009300 | Concentrate for dispersion for infusion | EDQM | | 0‑L | 50009500 | Concentrate for emulsion for infusion | EDQM | | 0‑L | 50010000 | Concentrate for oral solution | EDQM | | 0‑L | 50011000 | Concentrate for oral/rectal solution | EDQM | | 0‑L | 50012000 | Concentrate for peritoneal dialysis solution | EDQM | | 0‑L | 50013000 | Concentrate for solution for intravesical use | EDQM | | 0‑L | 50013500 | Concentrate for spray emulsion | EDQM | | 0‑L | 50014000 | Concentrate for suspension for infusion | EDQM | | 0‑L | 50015000 | Cutaneous and nasal ointment | EDQM | | 0‑L | 50015300 | Cutaneous/oromucosal/oral solution | EDQM | | 0‑L | 50015400 | Cutaneous/oromucosal spray | EDQM | | 0‑L | 50015500 | Cutaneous spray, emulsion | EDQM | | 0‑L | 50016000 | Cutaneous spray, ointment | EDQM | | 0‑L | 50017000 | Dental paste | EDQM | | 0‑L | 50018000 | Ear/eye drops, solution | EDQM | | 0‑L | 50019000 | Ear/eye oinment | EDQM | | 0‑L | 50020000 | Ear/eye/nose drops, solution | EDQM | | 0‑L | 50020500 | Effervescent buccal tablet | EDQM | | 0‑L | 50021000 | Emulsion for injection/infusion | EDQM | | 0‑L | 50021500 | Emulsion and suspension for emulsion for injection | EDQM | | 0‑L | 50022000 | Endosinusial wash, suspension | EDQM | | 0‑L | 50023000 | Eye drops, solution in single-dose container | EDQM | | 0‑L | 50023500 | Film coated gastro-resistant tablet | EDQM | | 0‑L | 50024000 | Gargle/mouthwash | EDQM | | 0‑L | 50025000 | Gastro-resistant coated tablet | EDQM | | 0‑L | 50026000 | Gastro-resistant granules for oral suspension | EDQM | | 0‑L | 50026250 | Gastro-resistant prolonged-release tablet | EDQM | | 0‑L | 50026500 | Granules and solvent for oral suspension | EDQM | | 0‑L | 50027000 | Granules and solvent for suspension for injection | EDQM | | 0‑L | 50028000 | Granules for oral and rectal suspension | EDQM | | 0‑L | 50029000 | Granules for oral drops, solution | EDQM | | 0‑L | 50029250 | Granules for use in drinking water | EDQM | | 0‑L | 50029500 | Granules for vaginal solution | EDQM | | 0‑L | 50029600 | Hard capsule with gastro-resistant pellets | EDQM | | 0‑L | 50029700 | Herbal tea in bag | EDQM | | 0‑L | 50030000 | Inhalation powder, tablet | EDQM | | 0‑L | 50031000 | Inhalation vapour, effervescent tablet | EDQM | | 0‑L | 50032000 | Inhalation vapour, emulsion | EDQM | | 0‑L | 50033000 | Inhalation vapour, impregnated pad | EDQM | | 0‑L | 50033300 | Intrauterine foam | EDQM | | 0‑L | 50033500 | Intravitreal implant in applicator | EDQM | | 0‑L | 50034000 | Liquefied gas for dental use | EDQM | | 0‑L | 50035000 | Modified-release film-coated tablet | EDQM | | 0‑L | 50036000 | Modified-release granules for oral suspension | EDQM | | 0‑L | 50036100 | Muco-adhesive buccal prolonged-release tablet | EDQM | | 0‑L | 50036500 | Nasal/oromucosal solution | EDQM | | 0‑L | 50037000 | Nasal spray and oromucosal solution | EDQM | | 0‑L | 50037250 | Nasal spray, solution in single-dose container | EDQM | | 0‑L | 50037750 | Oral drops, liquid | EDQM | | 0‑L | 50038000 | Oral/rectal suspension | EDQM | | 0‑L | 50038500 | Oral solution/concentrate for nebuliser solution | EDQM | | 0‑L | 50039000 | Oromucosal patch | EDQM | | 0‑L | 50039300 | Oromucosal powder in pouch | EDQM | | 0‑L | 50039500 | Oromucosal/laryngopharyngeal solution | EDQM | | 0‑L | 50040000 | Oromucosal/laryngopharyngeal solution/spray | EDQM | | 0‑L | 50041000 | Pillules in single-dose container | EDQM | | 0‑L | 50041500 | Powder and solution for solution for injection | EDQM | | 0‑L | 50042000 | Powder and solvent for concentrate for solution for infusion | EDQM | | 0‑L | 50043000 | Powder for concentrate for solution for infusion | EDQM | | 0‑L | 50044000 | Powder and solvent for cutaneous solution | EDQM | | 0‑L | 50044500 | Powder and solvent for dispersion for injection | EDQM | | 0‑L | 50045000 | Powder and solvent for endosinusial solution | EDQM | | 0‑L | 50045500 | Powder and solvent for epilesional solution | EDQM | | 0‑L | 50046000 | Powder and solvent for gingival gel | EDQM | | 0‑L | 50047000 | Powder and solvent for instillation solution for intraocular use | EDQM | | 0‑L | 50047500 | Powder and solvent for intravesical solution | EDQM | | 0‑L | 50047700 | Powder and solvent for nebuliser solution | EDQM | | 0‑L | 50048000 | Powder and solvent for prolonged-release suspension for injection | EDQM | | 0‑L | 50048250 | Powder and solvent for solution for injection in pre-filled syringe | EDQM | | 0‑L | 50048300 | Powder and solvent for suspension for injection in pre-filled syringe | EDQM | | 0‑L | 50048500 | Powder and suspension for suspension for injection | EDQM | | 0‑L | 50048600 | Powder, dispersion and solvent for concentrate for dispersion for injection | EDQM | | 0‑L | 50048750 | Powder for concentrate for dispersion for infusion | EDQM | | 0‑L | 50049000 | Powder for concentrate for haemodialysis solution, | EDQM | | 0‑L | 50049100 | Powder for concentrate for intravesical suspension | EDQM | | 0‑L | 50049250 | Powder for concentrate for solution for injection/infusion | EDQM | | 0‑L | 50049270 | Powder for dental solution | EDQM | | 0‑L | 50049300 | Powder for epilesional solution | EDQM | | 0‑L | 50049500 | Powder for implantation suspension | EDQM | | 0‑L | 50050000 | Powder for intravesical solution | EDQM | | 0‑L | 50051000 | Powder for intravesical suspension | EDQM | | 0‑L | 50051100 | Powder for mouth wash | EDQM | | 0‑L | 50052000 | Powder for oral/rectal suspension | EDQM | | 0‑L | 50053000 | Powder for solution for injection or infusion | EDQM | | 0‑L | 50053500 | Powder for solution for injection/infusion | EDQM | | 0‑L | 50054000 | Powder for solution for intravesical use | EDQM | | 0‑L | 50055000 | Powder for solution for nasal spray | EDQM | | 0‑L | 50055500 | Prolonged-release film-coated tablet | EDQM | | 0‑L | 50056000 | Prolonged-release granules for oral suspension | EDQM | | 0‑L | 50056500 | Radiopharmaceutical precursor, solution | EDQM | | 0‑L | 50057000 | Solution for haemodialysis/haemofiltration | EDQM | | 0‑L | 50058000 | Solution for infusion and oral solution | EDQM | | 0‑L | 50059000 | Solution for injection/concentrate for solution for infusion | EDQM | | 0‑L | 50060000 | Solution for injection/infusion | EDQM | | 0‑L | 50060100 | Solution for injection in cartridge | EDQM | | 0‑L | 50060200 | Solution for injection in pre-filled pen | EDQM | | 0‑L | 50060300 | Solution for injection in pre-filled syringe | EDQM | | 0‑L | 50060400 | Solution for injection in pre-filled syringe with automatic needle guard | EDQM | | 0‑L | 50060500 | Solution for injection/infusion in pre-filled syringe | EDQM | | 0‑L | 50061000 | Solution for intraperitoneal use | EDQM | | 0‑L | 50061300 | Solution for use in drinking water | EDQM | | 0‑L | 50061500 | Solution for sealant | EDQM | | 0‑L | 50061600 | Solvent for nasal use | EDQM | | 0‑L | 50062000 | Suspension and effervescent granules for oral suspension | EDQM | | 0‑L | 50062500 | Suspension and solution for spray | EDQM | | 0‑L | 50063000 | Suspension for infusion | EDQM | | 0‑L | 50063100 | Suspension for injection in cartridge | EDQM | | 0‑L | 50063200 | Suspension for injection in pre-filled pen | EDQM | | 0‑L | 50063300 | Suspension for injection in pre-filled syringe | EDQM | | 0‑L | 50063500 | Suspension for use in drinking water | EDQM | | 0‑L | 50064000 | Tablet and solvent for rectal suspension | EDQM | | 0‑L | 50065000 | Tablet and powder for oral solution | EDQM | | 0‑L | 50066000 | Tablet for oral suspension | EDQM | | 0‑L | 50070000 | Oral suspension for use in drinking water | EDQM | | 0‑L | 50071000 | Powder and solvent for dental gel | EDQM | | 0‑L | 50072000 | Powder for use in drinking water | EDQM | | 0‑L | 50073000 | Powder for solution for intraocular irrigation | EDQM | | 0‑L | 50074000 | Solvent for solution for intraocular irrigation | EDQM | | 0‑L | 50076000 | Solvent for solution for infusion | EDQM | | 0‑L | 50077000 | Dispersion for injection | EDQM | | 0‑L | 50078000 | Gas and solvent for dispersion for injection/infusion | EDQM | | 0‑L | 50079000 | Concentrate for solution for injection/infusion | EDQM | | 0‑L | 50080000 | Powder and solvent for solution for injection/infusion | EDQM | | 0‑L | 50081000 | Inhalation solution | EDQM | | 0‑L | 50082000 | Oral drops, powder for suspension | EDQM |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Healthcare Professional Roles
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.53 | Effective Date | 2017‑06‑21 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/healthcare-professional-roles-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSHealthcareProfessionalRoles | Display Name | IPS Healthcare Professional Roles |
|---|
| Usage: 3 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-header-template-2 | IPS CDA author (STU1) | DYNAMIC | | hl7ips-header-template-5 | IPS Patient Contacts (STU1) | DYNAMIC | | hl7ips-header-template-6 | IPS CDA documentationOf (STU1) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name | Code System |
|---|
| 0‑L | 22 | Health professionals | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 221 | Medical doctors | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 2211 | Generalist medical practitioners | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 2212 | Specialist medical practitioners | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 222 | Nursing and midwifery professionals | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 2221 | Nursing professionals | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 2222 | Midwifery professionals | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 223 | Traditional and complementary medicine professionals | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 224 | Paramedical practitioners | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 225 | Veterinarians | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 226 | Other health professionals | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 2261 | Dentists | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 2262 | Pharmacists | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 2263 | Environmental and occupational health and hygiene professionals | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 2264 | Physiotherapists | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 2265 | Dieticians and nutritionists | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 2266 | Audiologists and speech therapists | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 2267 | Optometrists and ophthalmic opticians | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 2269 | Health professionals not elsewhere classified | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 32 | Health associate professionals | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 321 | Medical and pharmaceutical technicians | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 3211 | Medical imaging and therapeutic equipment technicians | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 3212 | Medical and pathology laboratory technicians | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 3213 | Pharmaceutical technicians and assistants | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 3214 | Medical and dental prosthetic technicians | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 322 | Nursing and midwifery associate professionals | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 3221 | Nursing associate professionals | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 3222 | Midwifery associate professionals | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 323 | Traditional and complementary medicine associate professionals | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 325 | Other health associate professionals | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 3251 | Dental assistants and therapists | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 3252 | Medical records and health information technicians | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 3253 | Community health workers | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 3254 | Dispensing opticians | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 3255 | Physiotherapy technicians and assistants | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 3256 | Medical assistants | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 3257 | Environmental and occupational health inspectors and associates | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 3258 | Ambulance workers | 2.16.840.1.113883.2.9.6.2.7 | | 0‑L | 3259 | Health associate professionals not elsewhere classified | 2.16.840.1.113883.2.9.6.2.7 |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
Medicine Package
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.27 | Effective Date | 2017‑05‑03 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSPackage | Display Name | Medicine Package |
|---|
| Description | Value Set Definition:
Value Set Definition
DrawnFromCodeSystem 0.4.0.127.0.16.1.1.2.1 (EDQM)
PropertyBasedContentSet.IncludeWithProperty.Expression
(
([Concept Status] = ‘Current’)
AND ([Concept Class] IN (‘Administration device ; ‘Container ‘;’Closure’ )
AND ([Domain] = 'Human and Veterinary')
)
The list of values reported hereafter is provided only as example
|
|
| Usage: 6 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-3 | IPS Manufactured Material (STU1) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU1) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU1) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU2) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU2) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU2) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name | Code System |
|---|
| 0‑L | 30000500 | Administration system | EDQM | | 0‑L | 30001000 | Ampoule | EDQM | | 0‑L | 30002000 | Applicator | EDQM | | 0‑L | 30004000 | Bag | EDQM | | 0‑L | 30006000 | Barrel | EDQM | | 0‑L | 30007000 | Blister | EDQM | | 0‑L | 30008000 | Bottle | EDQM | | 0‑L | 30009000 | Box | EDQM | | 0‑L | 30010000 | Brush | EDQM | | 0‑L | 30011000 | Brush applicator | EDQM | | 0‑L | 30012000 | Cannula | EDQM | | 0‑L | 30013000 | Cap | EDQM | | 0‑L | 30014000 | Cartridge | EDQM | | 0‑L | 30015000 | Child-resistant closure | EDQM | | 0‑L | 30016000 | Cup | EDQM | | 0‑L | 30017000 | Dabbing applicator | EDQM | | 0‑L | 30019000 | Dredging applicator | EDQM | | 0‑L | 30020000 | Dredging container | EDQM | | 0‑L | 30022000 | Dropper applicator | EDQM | | 0‑L | 30023000 | Dropper container | EDQM | | 0‑L | 30023005 | Fixed cryogenic vessel | EDQM | | 0‑L | 30024000 | Gas cylinder | EDQM | | 0‑L | 30025000 | High pressure transdermal delivery device | EDQM | | 0‑L | 30026000 | Implanter | EDQM | | 0‑L | 30026500 | Inhaler | EDQM | | 0‑L | 30027000 | In-ovo injection device | EDQM | | 0‑L | 30028000 | Injection needle | EDQM | | 0‑L | 30029000 | Injection syringe | EDQM | | 0‑L | 30031000 | Intramammary syringe | EDQM | | 0‑L | 30032000 | Jar | EDQM | | 0‑L | 30033000 | Measuring device | EDQM | | 0‑L | 30034000 | Measuring spoon | EDQM | | 0‑L | 30035000 | Metering pump | EDQM | | 0‑L | 30036000 | Metering valve | EDQM | | 0‑L | 30036005 | Mobile cryogenic vessel | EDQM | | 0‑L | 30037000 | Mouthpiece | EDQM | | 0‑L | 30038000 | Multidose container | EDQM | | 0‑L | 30039000 | Multidose container with airless pump | EDQM | | 0‑L | 30040000 | Multipuncturer | EDQM | | 0‑L | 30041000 | Nasal applicator | EDQM | | 0‑L | 30042000 | Nebuliser | EDQM | | 0‑L | 30043000 | Needle applicator | EDQM | | 0‑L | 30044000 | Nozzle | EDQM | | 0‑L | 30045000 | Oral syringe | EDQM | | 0‑L | 30046000 | Pipette | EDQM | | 0‑L | 30047000 | Pipette applicator | EDQM | | 0‑L | 30048000 | Pour-on container | EDQM | | 0‑L | 30049000 | Pre-filled gastroenteral tube | EDQM | | 0‑L | 30050000 | Pre-filled pen | EDQM | | 0‑L | 30051000 | Pre-filled syringe | EDQM | | 0‑L | 30052000 | Pressurised container | EDQM | | 0‑L | 30053000 | Prick test applicator | EDQM | | 0‑L | 30053500 | Roll-on container | EDQM | | 0‑L | 30054000 | Sachet | EDQM | | 0‑L | 30055000 | Scarifier | EDQM | | 0‑L | 30056000 | Screw cap | EDQM | | 0‑L | 30057000 | Single-dose container | EDQM | | 0‑L | 30058000 | Spatula | EDQM | | 0‑L | 30059000 | Spot-on applicator | EDQM | | 0‑L | 30060000 | Spray container | EDQM | | 0‑L | 30061000 | Spray pump | EDQM | | 0‑L | 30062000 | Spray valve | EDQM | | 0‑L | 30063000 | Stab vaccinator | EDQM | | 0‑L | 30064000 | Stopper | EDQM | | 0‑L | 30064500 | Straw | EDQM | | 0‑L | 30065000 | Strip | EDQM | | 0‑L | 30066000 | Tablet container | EDQM | | 0‑L | 30067000 | Tube | EDQM | | 0‑L | 30069000 | Vial | EDQM |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Allergy Reaction
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.3 | Effective Date | 2024‑08‑02 09:21:05 |
|---|
| Canonical URI | http://hl7.org/fhir/ips/ValueSet/allergy-reaction-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSAllergyReaction | Display Name | IPS Allergy Reaction |
|---|
| Description | IPS Allergy Reaction Value Set. |
|---|
| Copyright | Licensing note: This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org.This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-6 | IPS Reaction Manifestation (STU2) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Intensional Definition | Code System |
|---|
| |  Include | code and descendants 4386001 Bronchospasm (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 9826008 Conjunctivitis (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 23924001 Pressure in chest (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 24079001 Atopic dermatitis (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 31996006 Vasculitis (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 39579001 Anaphylaxis (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 41291007 Angioedema (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 43116000 Eczema (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 49727002 Cough (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 51599000 Edema of larynx (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 62315008 Diarrhea (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 70076002 Rhinitis (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 73442001 Stevens-Johnson syndrome (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 76067001 Sneezing (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 91175000 Seizure (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 126485001 Urticaria (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 162290004 Dry eyes (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 195967001 Airway hyperreactivity (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 247472004 Weal (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 267036007 Dyspnea (finding | SNOMED Clinical Terms | | |  Include | code and descendants 271757001 Papular eruption (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 271759003 Bullous eruption (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 271807003 Eruption (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 410430005 Cardiorespiratory arrest (disorder) | SNOMED Clinical Terms | | |  Include | code and descendants 418363000 Itching of skin (finding | SNOMED Clinical Terms | | |  Include | code and descendants 422400008 Vomiting (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 422587007 Nausea (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 698247007 Arrhythmia (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 702809001 DRESS syndrome (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 768962006 Lyell syndrome (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 781682005 Bloodshot eye (finding) | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
Laterality (qualifier)
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.57 | Effective Date | 2017‑12‑12 19:27:25 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | lateralityQualifierCode | Display Name | Laterality (qualifier) |
|---|
| Copyright | This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 2 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-12 | IPS Radiology Result Observation (STU2) | DYNAMIC | | hl7ips-entry-template-12 | IPS Radiology Result Observation (STU1) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name | Code System | Designations |
|---|
| 0‑L | 7771000 | Left (qualifier value) | SNOMED Clinical Terms | | | 0‑L | 24028007 | Right (qualifier value) | SNOMED Clinical Terms | | | 0‑L | 51440002 | Right and left (qualifier value) | SNOMED Clinical Terms | |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Results Coded Values Laboratory/Pathology
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.74 | Effective Date | 2024‑08‑02 10:25:29 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/procedures-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSResultsCodedLaboratoryPathology | Display Name | IPS Results Coded Values Laboratory/Pathology |
|---|
| Description | IPS Results Coded Values Laboratory/Pathology value set. |
|---|
| Copyright | Licensing note: This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org.This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 2 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-13 | IPS Laboratory Result Observation (STU2) | DYNAMIC | | hl7ips-entry-template-11 | IPS Pathology Result Observation (STU2) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Intensional Definition | Code System |
|---|
| |  Include | descendants of code 365636006 Blood group | SNOMED Clinical Terms | | |  Include | descendants of code 260411009 Presence findings | SNOMED Clinical Terms | | |  Include | descendants of code 272519000 Absence findings | SNOMED Clinical Terms | | |  Include | descendants of code 409822003 Domain Bacteria (organism) | SNOMED Clinical Terms | | |  Include | descendants of code 441649000 Class Cestoda and/or Class Trematoda and/or Phylum Nemata (organism) | SNOMED Clinical Terms | | |  Include | descendants of code 414561005 Kingdom Fungi (organism) | SNOMED Clinical Terms | | |  Include | descendants of code 84676004 Prion | SNOMED Clinical Terms | | |  Include | descendants of code 49872002 Virus | SNOMED Clinical Terms | | |  Include | descendants of code 417396000 Kingdom Protozoa (organism) | SNOMED Clinical Terms | | |  Include | descendants of code 419036000 Superkingdom Archaea (organism) | SNOMED Clinical Terms | | |  Include | descendants of code 426785004 Kingdom Chromista (organism) | SNOMED Clinical Terms | | |  Include | descendants of code 370570004 Kingdom Protista (organism) | SNOMED Clinical Terms | | |  Include | descendants of code 417377004 Kingdom Viridiplantae (organism) | SNOMED Clinical Terms | | |  Include | descendants of code 243565002 Slime mold (finding (organism) | SNOMED Clinical Terms | | |  Include | descendants of code 106253005 Histologic grading differentiation AND/OR behavior | SNOMED Clinical Terms | | |  Include | descendants of code 373369003 Finding of histologic grading differentiation AND/OR behavior (finding) | SNOMED Clinical Terms | | |  Include | descendants of code 399981008 Neoplasm and/or hamartoma (disorder) | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Personal Relationship
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.54 | Effective Date | 2017‑07‑18 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/personal-relationship-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSPersonalRelationship | Display Name | IPS Personal Relationship |
|---|
| Description | The Value Set is used (optionally) to code the type of contact relationship between a person and the patient. |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-header-template-5 | IPS Patient Contacts (STU1) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name | Code System |
|---|
| 0‑L | AUNT | aunt | Role Code | | 0‑L | CHILD | child | Role Code | | 0‑L | CHLDADOPT | adopted child | Role Code | | 0‑L | CHLDFOST | foster child | Role Code | | 0‑L | CHLDINLAW | child in-law | Role Code | | 0‑L | COUSN | cousin | Role Code | | 0‑L | DAU | natural daughter | Role Code | | 0‑L | DAUADOPT | adopted daughter | Role Code | | 0‑L | DAUC | daughter | Role Code | | 0‑L | DAUFOST | foster daughter | Role Code | | 0‑L | DAUINLAW | daughter in-law | Role Code | | 0‑L | DOMPART | domestic partner | Role Code | | 0‑L | FAMMEMB | family member | Role Code | | 0‑L | FRND | unrelated friend | Role Code | | 0‑L | FTH | father | Role Code | | 0‑L | FTHINLAW | father-in-law | Role Code | | 0‑L | GGRPRN | great grandparent | Role Code | | 0‑L | GRNDCHILD | grandchild | Role Code | | 0‑L | GRPRN | grandparent | Role Code | | 0‑L | MTH | mother | Role Code | | 0‑L | MTHINLAW | mother-in-law | Role Code | | 0‑L | NBOR | neighbor | Role Code | | 0‑L | NCHILD | natural child | Role Code | | 0‑L | NIENEPH | niece/nephew | Role Code | | 0‑L | PRN | parent | Role Code | | 0‑L | PRNINLAW | parent in-law | Role Code | | 0‑L | ROOM | roomate | Role Code | | 0‑L | SIB | sibling | Role Code | | 0‑L | SIGOTHR | significant other | Role Code | | 0‑L | SON | natural son | Role Code | | 0‑L | SONADOPT | adopted son | Role Code | | 0‑L | SONC | son | Role Code | | 0‑L | SONFOST | foster son | Role Code | | 0‑L | SONINLAW | son in-law | Role Code | | 0‑L | SPS | spouse | Role Code | | 0‑L | STPCHLD | step child | Role Code | | 0‑L | STPDAU | stepdaughter | Role Code | | 0‑L | STPSON | stepson | Role Code | | 0‑L | UNCLE | uncle | Role Code |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Procedures
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.35 | Effective Date | 2024‑08‑02 10:25:29 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/procedures-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSProcedures | Display Name | IPS Procedures |
|---|
| Description | IPS Procedure codes value set. |
|---|
| Copyright | Licensing note: This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org.This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 4 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-17 | IPS Procedure Entry (2021) | DYNAMIC | | hl7ips-entry-template-17 | IPS Procedure Entry (STU1) | DYNAMIC | | hl7ips-entry-template-17 | IPS Procedure Entry (STU2) | DYNAMIC | | hl7ips-entry-template-38 | IPS Planned Procedure (TI-2020) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name / Intensional Definition | Code System |
|---|
| |  Include | descendants of code 71388002 Procedure (procedure) | SNOMED Clinical Terms | | 0‑L | 787480003 | No known procedures | SNOMED Clinical Terms | | |  Exclude | code and descendants 14734007 Administrative procedure (procedure) | SNOMED Clinical Terms | | |  Exclude | code and descendants 59524001 Blood bank procedure (procedure) | SNOMED Clinical Terms | | |  Exclude | code and descendants 389067005 Community health procedure (procedure) | SNOMED Clinical Terms | | |  Exclude | code and descendants 442006003 Determination of information related to transfusion (procedure) | SNOMED Clinical Terms | | |  Exclude | code and descendants 225288009 Environmental care procedure (procedure) | SNOMED Clinical Terms | | |  Exclude | code and descendants 308335008 Patient encounter procedure (procedure) | SNOMED Clinical Terms | | |  Exclude | code and descendants 710135002 Promotion (procedure) | SNOMED Clinical Terms | | |  Exclude | code and descendants 389084004 Staff related procedure (procedure) | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Condition Status Code
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.24 | Effective Date | 2017‑05‑02 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSConditionStatusCode | Display Name | IPS Condition Status Code |
|---|
| Description | This is a placeholder for the condition status code based on SNOMED CT |
|---|
| Usage: 2 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-template-56 | IPS Entry Problem Status Observation | STATIC | | hl7ips-entry-template-20 | IPS Problem Status Observation (STU1) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name | Code System | Description |
|---|
| 0‑S | active | Active | CareTeamCategory | The subject is currently experiencing the symptoms of the condition or there is evidence of the condition | | 1‑L | recurrence | Recurrence | CareTeamCategory | The subject is experiencing a re-occurrence or repeating of a previously resolved condition
- Example: recurrence of (previously resolved) urinary tract infection, pancreatitis, cholangitis, conjunctivitis
| | 1‑L | relapse | Relapse | CareTeamCategory | The subject is experiencing a return of a condition, or signs and symptoms after a period of improvement or remission
- Examples: relapse of cancer, multiple sclerosis, rheumatoid arthritis, systemic lupus erythematosus, bipolar disorder, [psychotic relapse of] schizophrenia, etc
| | 0‑S | inactive | Inactive | CareTeamCategory | The subject is no longer experiencing the symptoms of the condition or there is no longer evidence of the condition | | 1‑L | remission | Remission | CareTeamCategory | The subject is no longer experiencing the symptoms of the condition, but there is a risk of the symptoms or condition returning | | 1‑L | resolved | Resolved | CareTeamCategory | The subject is no longer experiencing the symptoms of the condition and there is a negligible perceived risk of the symptoms returning |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Results Specimen Collection Method
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.76 | Effective Date | 2024‑08‑02 10:53:20 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSResultsSpecimenCollectionMethod | Display Name | IPS Results Specimen Collection Method |
|---|
| Copyright | This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-30 | IPS Specimen Collection (STU2) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name / Intensional Definition | Code System |
|---|
| |  Include | descendants of code 129316008 Aspiration - action (qualifier value) | SNOMED Clinical Terms | | |  Include | descendants of code 129314006 Biopsy - action (qualifier value) | SNOMED Clinical Terms | | |  Include | descendants of code 129300006 Puncture - action (qualifier value) | SNOMED Clinical Terms | | |  Include | descendants of code 129304002 Excision - action (qualifier value) | SNOMED Clinical Terms | | |  Include | descendants of code 129323009 Scraping - action (qualifier value) | SNOMED Clinical Terms | | |  Include | descendants of code 73416001 Urine specimen collection, clean catch (procedure) | SNOMED Clinical Terms | | |  Include | descendants of code 225113003 Timed urine collection (procedure) | SNOMED Clinical Terms | | |  Include | descendants of code 70777001 Urine specimen collection, catheterized (procedure) | SNOMED Clinical Terms | | 0‑L | 386089008 | Collection of coughed sputum (procedure) | SNOMED Clinical Terms | | 0‑L | 278450005 | Finger-prick sampling (procedure) | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Results Radiology Textual Observations
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.66 | Effective Date | 2024‑08‑02 10:50:26 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSResultsRadiologyTextualObservations | Display Name | IPS Results Radiology Textual Observations |
|---|
| Description | IPS Results Radiology Textual Observation |
|---|
| Copyright | This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org.This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-document-template- | IPS Radiology Result Observation Component (STU2) | DYNAMIC |
|
|
| 3 Source Code Systems | 2.16.840.1.113883.6.1 - Logical Observation Identifier Names and Codes - FHIR: http://loinc.org - HL7 V2: LN |
|---|
| Level/ Type | Code | Display Name | Code System |
|---|
| 0‑L | 11329-0 | History | Logical Observation Identifier Names and Codes | | 0‑L | 55115-0 | Request | Logical Observation Identifier Names and Codes | | 0‑L | 121065 | Procedure Description | DICOM Controlled Terminology | | 0‑L | 121069 | Previous Finding | DICOM Controlled Terminology | | 0‑L | 121071 | Finding | DICOM Controlled Terminology | | 0‑L | 121073 | Impression | DICOM Controlled Terminology | | 0‑L | 121075 | Recommendation | DICOM Controlled Terminology | | 0‑L | 121077 | Conclusion | DICOM Controlled Terminology | | 0‑L | 121110 | Patient Presentation | DICOM Controlled Terminology | | 0‑L | 121111 | Summary | DICOM Controlled Terminology | | 0‑L | 116224001 | Complication of Procedure | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Allergy or Intolerance Conditions
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.10 | Effective Date | 2024‑08‑02 09:16:33 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSAllergyOrIntoleranceConditions | Display Name | IPS Allergy or Intolerance Conditions |
|---|
| Description | IPS Allergy intolerance codes value set with focus on Conditions. |
|---|
| Copyright | Licensing note: This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org.This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 2 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-1 | IPS Allergy or Intolerance (STU1) | DYNAMIC | | hl7ips-entry-template-1 | IPS Allergy or Intolerance (STU2) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Intensional Definition | Code System |
|---|
| |  Include | descendants of code 420134006 Propensity to adverse reaction (finding) | SNOMED Clinical Terms | | |  Include | code and descendants 716186003 No known allergy (situation) | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
Allergy or Intolerance Type
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.2 | Effective Date | 2016‑12‑06 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | allergyOrIntollerance_VS | Display Name | Allergy or Intolerance Type |
|---|
| Usage: 2 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-1 | IPS Allergy or Intolerance (STU1) | DYNAMIC | | hl7ips-entry-template-1 | IPS Allergy or Intolerance (STU2) | DYNAMIC |
|
|
| 2 Source Code Systems | |
|---|
| Level/ Type | Code | Display Name | Code System | Description |
|---|
| 1‑L | OINT | Allergy or Intolerance | Act Code | Hypersensitivity resulting in an adverse reaction upon exposure to an agent.
| | 1‑L | allergy | Allergy | 2.16.840.1.113883.4.642.1.122 | A propensity for hypersensitivity reaction(s) to a substance. These reactions are most typically type I hypersensitivity, plus other "allergy-like" reactions, including pseudoallergy.
| | 1‑L | intolerance | Intolerance | 2.16.840.1.113883.4.642.1.122 | A propensity for adverse reactions to a substance that is not judged to be allergic or "allergy-like". These reactions are typically (but not necessarily) non-immune. They are to some degree idiosyncratic and/or individually specific (i.e. are not a reaction that is expected to occur with most or all patients given similar circumstances).
|
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Current Smoking Status
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.59 | Effective Date | 2024‑08‑02 09:32:33 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/current-smoking-status-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSCurrentSmokingStatus | Display Name | IPS Current Smoking Status |
|---|
| Description | HL7 IPS SNOMED value set for smoking status. |
|---|
| Copyright | This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-34 | IPS Social History Tobacco Use (STU1) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name | Code System |
|---|
| 0‑L | 449868002 | Smokes tobacco daily (finding) | SNOMED Clinical Terms | | 0‑L | 428041000124106 | Occasional tobacco smoker (finding) | SNOMED Clinical Terms | | 0‑L | 8517006 | Ex-smoker (finding) | SNOMED Clinical Terms | | 0‑L | 266919005 | Never smoked tobacco (finding) | SNOMED Clinical Terms | | 0‑L | 77176002 | Smoker (finding) | SNOMED Clinical Terms | | 0‑L | 266927001 | Tobacco smoking consumption unknown (finding) | SNOMED Clinical Terms | | 0‑L | 230063004 | Heavy cigarette smoker (finding) | SNOMED Clinical Terms | | 0‑L | 230060001 | Light cigarette smoker (finding) | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Medications Products
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.71 | Effective Date | 2024‑08‑02 09:41:07 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSMedicationsProducts | Display Name | IPS Medications Products |
|---|
| Description | IPS Medication codes value set. |
|---|
| Copyright | Licensing note: This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org.This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-3 | IPS Manufactured Material (STU2) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Intensional Definition | Code System |
|---|
| |  Include | descendants of code 763158003 Medicinal product (product) | SNOMED Clinical Terms | | |  Exclude | code and descendants 787859002 Vaccine product (medicinal product) | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Medical Devices
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.23 | Effective Date | 2024‑08‑02 09:37:16 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/medical-devices-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSMedicalDevices | Display Name | IPS Medical Devices |
|---|
| Description | IPS Medical device codes value set. |
|---|
| Copyright | Licensing note: This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org.This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 3 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-17 | IPS Procedure Entry (2021) | DYNAMIC | | hl7ips-entry-template-17 | IPS Procedure Entry (STU2) | DYNAMIC | | hl7ips-entry-template-26 | IPS Medical Device (STU1) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name / Intensional Definition | Code System |
|---|
| |  Include | descendants of code 49062001 Device (physical object) | SNOMED Clinical Terms | | 0‑L | 787483001 | No known device use (situation) | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Allergy Verification Status
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.62 | Effective Date | 2018‑02‑07 12:04:21 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSAllergyVerificationStatus | Display Name | IPS Allergy Verification Status |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-document-template- | IPS Allergy Certainty Observation (STU1) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name | Code System | Description |
|---|
| 0‑S | unconfirmed | Unconfirmed | AllergyIntoleranceVerificationStatus | There is not sufficient diagnostic and/or clinical evidence to treat this as a confirmed condition. | | 0‑L | confirmed | Confirmed | AllergyIntoleranceVerificationStatus | A high level of certainty about the propensity for a reaction to the identified substance, which may include clinical evidence by testing or rechallenge. | | 0‑L | refuted | Refuted | AllergyIntoleranceVerificationStatus | A propensity for a reaction to the identified substance has been disproven with a high level of clinical certainty, which may include testing or rechallenge, and is refuted. |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Vaccines
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.44 | Effective Date | 2024‑08‑02 11:41:23 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/vaccines-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSVaccines | Display Name | IPS Vaccines |
|---|
| Description | IPS Vaccine codes value set. |
|---|
| Copyright | Licensing note: This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org.This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 2 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-16 | IPS Immunization Medication Information (STU1) | DYNAMIC | | hl7ips-entry-template-16 | IPS Immunization Medication Information (STU2) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name / Intensional Definition | Code System |
|---|
| |  Include | descendants of code 71181003 Vaccine (product) | SNOMED Clinical Terms | | 0‑L | 787482006 | No known immunizations (situation) | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Problems
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.73 | Effective Date | 2024‑08‑02 09:41:07 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSProblems | Display Name | IPS Problems |
|---|
| Description | IPS Problem (Condition) codes value set. |
|---|
| Copyright | This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-8 | IPS Problem Entry (2021) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name / Intensional Definition | Code System |
|---|
| |  Include | descendants of code 404684003 Clinical finding (finding) | SNOMED Clinical Terms | | |  Include | descendants of code 243796009 Situation with explicit context (situation) | SNOMED Clinical Terms | | |  Include | descendants of code 272379006 Event (event) | SNOMED Clinical Terms | | 0‑L | 160245001 | No current problems or disability (situation) | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
Allergy-intolerance Criticality
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.60 | Effective Date | 2017‑12‑14 10:22:48 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | allergy-intolerance-criticality | Display Name | Allergy-intolerance Criticality |
|---|
| Description |
description:
Recommended values for criticality observations
|
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-18 | IPS Criticality Observation (STU1) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name | Code System | Description |
|---|
| 1‑L | high | High Risk | 2.16.840.1.113883.4.642.1.120 | Worst case result of a future exposure is assessed to be life-threatening or having high potential for organ system failure.
| | 1‑L | low | Low Risk | 2.16.840.1.113883.4.642.1.120 | Worst case result of a future exposure is not assessed to be life-threatening or having high potential for organ system failure.
| | 1‑L | unable-to-assess | Unable to Assess Risk | 2.16.840.1.113883.4.642.1.120 | Unable to assess the worst case result of a future exposure.
|
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Results Laboratory/Pathology Observation
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.75 | Effective Date | 2024‑08‑02 10:25:29 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/procedures-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSResultsLaboratoryPathology | Display Name | IPS Results Laboratory/Pathology Observation |
|---|
| Description | IPS Results Values Laboratory/Pathology value set. Value Set Definition: LOINC {STATUS in {ACTIVE}, CLASSTYPE in {Laboratory class (1)}, CLASS exclude {LP62148-9 (NR STATS), LP175679-2 (H&P.HX.LAB), LP7785-1 (CHALSKIN), LP94892-4 (LABORDERS)}} |
|
| Usage: 2 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-13 | IPS Laboratory Result Observation (STU2) | DYNAMIC | | hl7ips-entry-template-11 | IPS Pathology Result Observation (STU2) | DYNAMIC |
|
|
| Source Code System | 2.16.840.1.113883.6.1 - Logical Observation Identifier Names and Codes - FHIR: http://loinc.org - HL7 V2: LN |
|---|
| Level/ Type | Code | Intensional Definition | Code System |
|---|
| |  Include | A valid code from the code system where:- Property STATUS equal "ACTIVE"
- Property CLASSTYPE equal "Laboratory class"
| Logical Observation Identifier Names and Codes | | |  Exclude | A valid code from the code system where:- Property CLASS equal "LP62148-9"
| Logical Observation Identifier Names and Codes | | |  Exclude | A valid code from the code system where:- Property CLASS equal "LP175679-2"
| Logical Observation Identifier Names and Codes | | |  Exclude | A valid code from the code system where:- Property CLASS equal "LP7785-1"
| Logical Observation Identifier Names and Codes | | |  Exclude | A valid code from the code system where:- Property CLASS equal "LP94892-4"
| Logical Observation Identifier Names and Codes |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Allergy Status Code
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.42 | Effective Date | 2017‑05‑24 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSAllergyStatusCode | Display Name | IPS Allergy Status Code |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-21 | IPS Allergy Status Observation (STU1) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name | Code System | Description |
|---|
| 0‑S | active | Active | CareTeamCategory | The subject is currently experiencing the symptoms of the condition or there is evidence of the condition | | 0‑S | inactive | Inactive | CareTeamCategory | The subject is no longer experiencing the symptoms of the condition or there is no longer evidence of the condition | | 1‑L | resolved | Resolved | CareTeamCategory |
The subject is no longer experiencing the symptoms of the condition and there is a negligible perceived risk of the symptoms returning
|
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Medicine Active Substances
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.32 | Effective Date | 2024‑08‑02 09:41:07 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/medicine-active-substances-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSActiveSubstances | Display Name | IPS Medicine Active Substances |
|---|
| Description | IPS Medicine active substance codes value set. |
|---|
| Copyright | Licensing note: This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org.This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 2 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-3 | IPS Manufactured Material (STU1) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU2) | DYNAMIC |
|
|
| Level/ Type | Code | Intensional Definition | Code System |
|---|
| |  Include | descendants of code 410942007 Drug or medicament (substance) | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Body Site
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.55 | Effective Date | 2024‑08‑02 09:31:31 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/target-site-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSTargetSiteCode | Display Name | IPS Body Site |
|---|
| Description | IPS body site value set.
|
|---|
| Copyright | Licensing note: This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org.This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 8 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-17 | IPS Procedure Entry (2021) | DYNAMIC | | hl7ips-entry-template-12 | IPS Radiology Result Observation (STU2) | DYNAMIC | | hl7ips-entry-template-17 | IPS Procedure Entry (STU1) | DYNAMIC | | hl7ips-entry-template-17 | IPS Procedure Entry (STU2) | DYNAMIC | | hl7ips-entry-template-30 | IPS Specimen Collection (STU2) | DYNAMIC | | hl7ips-entry-template-12 | IPS Radiology Result Observation (STU1) | DYNAMIC | | hl7ips-entry-template-38 | IPS Planned Procedure (TI-2020) | DYNAMIC | | hl7ips-entry-template-10 | IPS Result Observation (STU1) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Intensional Definition | Code System |
|---|
| |  Include | descendants of code 442083009 Anatomical or acquired body structure (body structure) | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Results Radiology Observation
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.40 | Effective Date | 2024‑08‑02 10:48:07 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/imaging-observations-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSRadResultsObservation | Display Name | IPS Results Radiology Observation |
|---|
| Description | Value Set Definition: LOINC {STATUS in {ACTIVE}, CLASS in LP29684-5 ("RAD")} |
|---|
| Usage: 3 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-12 | IPS Radiology Result Observation (STU2) | DYNAMIC | | hl7ips-document-template- | IPS Radiology Result Observation Component (STU2) | DYNAMIC | | hl7ips-entry-template-12 | IPS Radiology Result Observation (STU1) | DYNAMIC |
|
|
| Source Code System | 2.16.840.1.113883.6.1 - Logical Observation Identifier Names and Codes - FHIR: http://loinc.org - HL7 V2: LN |
|---|
| Level/ Type | Code | Intensional Definition | Code System |
|---|
| |  Include | A valid code from the code system where:- Property STATUS equal "ACTIVE"
- Property CLASS equal "LP29684-5"
| Logical Observation Identifier Names and Codes |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Pregnancy Status
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.68 | Effective Date | 2024‑08‑02 10:17:58 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/pregnancy-status-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | Pregnancy_status_uv_ips | Display Name | IPS Pregnancy Status |
|---|
| Copyright | This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 3 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-27 | IPS Pregnancy Status Observation | DYNAMIC | | hl7ips-entry-template-27 | IPS Pregnancy Status Observation (2021) | DYNAMIC | | hl7ips-entry-template-27 | IPS Pregnancy Status Observation (TI-2020) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name | Code System |
|---|
| 0‑L | 77386006 | Pregnancy (finding) | SNOMED Clinical Terms | | 0‑L | 60001007 | Not pregnant (finding) | SNOMED Clinical Terms | | 0‑L | 152231000119106 | Pregnancy not yet confirmed (finding) | SNOMED Clinical Terms | | 0‑L | 146799005 | Possible pregnancy (situation) | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
Absent or Unknown Devices
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.61 | Effective Date | 2018‑01‑27 22:19:47 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/absent-or-unknown-devices-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSNoDevicesInfos | Display Name | Absent or Unknown Devices |
|---|
| Usage: 3 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-17 | IPS Procedure Entry (2021) | DYNAMIC | | hl7ips-entry-template-17 | IPS Procedure Entry (STU2) | DYNAMIC | | hl7ips-entry-template-26 | IPS Medical Device (STU1) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name | Code System | Description |
|---|
| 0‑L | no-device-info | No information about device | IPS CodeSystem - Absent and Unknown Data | There is no information available regarding implanted or external devices for the subject. | | 0‑L | no-known-devices | No known devices in use | IPS CodeSystem - Absent and Unknown Data | There are no devices known to be implanted in or used by the subject that have to be reported in this record. This can mean either that there are none known, or that those known are not relevant for the purpose of this record. |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Allergy or Intolerance Substances
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.65 | Effective Date | 2024‑08‑02 09:13:15 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/allergy-intolerance-substance-condition-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | Allergyintolerancesubstancecondition | Display Name | IPS Allergy or Intolerance Substances |
|---|
| Description | IPS Allergy intolerance codes value set with focus on Substances. |
|---|
| Copyright | This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-1 | IPS Allergy or Intolerance (STU2) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Intensional Definition | Code System |
|---|
| |  Include | descendants of code 105590001 Substance (substance) | SNOMED Clinical Terms | | |  Include | descendants of code 373873005 Pharmaceutical / biologic product (product) | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Vaccines WHO ATC
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.78 | Effective Date | 2024‑08‑02 11:41:23 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/vaccines-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSWHOVaccines | Display Name | IPS Vaccines WHO ATC |
|---|
| Description | IPS Vaccine codes value set. This value set includes codes from the World Health Organization Anatomical Therapeutic Chemical (ATC) classification system: all descendants of J07 “VACCINES” |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-16 | IPS Immunization Medication Information (STU2) | DYNAMIC |
|
|
| Source Code System | 2.16.840.1.113883.6.73 - WHO Anatomical Therapeutic Chemical classification - FHIR: http://www.whocc.no/atc - HL7 V2: WC |
|---|
| Level/ Type | Code | Intensional Definition | Code System |
|---|
| |  Include | descendants of code J07 VACCINES | WHO Anatomical Therapeutic Chemical classification |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Pregnancies Summary
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.21 | Effective Date | 2017‑04‑13 |
|---|
| Canonical URI | http://hl7.org/fhir//ips/ValueSet/pregnancies-summary-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSPregnanciesSummary | Display Name | IPS Pregnancies Summary |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-28 | IPS Pregnancy Outcome Observation (STU1) | DYNAMIC |
|
|
| Source Code System | 2.16.840.1.113883.6.1 - Logical Observation Identifier Names and Codes - FHIR: http://loinc.org - HL7 V2: LN |
|---|
| Level/ Type | Code | Display Name | Code System |
|---|
| 0‑L | 11636-8 | [#] Births.live | Logical Observation Identifier Names and Codes | | 0‑L | 11637-6 | [#] Births.preterm | Logical Observation Identifier Names and Codes | | 0‑L | 11638-4 | [#] Births.still living | Logical Observation Identifier Names and Codes | | 0‑L | 11639-2 | [#] Births.term | Logical Observation Identifier Names and Codes | | 0‑L | 11640-0 | [#] Births total | Logical Observation Identifier Names and Codes | | 0‑L | 11612-9 | [#] Abortions | Logical Observation Identifier Names and Codes | | 0‑L | 11613-7 | [#] Abortions.induced | Logical Observation Identifier Names and Codes | | 0‑L | 11614-5 | [#] Abortions.spontaneous | Logical Observation Identifier Names and Codes | | 0‑L | 33065-4 | [#] Ectopic pregnancy | Logical Observation Identifier Names and Codes |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Specimen Type
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.56 | Effective Date | 2024‑08‑02 10:55:07 |
|---|
| Canonical URI | http://hl7.org/uv/ips/fhir/ValueSet/specimen-type |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPS_SpecimenType | Display Name | IPS Specimen Type |
|---|
| Description | IPS Specimen Type codes value set. |
|---|
| Copyright | This artefact includes content from SNOMED Clinical Terms® (SNOMED CT®) which is copyright of the International Health Terminology Standards Development Organisation (IHTSDO). Implementers of these artefacts must have the appropriate SNOMED CT Affiliate license - for more information contact http://www.snomed.org/snomed-ct/getsnomed-ct or info@snomed.org. |
|---|
| Usage: 2 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-30 | IPS Specimen Collection (STU2) | DYNAMIC | | hl7ips-entry-template-30 | IPS Specimen Collection (STU1) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Intensional Definition | Code System |
|---|
| |  Include | descendants of code 123038009 Specimen | SNOMED Clinical Terms |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Pregnancy Expected Delivery Date Method
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.72 | Effective Date | 2024‑08‑02 09:41:07 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSPregnancyEDDMethod | Display Name | IPS Pregnancy Expected Delivery Date Method |
|---|
| Description | IPS Expected Delivery Date Method |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-29 | IPS Pregnancy Expected Delivery Date Observation (STU2) | DYNAMIC |
|
|
| Source Code System | 2.16.840.1.113883.6.1 - Logical Observation Identifier Names and Codes - FHIR: http://loinc.org - HL7 V2: LN |
|---|
| Level/ Type | Code | Display Name | Code System |
|---|
| 0‑L | 11778-8 | Delivery date Estimated | Logical Observation Identifier Names and Codes | | 0‑L | 11779-6 | Delivery date Estimated from last menstrual period | Logical Observation Identifier Names and Codes | | 0‑L | 11780-4 | Delivery date Estimated from ovulation date | Logical Observation Identifier Names and Codes |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
ActStatusActiveCompletedAbortedSuspended
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.12 | Effective Date | 2017‑03‑30 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | ActStatusCodeActiveCompletedAbortedSuspended | Display Name | ActStatusActiveCompletedAbortedSuspended |
|---|
| Usage: 4 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-4 | IPS Medication Statement (STU1) | DYNAMIC | | hl7ips-entry-template-4 | IPS Medication Statement (2021) | DYNAMIC | | hl7ips-entry-template-33 | IPS Subordinate SubstanceAdministration (STU1) | STATIC | | hl7ips-entry-template-4 | IPS Medication Statement (STU2) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name | Code System |
|---|
| 0‑L | completed | Completed | ActStatus | | 0‑L | aborted | Aborted | ActStatus | | 0‑L | active | Active | ActStatus | | 0‑L | suspended | Suspended | ActStatus |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
ActStatusActiveCompletedAbortedCancelled
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.22 | Effective Date | 2017‑05‑02 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | ActStatusActiveCompletedAbortedCancelled | Display Name | ActStatusActiveCompletedAbortedCancelled |
|---|
| Usage: 4 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-17 | IPS Procedure Entry (2021) | DYNAMIC | | hl7ips-entry-template-17 | IPS Procedure Entry (STU1) | DYNAMIC | | hl7ips-entry-template-17 | IPS Procedure Entry (STU2) | DYNAMIC | | hl7ips-entry-template-38 | IPS Planned Procedure (TI-2020) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name | Code System |
|---|
| 0‑L | completed | Completed | ActStatus | | 0‑L | active | Active | ActStatus | | 0‑L | aborted | Aborted | ActStatus | | 0‑L | cancelled | Cancelled | ActStatus |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Medicine Route of Administration
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.33 | Effective Date | 2024‑08‑02 10:12:19 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/medicine-route-of-administration |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSRouteOfAdmin | Display Name | IPS Medicine Route of Administration |
|---|
| Description | EDQM (European Directorate for the Quality of Medicines and Healthcare) Route of Administration codes. This Value Set includes all the EDQM Standard Terms having:
[Concept Status] = ‘Current’ AND
[Concept Class] = ‘ROA’ AND
[Domain] = ‘Human and Veterinary’ ROA = ‘Route of administration’ |
|
| Usage: 3 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-4 | IPS Medication Statement (STU1) | DYNAMIC | | hl7ips-entry-template-4 | IPS Medication Statement (2021) | DYNAMIC | | hl7ips-entry-template-4 | IPS Medication Statement (STU2) | DYNAMIC |
|
|
A valid code from the code system:| Code System Name | Code System Id | EDQM | 0.4.0.127.0.16.1.1.2.1 |
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
Problem Severity
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.18 | Effective Date | 2017‑04‑07 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/condition-severity-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSProblemSeverity | Display Name | Problem Severity |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-25 | IPS Severity Observation (STU1) | DYNAMIC |
|
|
| Source Code System | 2.16.840.1.113883.6.1 - Logical Observation Identifier Names and Codes - FHIR: http://loinc.org - HL7 V2: LN |
|---|
| Level/ Type | Code | Display Name | Code System |
|---|
| 0‑L | LA6752-5 | Mild | Logical Observation Identifier Names and Codes | | 0‑L | LA6751-7 | Moderate | Logical Observation Identifier Names and Codes | | 0‑L | LA6750-9 | Severe | Logical Observation Identifier Names and Codes |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Problem Type
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.16 | Effective Date | 2017‑04‑07 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/problem-type-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSProblemType | Display Name | IPS Problem Type |
|---|
| Description | This value set indicates the level of medical judgment used to determine the existence of a problem. |
|---|
| Usage: 5 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-8 | IPS Problem Entry (2021) | DYNAMIC | | hl7ips-entry-template-8 | IPS Problem Entry (2021) | DYNAMIC | | hl7ips-entry-template-8 | IPS Problem Entry (STU1) | DYNAMIC | | hl7ips-entry-template-6 | IPS Reaction Manifestation (STU1) | STATIC | | hl7ips-entry-template-6 | IPS Reaction Manifestation (STU2) | STATIC |
|
|
| Source Code System | 2.16.840.1.113883.6.1 - Logical Observation Identifier Names and Codes - FHIR: http://loinc.org - HL7 V2: LN |
|---|
| Level/ Type | Code | Display Name | Code System |
|---|
| 0‑L | 75326-9 | Problem | Logical Observation Identifier Names and Codes |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
DRUGActCode
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.14 | Effective Date | 2017‑04‑01 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | DRUGActCode | Display Name | DRUGActCode |
|---|
| Usage: 3 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-4 | IPS Medication Statement (STU1) | DYNAMIC | | hl7ips-entry-template-4 | IPS Medication Statement (2021) | DYNAMIC | | hl7ips-entry-template-4 | IPS Medication Statement (STU2) | DYNAMIC |
|
|
| Source Code System | |
|---|
| Level/ Type | Code | Display Name | Code System |
|---|
| 0‑L | DRUG | Drug therapy | Act Code |
|
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
Medicine Strength Denominator
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.31 | Effective Date | 2017‑05‑03 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/strengthDenominator |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSMedicineStrengthDenominator | Display Name | Medicine Strength Denominator |
|---|
| Usage: 2 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-3 | IPS Manufactured Material (STU1) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU2) | DYNAMIC |
|
|
A valid code from the code system:| Code System Name | Code System Id | Unified Code for Units of Measure | 2.16.840.1.113883.6.8 |
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
Topographical modifier (qualifier)
| Id | 2.16.840.1.113883.11.22.58 | Effective Date | 2017‑12‑12 19:33:52 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | topographicalModifierQualifier | Display Name | Topographical modifier (qualifier) |
|---|
| Description | << 106233006 |Topographical modifier (qualifier value)|
|
|---|
| Usage: 2 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-12 | IPS Radiology Result Observation (STU2) | DYNAMIC | | hl7ips-entry-template-12 | IPS Radiology Result Observation (STU1) | DYNAMIC |
|
|
IPS Results Organizer
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.37 | Effective Date | 2017‑05‑23 |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSResultsOrganizer | Display Name | IPS Results Organizer |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-9 | IPS Result Organizer (STU1) | DYNAMIC |
|
|
A valid code from the code system:| Code System Name | Code System Id | Logical Observation Identifier Names and Codes | 2.16.840.1.113883.6.1 |
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
Medicine Strength Numerator
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.30 | Effective Date | 2017‑05‑03 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/strengthNumerator |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSStrengthNumerator | Display Name | Medicine Strength Numerator |
|---|
| Usage: 2 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-3 | IPS Manufactured Material (STU1) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU2) | DYNAMIC |
|
|
A valid code from the code system:| Code System Name | Code System Id | Unified Code for Units of Measure | 2.16.840.1.113883.6.8 |
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS WHO ATC
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.29 | Effective Date | 2017‑05‑03 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/whoatc-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | WHOATC | Display Name | IPS WHO ATC |
|---|
| Usage: 3 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-3 | IPS Manufactured Material (STU1) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (2021) | DYNAMIC | | hl7ips-entry-template-3 | IPS Manufactured Material (STU2) | DYNAMIC |
|
|
A valid code from the code system:| Code System Name | Code System Id | WHO Anatomical Therapeutic Chemical classification | 2.16.840.1.113883.6.73 |
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
IPS Results Observation
| This terminology is a snapshot as of . Terminologies may evolve over time. If you need recent (dynamic) versions of this terminology, please retrieve it from the source. |
| Id | 2.16.840.1.113883.11.22.38 | Effective Date | 2017‑05‑23 |
|---|
| Canonical URI | http://hl7.org/fhir/uv/ips/ValueSet/observation-codes-uv-ips |
|---|
| Status |  Draft Draft | Version Label | |
|---|
| Name | IPSResultsObservation | Display Name | IPS Results Observation |
|---|
| Usage: 1 | | Id | Name | Type |
|---|
| Template |
|---|
| hl7ips-entry-template-10 | IPS Result Observation (STU1) | DYNAMIC |
|
|
A valid code from the code system:| Code System Name | Code System Id | Logical Observation Identifier Names and Codes | 2.16.840.1.113883.6.1 |
|
| Legenda: Type L=leaf, S=specializable, A=abstract, D=deprecated. NullFlavor OTH (other) suggests text in originalText. HL7 V3: NullFlavors to appear in @nullFlavor attribute instead of @code. |
List of all artifacts used in this guide
CDA Templates
Document
Section
Entry
Other
CDA Template References (Pharmacy)
CDA Template References (IHE)
- 1.3.6.1.4.1.19376.1.5.3.1.1.5.3.2 IHE Coded Vital Signs Section
- 1.3.6.1.4.1.19376.1.5.3.1.4.13.1 Vital Signs Organizer
Unconstrained Templates from the original CDA specification
- 2.16.840.1.113883.10.12.151 CDA Organization
- 2.16.840.1.113883.10.12.152 CDA Person
- 2.16.840.1.113883.10.12.153 CDA AssignedEntity
- 2.16.840.1.113883.10.12.318 CDA Author (Body)
- 2.16.840.1.113883.10.12.315 CDA Device
- 2.16.840.1.113883.10.12.319 CDA Informant (Body)
- 2.16.840.1.113883.10.12.323 CDA Performer (Body)
- 2.16.840.1.113883.10.12.313 CDA PlayingEntity
- 2.16.840.1.113883.10.12.316 CDA RelatedEntity
Value Sets
Value Sets References
- 2.16.840.1.113883.1.11.1 AdministrativeGender
- 2.16.840.1.113883.1.11.10706 Timing Event
- 2.16.840.1.113883.1.11.11610 x_ActRelationshipDocument
- 2.16.840.1.113883.1.11.15933 ActStatus
- 2.16.840.1.113883.1.11.16926 HL7 BasicConfidentialityKind
- 2.16.840.1.113883.1.11.19446 x_ActRelationshipEntry
- 2.16.840.1.113883.1.11.19563 PersonalRelationshipRoleType
- 2.16.840.1.113883.1.11.19601 x_ServiceEventPerformer
- 2.16.840.1.113883.1.11.19709 ActSubstanceAdministrationImmunizationCode
- 2.16.840.1.113883.1.11.19890 x_ActStatusActiveComplete
- 2.16.840.1.113883.1.11.201 TelecommunicationAddressUse
- 2.16.840.1.113883.1.11.20386 SeverityObservationCode
- 2.16.840.1.113883.1.11.78 Observation Interpretation
- 2.16.840.1.113883.11.20.9.18 MoodCodeEvnInt
- 2.16.840.1.113883.11.20.9.33 INDRoleclassCodes
- 2.16.840.1.113883.3.88.12.80.60 Social History Type
Appendix (Informative)
Acronyms and abbreviations
-
CCD: Continuity of Care Document
-
C-CDA: Consolidated CDA
-
CDA: Clinical Document Architecture
-
CEN: Comité Européen de Normalisation (European Committee for Standardization)
-
CEN/TC 251 : CEN Technical Committe 251
-
DSTU: Draft Standard for Trial Use
-
EC: European Commission
-
EDQM: European Directorate for the Quality of Medicines & Healthcare
-
eHDSI: Digital Service Infrastructure for eHealth
-
eHN: eHealth Network
-
EHR: Electronic Healthcare Record
-
EN: European Normative [or Standard] (CEN)
-
epSOS: European Patient Smart Open Services
-
EU: European; Europe
-
FDA: Food and Drug Administration (USA)
-
GP: General Practitioner
-
HL7: Health Level Seven
-
HP: Healthcare Professional
-
IDMP: IDentification of Medicinal Products (ISO Standard)
-
IHE: Integrating the Healthcare Enterprise
-
INTERPAS: International Patient Summary (HL7 International Project)
-
IPS: International Patient Summary
-
ISO: International Organization for Standardization
-
JAseHN: Joint Action to Support the eHealth Network
-
JIC: Joint Initiative Council on SDO Global Health Informatics Standardization
-
LOINC: Logical Observation Identifiers Names & Codes
-
MOU: Memorandum of Understanding (on cooperation surrounding health related information and communication technologies, that between EU and US)
-
MPID: Medicinal Product Identifier
-
ONC: Office of the National Coordinator for Health Information Technology (USA)
-
PCC: Patient Care Coordination
-
PCID : Medicinal Product Package Identifier
-
PhPID(s): Pharmaceutical Product Identifier(s)
-
prEN: Draft European Normative [or Standard] (CEN)
-
prTS: Draft Technical Specifications (CEN)
-
PS: Patient Summary
-
S&I: Standards and Interoperability (S&I) Framework (run by ONC)
-
SAIF: Service Aware Interoperability Framework
-
SDO: Standards Developing Organization
-
STU: Standard for Trial Use
-
TS: Technical Specifications (CEN)
-
UCUM: Unified Code for Units of Measure
-
WHO: World Health Organization
Glossary
-
Compliance. A standard or specification is compliant with another standard or specification if all propositions that are true in the initial standard are also true in the complying standard. The target artifact is compliant with the source artifact if and only if all conforming implementations of the target are also conforming with the source (RM-ODP).
The term compliance is also used to state expectations as to how certain specifications need to satisfy possible legislative or regulatory constraints or requirements.
-
Conformance relates an implementation to a standard. Any proposition that is true of the specification must be true in its implementation. (ISO, 2010).
-
Conformance Assessment is a process whereby a given implementation instance is evaluated to determine which of its various Conformance Assertions are valid implementations of a given specification’s Conformance Statements.
-
Conformance Statement is a statement that identifies testable requirements at a specified Conformance Point within a specification, explicitly defining the behavior which must be satisfied at these points. Conformance Statements will only occur in standard which are intended to constrain some feature of a real implementation, so that there exists, in
principle, the possibility of testing.
-
Conformance Assertion is a testable, verifiable statement made about a specific implementation instance against a corresponding Conformance Statement.
-
Conformance Points are the evaluation of conformance at specific points in the implementation or specification. See Conformance.
-
Electronic Patient Summary: electronic health record extract containing essential healthcare information intended for specific uses . (EN ISO 13940: 2016)
-
International Patient Summary : electronic patient summary for use in the unscheduled, cross-border care scenario comprising at least the required elements of the IPS dataset.
-
International Patient Summary dataset: a minimal and non-exhaustive patient summary dataset, specialty-agnostic, condition-independent, but readily usable by clinicians for the cross-border unscheduled care of a patient.
Real World User Stories
This section reports a series of real world user stories adapted from the Trillium Bridge project [The Trillium Bridge Project http://www.trilliumbridge.eu] and the eHDSI initiative [The eHDSI initiative https://ec.europa.eu/cefdigital/wiki/display/EHOPERATIONS/eHealth+DSI+Operations+Home].
IPS Storyboard 1: Martha, a traveling corporate executive
Martha, a 45-year old corporate executive and breast cancer survivor travels frequently on business between the US and EU countries. She carries a clinical summary on her mobile phone and on paper just in case she needs to seek medical care regarding recurring symptoms. Martha’s summary includes
-
Breast cancer Stage II with no evidence of recurrence following treatment
-
hot flashes as problems
-
Anastrozole 1 mg. once daily
-
Black Cohosh Extract herbal supplement as medications
-
the indication of an allergy to Penicillin
-
and finally as Plan of Care, to continue hormone medication with Anastrozole for total of 5 years
-
and monitor for potential breast cancer recurrence.
During a visit in Austria, Martha walks up a hill and experiences shortness of breath, faints, and wakes up a few minutes later after hitting her head on a stone step. A passerby helps her get to the emergency department of a local hospital. An ambulance is called and she is brought to the emergency ward.
During registration and admission, Martha hands in her patient summary in a USB key. At the hospital, Martha is evaluated by an oncologist and a cardiologist.
Following care provision, Martha receives an encounter report. When back home she hands in the encounter report to her primary physician, who updates her record.
IPS Storyboard 2: Paolo, a retired businessman
Paolo Cerruti is a 67-year-old retired businessman, who normally lives in the outskirts Bergamo, near Lake Como, in Lombardy. He is generally healthy, but has long-standing hypertension. His regular physician changed his medication two weeks ago because of poor blood pressure control on his previous medication. He is on holiday going through New England, US, travelling on his
own to enjoy the autumn foliage, and is presently in Boston, MA. He is nearing the end of his holiday, and will be returning to Italy in three days’ time. Two days ago he lost his day bag. The bag included his hypertension medication, and he has not been able to take his tablets for two days.
This morning he has woken up feeling dizzy and has blurred vision. The hotel is able to put him in urgent contact with a local general practitioner (GP). Having assessed him, the GP noted a raised blood pressure, but is uncertain about whether to attribute these symptoms to the raised blood pressure or a side effect of the new medication. Now, the GP in Boston needs to know the
medication, and the past few blood pressure readings to determine how exceptional the present reading is and manage Paolo appropriately.
Immediate access to his International Patient Summary would be the perfect answer. Paolo may retrieve his online European Patient Summary for emergency access that is retrieved, transformed into an IPS and shown its content translated in English.
The GP notes that visual disturbances are a recognized side effect of this medication. No specific treatment is indicated, and Paolo is reassured that side effects will gradually subside, and his GP can prescribe a suitable antihypertensive medication upon his return to Lake Como.
IPS Storyboard 3: Diana, a pregnant woman
Diana is a 34-year-old pregnant woman from Lisbon with a past medical history of allergic asthma and thyroid cancer during adolescence; for the latter she had a surgical procedure done (thyroidectomy) and, as a consequence, suffers hypothyroidism which requires hormone replacement for life (levothyroxine). At the age of 31 she was diagnosed with a hereditary cardiac disorder –
Brugada Syndrome – and had a cardioverter defibrillator implanted to control potentially lethal arrhythmias.
During the pregnancy of her first child (C-section delivery), she suffered gestational diabetes that developed into type 2 diabetes after giving birth and needs now to receive subcutaneous insulin. As chronic treatment she also needs nebulizations three-time per day for her asthma - this condition is aggravated in her case by being a smoker (1 pack per day) as included in the
Social History Section.
At this moment, she presents severe pre-eclampsia (hypertension during pregnancy) in treatment with two oral antihypertensive agents (a combination medication). Additionally, she is following a 14-day-course of antibiotic treatment due to an acute pyelonephritis (kidney infection more likely to be develop in pregnant women due to the physiological changes that may interfere with
the flow of urine).
Other sections of her Summary include allergies to latex and kiwi (which are very often associated) and to aspirin, and intolerance to lactose; immunizations administered during childhood and adolescence are also present.
Although being real choices for the different diseases and conditions, the selection of the patient's current medication tries to present some easily described medication as well as not so easily ones: e.g. insulin degludec, amoxicilin+clavulanic acid, and the combination of ipratropium bromide+salbutamol for nebulization. For the oral treatment of the pre-eclampsia the agents
selected would not be used in real practice during pregnancy.
Integrated examples
The IPS specification releases are published at hl7intl.art-decor.org the International Patient Summary Project Publication Page [International Patient Summary Project Publication Page https://hl7intl.art-decor.pub/index.php?prefix=hl7ips-]. The actual release has a link to
the XML materials that the W3C schemas are part of; it also includes example CDA document instances. A set of use cases have been defined and represented in IPS format. Also multiple languages are covered.
It is likely that the publication site will move to hl7.org permanently, and we will inform the community about that process.
Validation artifacts
You can test your implementation (instances) against the IPS specification. To download materials to your computer for local testing and validation consider...
-
...the W3C schemas (actually valid for any CDA specification) located at the Publication Page [https://hl7intl.art-decor.pub/index.php?prefix=hl7ips-]. The actual release has a link to the XML materials as which the W3C schemas are part of; it also includes example CDA
document instances.
-
..the ISO schematron, automatically generated by the tool. These are files to do validation locally by associating IPS CDA instances with the main schematron using an XML editor or to use the derived XSLT conversions and apply the according XSLT derivation to your local IPS CDA instance.
For further information you can follow the documentation.
Operational information
-
The IPS project has an official mailing list address ips(at)lists.hl7.org, hosted at the HL7 listserver. Visit your Listserv Subscriptions at hl7.org and subscribe to the International Patient Summary (IPS)that is summarised under the Structured
Documents Work Group.
-
The original specification is hosted on the logical ART-DECOR main server art-decor.org under the Governance Group
HL7 International, the project is reachable at the Live Project Landing Page.
-
Any IPS specification release in HTML format resides at the Publication Page [https://hl7intl.art-decor.pub/index.php?prefix=hl7ips-]. It is likely that the publication site will move to hl7.org permanently, we will inform about that process.
-
The IPS specification on the wiki is hosted here (international-patient-summary.net). It is likely that the publication site will move to hl7.org permanently, we will inform about that process.
Licenses
Following is a non-exhaustive list of third-party terminologies that may require a separate license:
-
SNOMED CT: SNOMED International (formerly know as International Healthcare Terminology Standards Development Organization IHTSDO, see Get SNOMED CT http://www.ihtsdo.org/snomed-ct/get-snomed-ct) or info@ihtsdo.org
-
Logical Observation Identifiers Names & Codes (LOINC): This material contains content from LOINC® (http://loinc.org). The LOINC Table, LOINC Table Core, LOINC Panels and Forms File, LOINC Answer File, LOINC Part File, LOINC Group File, LOINC Document Ontology File, LOINC Hierarchies,
LOINC Linguistic Variants File, LOINC/RSNA Radiology Playbook, and LOINC/IEEE Medical Device Code Mapping Table are copyright © 1995-2017, Regenstrief Institute, Inc. and the Logical Observation Identifiers Names and Codes (LOINC) Committee and is available at no cost under the license at http://loinc.org/license.
-
Unified Code for Units of Measure (UCUM) : Regenstrief Institute, Inc. and the UCUM Organization
-
EDQM Standard Terms : European Directorate for the Quality of Medicines & Healthcare (EDQM, see EDQM Standard Terms https://standardterms.edqm.eu).
FAQ’s
This is a placeholder for future Frequently Asked Questions about the International Patient Summary.
How to read the table view for templates
The template definitions are shown in a table view. It is comprised of Template Meta data and the Template Design. For further information please refer to the HL7 Templates Standard: Specification and Use of Reusable Information Constraint Templates, Release 1[1].
Templates may also be included in the hierarchical graph view (often used for CDA), see below.
Template Meta data
The upper right part of the template table contains the template meta data. Template id, status and the template name are shown (1). Furthermore the Version (effective date), a possible version label and the display name are shown (2).
The description area (plain or an accordion) contains the template descriptions/purpose (3), followed by classifications and whether the template is defined as open or closed (4).
The usage part (5) may list templates that uses this template or what templates this templates uses. A relationship list (6) may show all relationships to other templates or models.
Examples may show the correct use of the template by an XML fragment (7).
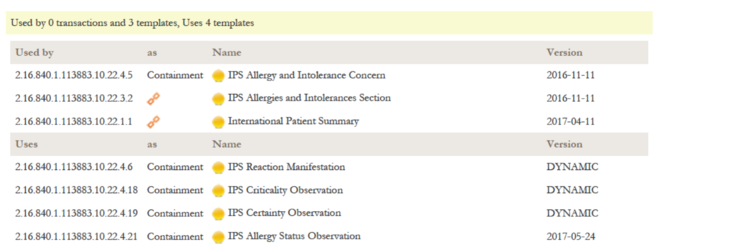
The relationship list shows all relationships to other templates or models for this template. It is divided in the "Used by" part listing templates that make use of this template, and a "Uses" listing all templates that are used by this templates, either as inclusion or containment. Indirect relationships like the parent Document Level Template for a Section Level Template are marked with a chain symbol.
The PDF version is rendered in the same way, but maybe with different fonts etc. to fit customized publication requirements.
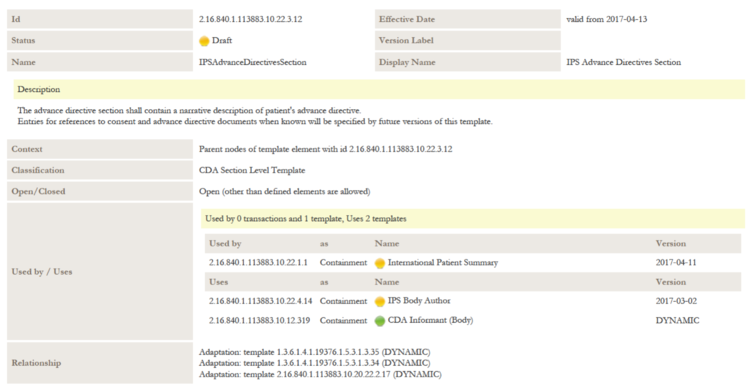
Table view of Template Design

The headings of the table view of a template design are:
Item (1) contains the XML document tree view of all elements and attributes specified in the template design. Elements are denoted by a preceding triangle and attributes by a preceding "@".
DT (2) data types, contains the data type of the item, for more information on valid data types for element and attributes (see [1]).
Card / Conf (3) cardinality (Card) and conformance (Conf) of the item.
Cardinality is the usual notion of min and max occurrences of the element. For attributes 0..1 denotes optionality, 1..1 say that the attribute is required and NP denotes prohibited attributes.
Conformance may display values as shown in the following table.
Values of the conformance column
| Conf |
Short |
Description
|
| O |
optional |
Data is truly optional
|
| R |
required |
If data is present and not masked (e.g. for privacy reasons), it must be provided, otherwise it may be omitted or explicitly null flavored.
Sender and receiver must support this element.
|
| M |
mandatory |
The data must be populated with a valid value from the associated value domain, otherwise the instance is not valid and may not be communicated.
Sender and receiver must support this element.
|
| C |
conditional |
There are conditions when data has to be provided (e.g. co-constraints like "information about pregnancy IF the patient is "female".
Sender and receiver must support this element.
|
| F |
fixed |
The data has a fixed value.
|
| NP |
not permitted |
Data shall not be present
|
Description (4) contains a textual description of the item, may also contain constraints and values for fixed attributes.
Label (5) is a human readable label that is displayed upon errors, warnings or notes during validation.
Details of the table view
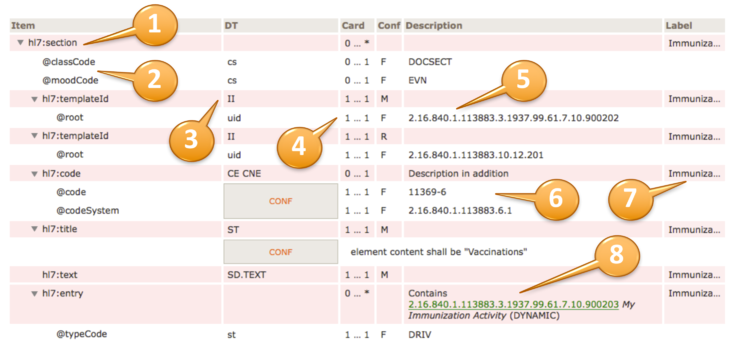
The actual template design shows the XML structure in a hierarchical list of elements (items) that are typically prefixed by the namespace "hl7:" or "cda:" (1).
Elements are denoted with a triangle, attributes with an @ sign (2).
Data types are specified according to the list of supported data types (3). They may be simple data types (lowercase), regular data types (uppercase) or flavors thereof.
In case of coded elements, the coding strength (Required/CNE, Extensible/CWE, Preferred or Example) can be highlighted near the datatype (e.g. “CD.IPS (Extensible/CWE)”) ; the absence of indications about the strength (e.g. “CE.IPS”) shall be interpreted as “Required/CNE”.
Values of the coding strength column
| Strength |
Displayed as |
Description
|
| Required |
Required/CNE |
Coded with no exceptions; this element SHALL be from the specified value set
|
| Extensible |
Extensible/CWE |
Coded with Exceptions; this element SHALL be from the specified value set if any of the codes within the value set can apply to the concept being communicated. If the value set does not cover the concept (based on human review), alternate codings (or, data type allowing, text) may be included instead.
|
| Preferred |
Preferred |
Instances are encouraged to draw from the specified codes for interoperability purposes but are not required to do so to be considered conformant.
|
| Example |
Example |
Instances are not expected or even encouraged to draw from the specified value set. The value set merely provides examples of the types of concepts intended to be included.
|
The cardinality and conformance column is explained above (4).
Fixed values for e.g. attributes are also shown in the "description" column (5), preceded by a "F" in the Conf column.
Conformance statements are shown together with a CONF box, e.g. a @code and a @codeSystem with fixed and required values (6).
An optional label is displayed at the rightmost column (7).
Inclusion or containments of other templates, e.g. an entry within a section, are shown accordingly (8) along with their template id, display name and flexibility/stability indication, i.e. "DYNAMIC" (the most recent version) or a STATIC binding together with a version date.

Choices of elements are shown as a choice list with the elements in questions summarised in a bullet point list.

A typical Conformance Statement is the binding of a coded element to a value set. This is expressed in the way shown. The value set is represented with the id, display name and the flexibility/stability of the binding.

In case a constraint is expressed in words, a box "Constraint" accompanies the textual expression of the constraint.

In cases where constraints are expressed by formalised rules in ISO Schematron, the rule along with the role (error, warning), the test and the assertion message is shown.
How to read the Templates hierarchical graph view

Templates are often included in the hierarchical graph view (often used for CDA). It gives an overview of e.g. section and entries and their nesting/relationships.

In case a template has more that one type (CDA Person for header, section and entry templates), it is denoted with a *, if a recursive definition is detected, this is shown with the symbol @.
How to read the where criteria
Templates sometimes include criteria for identifying distinct elements from a list (e.g. in a choice).
The criteria used to identify the items are shown in square brackets using the assertion where [ criteria ]
Criteria can be:
- an xpath expression as in the example : where [hl7:low or hl7:high]
- or an integer indexing the items of the list: e.g. where [1]; where [2]
- ↑ 1.0 1.1 Cite error: Invalid
<ref> tag;
no text was provided for refs named teits